| Issue |
Aquat. Living Resour.
Volume 35, 2022
Topical Issue - NORA 4 - Native Oyster Restoration Alliance 4th Conference
|
|
|---|---|---|
| Article Number | 5 | |
| Number of page(s) | 13 | |
| DOI | https://doi.org/10.1051/alr/2022005 | |
| Published online | 20 May 2022 | |
Research Article
Molecular identification, life cycle characterization, and hatchery seed production of dwarf oysters from the Ebro Delta (Spain)
1
IRTA-Sant Carles de la Ràpita. Ctra. Poble Nou Km 5.5, Sant Carles de la Ràpita, 43540 Tarragona, Spain
2
Institute of Environment and Marine Science Research (IMEDMAR-UCV), Universidad Católica de Valencia SVM,C/Explanada del Puerto S/n, Calpe, 03710 Alicante, Spain
3
Instituto de Acuicultura Torre de la Sal, Consejo Superior de Investigaciones Científicas, 12595 Ribera de Cabanes (Castellón), Spain
4
University of Naples Federico II, Department of Biology, Corso Umberto I 40, 80138 Naples, Italy
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Pauline Kamermans
Received:
23
December
2021
Accepted:
11
March
2022
Abstract
Dwarf oysters in the Ebro Delta are only observed growing on local pen shells, Pinna nobilis L., currently under extinction risk. The species identification of these populations is uncertain, given recent genetic classifications of dwarf oysters of Ostrea stentina species complex. Hence, the first objective of this study was to confirm the identity of dwarf oysters associated to P. nobilis in the Ebro Delta. Then, we aimed to assess the viability of hatchery and nursery production of O. stentina for potential conservation programs using diets based on a mix of live microalgae species or the commercial Shellfish Diet 1800®. Our results showed that COI sequences obtained were specific to O. stentina. Significant differences in the growth and survival of larvae between diet treatments were observed in 3 days. For the live diet, the larval period lasted from 15 to 22 days (299.2 ± 4 µm) whereas it took up to 36 days using the Shellfish Diet 1800® (280 ± 7.2 µm), with an overall mortality of 72.3 vs. 99.3%, respectively for each diet. Besides, no seed survival was observed for the commercial diet after a 10-day-fixation attempt. In contrast, ca. 16% of the individuals on the live diet that were allowed to settle reached the juvenile stage and could be released to the environment. Histological examination of obtained seed did not present any sign of disease and showed gonadal development for both sexes at ca. 6 months of age (16.1–19.1 mm). The growth curve obtained was fitted to a Schnute-Richards growth model which returned ages of up to 10 years for maximum local sizes of 45 mm. We conclude that although the Shellfish Diet 1800® is not an adequate diet for O. stentina, its hatchery production is feasible and might provide support to future conservation actions.
Key words: Ostrea stentina / DNA barcoding / Shellfish Diet 1800® / microalgae diets / Schnute-Richards growth equation / age at first maturity
© P. Prado et al., Published by EDP Sciences 2022
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Oysters are widely distributed marine engineer species, capable of building reef habitat for entire ecosystems (Lenihan and Peterson, 1998) and providing services of great ecological and economical value including fisheries resources (Beck et al., 2011), effective attenuation against wave action and shoreline erosion (Meyer et al., 1997) and reduction of coastal eutrophication thanks to their ability to filter large volumes of water (Cerco and Noel, 2007; Carmichael et al., 2012). Unfortunately, over-harvesting, and pollution have contributed to globally reducing natural oyster habitats by as much as 85 percent over the last two centuries (Beck et al., 2011). Besides, different oysters’ populations and species have been also severely affected by a range of parasitic, bacterial, and viral diseases, possibly enhanced by exposure to environmental stressors (Lenihan et al., 1999) and/or transfers of nonnative oysters for aquaculture and from ballast waters (Brenner et al., 2014). Among proposed solutions, a popular one has been the construction of artificial reefs to promote larval settlement on empty oyster shells or different man-made substrates (Goelz et al., 2020). Yet, most of such restoration efforts have been focused on enhancing oyster populations targeted by seafood markets to continue their exploitation (Coen and Luckenbach, 2000). In doing this, the conservation of other oyster species without commercial interest has been neglected. In addition, most of the hatchery production of bivalves is also focused on satisfying the demands of species of commercial interest (Helm et al., 2004), even when aimed at restoring natural banks to mitigate overharvesting (e.g., Loor et al., 2016; Lodeiros et al., 2016). To our knowledge, the only non-commercial bivalve species for which still infructuous efforts have been conducted to develop viable hatchery and nursery protocols is the critically endangered Mediterranean fan mussel, Pinna nobilis Linnaeus, 1758 (Trigos et al., 2018). Another key issue for restoration is the availability of local natural populations (Brumbaugh and Coen, 2009), whose shortage could potentially constrict hatchery production since a minimum genetic diversity is necessary in order to obtain viable populations in the long-term (Hughes et al., 2019).
The ostreid genus Ostrea Linnaeus, 1758 typically includes sequential hermaphrodites, viviparous species maintaining larvae within a brood chamber from days to weeks before releasing the swarm into the water column (Gray et al., 2019). Aside some economically important species such as O. edulis, O. chilensis and O. angasi, the genus also contains several less known and closely related species of dwarf oysters in the Ostrea stentina/equestris/aupouria complex (Hu et al., 2019). In particular, the O. stentina group appears to feature a geographic distribution across the Eastern Atlantic (from Morocco to the Iberian Peninsula) and Southern/Southwestern Mediterranean coast (Lapègue et al., 2006; Hu et al., 2019), although genetic confirmation is needed for most of the range. The species has been suggested to be a protandric hermaphrodite starting gametogenic development in February–March for females and in September for males (Ben Salah et al., 2012). It dwells on intertidal and shallow subtidal rocks and gravel beds, or aggregated and low lying in the mudbank within marine and estuarine environments (Richards and Vita-Finzi, 1982; González-Wangüemert et al., 2004; Lapègue et al., 2006) and reach maximum sizes of ca. 4.5 cm (Rosique et al., 1995). In the Catalonian and Balearic region, fossil records of so called ‘O. stentina’ range back to the Miocene and Pleistocene epochs (De Porta, 1987; Cuerda et al., 1989). In biological records from more recent periods, they have also been mentioned in numerous studies (e.g., Aguilar-Amat, 1935; Altimira et al., 1981; Bonnin and Rodrígez-Babío, 1990, Tarruella and López, 2006), but no population studies have been conducted, possibly because of the small size and lack of commercial value compared to other exploited species of oyster such as Ostrea edulis Linnaeus, 1758 or Crassostrea gigas Thunberg, 1793 (Beck et al., 2011). Yet, they might have an important role in the food web of marine ecosystems, enhance the availability of microhabitats, and contribute to positive effects on water quality. Hence, there is a need to overcome this overall lack of information on the genetic identity of dwarf oysters in order to assess possible alterations in the abundance of populations and associated ecological functions and allow addressing possible conservation efforts.
The same difficulties arise in Alfacs Bay (south Ebro Delta), where there is a historic record of the species (Brunet and Capdevila, 2005), but no population or genetic studies have been conducted. Currently, the presence of different native species of oysters, including O. stentina Payraudeau, 1826, O. edulis or Anomia ephippium Linnaeus, 1758, appears to be relegated to few isolated individuals settled on the spare hard substrate available within local seagrass beds of Cymodocea nodosa (Ucria) Ascherson 1870, but frequently they appear attached to the large shells of the Mediterranean fan mussel, Pinna nobilis which is currently in risk of extinction due to the expansion of the disease caused by Haplosporidium pinnae (García-March et al., 2020). Previously to the disease, the pen shell population of the Alfacs Bay had been indicated as the second largest in the Mediterranean Sea, with over 90,000 individuals (Prado et al., 2014). Currently, the spread of the disease appears to be constrained by the salinity gradient induced by freshwater discharges from the local rice field agriculture (Prado et al., 2021a). Yet, a large part of the population has experienced mass mortality, thus compromising the presence of available substrate for the settlement of other organisms, including oysters. Present low abundances might be additionally influenced by the same confluence of factors such as pollution and disease also contributing to oyster reef decline at the global scale (Lenihan et al., 1999; Beck et al., 2011; Brenner et al., 2014), as well as to the local presence of the invasive blue crab, Callinectes sapidus Rathbun, 1896, which was first detected in the Alfacs Bay in 2012 (Castejón and Guerao, 2013) and currently supports a growing local fishery (López and Rodon 2018). According to a recent study conducted with local commercial bivalves (Crassostrea gigas and Mytilus galloprovincialis Lamarck, 1819), only oysters larger than 5–7 cm are relatively protected from predation (Prado et al., 2020a), although experiments were conducted with detached animals, which ease manipulability compared to wild individuals (Lin, 1991).
In this context, given the current risk for the persistence of local dwarf oysters populations in the Ebro Delta, the main objectives of this study were: (1) to conduct a phylogenetic assessment to determine the genetic identity of local individuals relative to other dwarf oyster species within the Ostrea genus (Hu et al., 2019) and (2) to document the life cycle of the species and evaluate the viability of developing hatchery and nursery production techniques using different feeding methods. To this end, two different diets were tested on produced larvae, one based on live microalgae cultured in situ within experimental facilities, and other based on a popular commercial brand, Shellfish Diet 1800®, which has been successfully used in other species of bivalves such as the eastern oyster Crassostrea virginica Gmelin, 1791, the giant clam, Tridacna noae Röding, 1798, and the winged pearl oyster, Pteria penguin Röding, 1798 larvae (Rikard and Walton, 2012; Southgate et al., 2016; Wassnig and Southgate 2016; Southgate et al., 2017). An assessment of health conditions was also performed on grown seed to determine whether individuals could be safely introduced in the wild. Finally, size increments were monitored for several months in order to have an estimation of the time required to attain maximum sizes observed in the field.
2 Materials and methods
2.1 Broodstock collection and conditioning
Broodstock collection was conducted on August the 19th 2020 on the southern shore of Alfacs Bay (south Ebro Delta), at a site hosting one of the few remaining populations of the Mediterranean fan mussel, Pinna nobilis, which provides substrate for the settlement of other bivalve species. Adult individuals (N = 2, ca. 45 mm in length) similar in appearance to O. stentina were transported to IRTA facilities attached to one of the pen shells (Fig. 1), which was also captured as a part of a reproductive program to preserve the species (Pinna spat project). The pen shell and the two attached oysters were kept in an aerated 350 L truncated cone tank at 22 °C within an acclimatized room with 50% daily seawater renovation filtered and sterilized as for larval cultures (see later). Individuals were maintained with a daily dose of 15 X 104 cells/mL of Tisochrysis lutea El M. Bendif and I. Probert 2013 (T-ISO), 105 cells/mL of Chaetoceros neogracilis VanLandingham 1968, and 25 X 103 cells/mL of Tetraselmis suecica (Kylin) Butcher 1959. The strains were bought from the University of Santiago de Compostela and had the identification codes: ECC007 (T. lutea, 4–5 µm), ECC022 (C. neogracilis, 3–5 X 6–8 µm), and EC039 (T. suecica, 7–9 X 15–17 µm).
 |
Fig. 1 Close up of a P. nobilis shell showing two attached adult individuals of O. stentina used for maturation. |
2.2 Molecular identification of the oysters
A DNA barcoding strategy was used to identify the oyster species. DNA from 4 offspring from the two captive adults was extracted from a piece of fresh mantle tissue using the EZNA-Mollusc DNA extraction kit. A fragment of ca. 700 bp of the COI gene was amplified with the universal primers HC02198 (5’-taaacttcagggtgaccaaaaaatca-3’) and LCO1490 (5’-ggtcaacaaatcataaagatattgg-3’) (Folmer et al., 1994). PCR was carried out in 20 µl reactions with a final concentration of 2.5 ng/µl of template DNA, 0.2 mM of each dNTP, 1 µM of each primer, 1.5 mM of MgCl2, and 0.025 U/µl of Taq DNA Polymerase (Invitrogen) in the buffer supplied by the manufacturer. After 4 min at 95 °C, the PCR was subjected to 35 cycles of 1 min at 95 °C, 30 s at 56 °C, and 30 s at 72 °C, followed by a final 3 min step at 72 °C. PCR products were purified with PureLink Quick PCR Purification Kit (Invitrogen) and Sanger sequencing was carried out in both directions by the genomics service of the University of Valencia (Spain), using an ABI 3730XL automated sequencer and the same primers used for PCR amplification.
The sequences obtained were edited with Geneious Prime® 2019.2.3 and aligned with Clustal W 2.1 (Thompson et al., 1994) as implemented in Genious Prime. Seventeen additional COI sequences from different Ostrea species were downloaded from the GenBank database and added to the previous sequences in order to conduct a phylogenetic analysis with the software MEGA X (Kumar et al., 2018). These sequences were chosen among those used by Hu et al (2019) to obtain a set of representative sequences of the different Ostrea taxa recognized in that study. Maximum likelihood composite (MLC) distances (Tamura et al., 2004) were estimated between pairs of sequences and the resulting matrix was used for the construction of a neighbour-joining (NJ) tree (Saitou and Nei, 1987). The statistical support of the tree nodes was estimated by a bootstrap test (Felsenstein 1985) based on 1000 replicates.
2.3 Larval and seed culture
Once broodstock released the D-veliger larvae, water was filtered through a 40 µm sieve and subsamples counted under a stereomicroscope. A total of 700,000 larvae were obtained from one or both individuals (production of larvae was initially unintended but they were kept and cared for when they were released) attached to the pen shell: half of them (350,000 ± 14,530 D-veliger larvae) were fed the same three live microalgae that were used for broodstock conditioning, since these species are permanently produced at IRTA facilities. Mixed diets with different species of microalgae, including some of larger size such as T. suecica, have been used in other studies assessing the survival of veliger larvae in other species (Helm, 1977; Neo et al., 2013; Aranda-Burgos et al., 2013) and were considered as suitable for a first dietary assay with larvae of ca. 132 µm. The other half of the larvae were fed the Shellfish Diet 1800®, which is a mix of five marine microalgae: Isochrysis sp., Pavlova sp., Tetraselmis sp., Thalassiosira weissdlogii, and T. pseudonana (https://reedmariculture.com/products/shellfish-diet). D-veliger larvae were distributed in smaller 100 L truncated cone tanks at an initial density of 3.5 larvae mL–1, resulting in only one tank per diet treatment. Although this fact poses the limitation of the absence of replicates for diet treatments, both tanks were located immediately adjacent to each other and exposed to the same conditions. Larvae and seed cultures were grown using filtered seawater through 1 µm and active C and irradiated with UV light (FSW) to minimize bacterial growth. Tanks were aerated and subjected to water change every 48 h. Temperature conditions were 22 °C and seawater salinity in our facilities ranged between 35 and 37 psu throughout the study period. For the live diet, microalgae cultures were prepared using the same filtered and irradiated seawater that was used for larval culture, under constant aeration and exposure to fluorescent light (47–67 μmol photons m2 s–1). In addition, seawater used to start microalgae cultures for larval feeding was autoclaved in round 5 L flasks for 20 min at 120 °C to achieve further sterilization. The cultures were maintained in a thermostatic chamber at 22 °C illuminated with cool-white fluorescent tubes at light intensity of 150–200 µmol photons m–2 s–1 (Heinz Walz GmbH Universal Light Meter – ULM-500) under a 12 light:12 dark photoperiod, continuous mix of air and CO2, and f/2 medium. During the first 10 days, larvae were feed every two days following each water change, with a total of 80,000 cells mL–1 at a ratio of 4 T. lutea: 2.5 C. neogracilis: 1.5 T. suecica, and then raised to 100.000 cells mL–1 (5:3:2 ratio) until the pediveliger stage. The Shellfish Diet 1800® consist of a mix of several marine microalgae: Isochrysis sp., Pavlova sp., Tetraselmis sp., Chaetoceros calcitrans, Thalassiosira weissfloggi, and Thalassiosira pseudonana. The diet was supplied in increasing daily doses following the instructions in the manufacturer web page (https://reedmariculture.com/collections/bivalve-hatchery-feeds). Both diets were supplied with a continuous feeding protocol, using a peristaltic pump controlled by a cycle timer that delivered the microalgae to larval rearing tanks every 60 min.
Once the postlarval stage was reached, production was transferred to a wider 100 L truncated cone tank in a closed, water recirculated system equipped with three settlement PVC cylinders with 150 µm mesh bottom and individual airlift systems. Each cylinder contained a fine layer of 300–500 µm sand made of crushed oyster shells.
Individuals on the live diet were fed with 120,000 cells mL–1 (5:3:2 ratio) for 12 days, and then the feed dose increased to 180,000 cells mL–1 for another 6 days, both at 48 h intervals. Then, seed started to be fed at increasing daily rates (10 days: 120,000 cells mL–1; 7 days: 150,000 cells mL–1; 7 days: 200,000cells mL–1; 12 days: 250,000cells mL–1; and 12 days: 300,000cells mL–1). For the Shellfish Diet 1800® doses were adjusted for size increases, following the same calculations described for the larval period, but stabilized when no growth was detected.
Remaining individuals that reached a size of ca. 5 mm (early November), were transferred to a larger 3000 L tank with an open-water system equipped with a 500 µm mesh bottom, PVC trays, with an airlift system connected to an automatic pumping system that delivered microalgae mix every 15 min. In mid-March 2021, ca. 25% of the produced seed (the maximum that we were able to process) were carefully cemented to concrete construction blocks (ca. 60 × 25 × 35 cm) and experimentally released in Alfacs Bay, beside a mixed seagrass meadow (Cymodocea nodosa and Zostera noltii Hornem), located in front of IRTA facilities, to further assess survival and growth. The remaining 75% was released into the same area or in Alfacs bay, where broodstock were captured, but without attachment to a hard substrate to allow dispersion by currents into nearby areas. Only a small contingent of ca. 200 individuals were kept under vivarium conditions (open-water circuit) for a pre-fattening phase with supplied daily rations of ca. 4% of body weight (1:3 T-ISO and Tetraselmis suecica).
2.4 Assessment of larval and seed sizes and number of surviving individuals
At each water change, a sample of larvae (N ≥ 20 individuals) under each dietary condition was measured to the nearest µm from photographs taken with a camera coupled to a NIKON Eclipse TE2000-S microscope and to a computer featuring the Analysis 5.1 software for image processing. Later at early seed stages, individuals were measured to the nearest 0.1 mm with a NIKON SMZ 1500 stereomicroscope every 2–7 days. Finally, when individuals reached a manipulative size and transferred to the 3000 L tank, they were measured to the nearest mm once a month using vernier calipers.
The number of surviving larvae was counted from 1 mL aliquots (N = 3) taken from a 2 L measuring cylinder containing the homogenized larval pool at each water change. Once they were allowed to settle to sand particles, settlement success was assessed by counting the proportion of attached seed within randomly collected samples under the stereomicroscope. This was done on 8 occasions over a 20-day period in order to incorporate temporal variability due to the possible death of individuals during the settlement period.
2.5 Histological examination of produced seed
Twenty animals ranging from ca. 1.9 to 1.4 cm were collected once from the vivarium facilities at IRTA in February 2021, removed from the shell for fixation in Davidson solution, and preserved for at least 48 h at room temperature. Subsequently, they were dehydrated in ascendant alcohol series and embedded in paraffin blocks, 3 µm thick sections were cut with a Rotary Microtome (Bioptica, Italy) and stained with Carazzi’s haematoxylin and eosin (HandE) and special stain Pas-BA pH 2.5 for detection of acid mucins (Mazzi, 1977). The slides were examined under a light microscope (Zeiss Axioscope) to assess the overall condition of the animal and possible presence of pathogens and diseases. Special attention was also paid to the presence of gonads and gametes in order to assess the age (and sex) at first maturity of the species. Samples for determining age at first maturity were collected during the first possible period of gonad development during the winter period and were not repeated because animals were already found to be mature.
2.6 Statistical analyses
Possible temporal differences in the size of individuals associated to the effect of diet were investigated with an independent samples two-tailed t-test for each sampling date. Similarly, differences in patterns of survival (i.e., estimated number of larvae in the tank) between the two diet treatments were also investigated with an independent samples two-tailed t-test but given the lower number of replicates per sampling date (N = 3 counts of each diet culture per date), all experimental period was considered together in order to increase the power of the test and obtain a more precise estimate. Student’s t-tests were conducted in IBM SPSS Statistics v. 23.
The measured shell lengths of O. stentina across days of life in larval benthic stages were used to estimate Schnute-Richards growth function (Schnute and Richards, 1990). The Schnute–Richards model is a five-parameter model, given by the equation L(t) = L8(1+ δe−ktv )1/γ; where L(t) is oyster mean shell length at age t, L∞ is the asymptotic shell length, δ, ν, and γ are dimensionless parameters, and K is a relative growth rate parameter with units days–ν. Growth equation were fitted with R software version 4.1.1 through the minpack.lm 1.2-1 package.
3 Results
3.1 Molecular identification
Sequences of ca. 586 base pairs in length were recovered after editing the Sanger sequenced COI fragments. The four sequences were deposited with accessions OL404965-OL404968 in the GenBank sequence database and showed no polymorphism.
The alignment with the 17 Ostrea sequences downloaded from the GenBank database was over 650 bp in length, but only a fragment of 513 sites common to all sequences was used for phylogenetic inference (Fig. 2). The sequences obtained in this study joined other 4 sequences that belonged to the taxon Ostrea stentina, which were originally obtained by Lapègue et al. (2006) and Pejovic et al. (2016) from Atlantic localities in Morocco and northern Spain, respectively. The five sequences together formed a cluster with 100% bootstrap support.
 |
Fig. 2 Neighbor joining (NJ) tree based on Maximum Composite Likelihood (MCL) distances showing the phylogenetic affinity of the Ostrea sp. specimens used for this study (Ostrea sp-IRTA - Ebro Delta) to Ostrea stentina. Numbers near nodes give bootstrap support (%). The inclusion in the different groups of dwarf oysters proposed by Hu et al. (2019) is indicated. Groups 1 and 2 are O. equestris and O. aupouria from close but divergent populations from the Americas and Asian Pacific, respectively. Group 3 is a new species Ostrea neostentina sp. nov. found in southeastern Spain (Mar Menor lagoon), eastern Tunisia, Hong Kong, and Japan, and group 4 is O. stentina occurring in northern (Avilés in the Atlantic Ocean) and northeastern Spain (Alfacs Bay in the Mediterranean Sea), Portugal, Morocco, and northern Tunisia. |
3.2 Dietary effects on larval size and survival
D-veliger larvae of O. stentina were swarmed by broodstock at an initial size of 132 ± 1.1 µm (Fig. 3A) two days after their arrival to our facilities and at a water temperature of 22 °C.
Results from t-test evidenced significant differences in larval size between the two tested diets only two days after the start of the culture, with the magnitude of differences increasing dramatically through time (Fig. 3A; Tab. 1). For larvae fed with the live diet, most individuals settled at 302.8 ± 3.2 µm in a total of 15 days, although a small proportion was allowed to grow for an additional week (Fig. 3A). In contrast, larvae under the Shellfish Diet 1800® took over a month to reach a size of only 280 ± 7.2 µm which was taken to attempt fixation (Fig. 3A).
There were also significant differences in the survival rates of individuals between dietary treatments throughout the larval period (Student’s t = –2.01, tcritic = 2.00, df = 52, P = 0.040) (Fig. 3B). Individuals under the live diet showed an overall mortality during the entire larval period of 72.3% whereas mortality for individuals under the Shellfish Diet 1800® was considerably higher and reached rates of 99.3% by the end of the pelagic larval period (Fig. 3B).
 |
Fig. 3 (A) Larval size (first 15 days of life) of individuals fed with live microalgae vs. those fed the Shellfish Diet 1800®. For the live diet, the size of a group of larvae that remained in the tank while the majority was taken to fixation is also indicated. (B) Temporal changes in the number of larvae in of O. stentina feed with each type of diet. Error bars are mean standard error. |
Results of two-tailed t-test for independent samples testing for differences between larval growth with the live microalgae diet and the Shellfish Diet 1800® at each experimental date. Significant differences are indicated in bold.
3.3 Larval and post-larval development
Larvae under the live diet lost their D shape in 4 days (Fig. 4A), started to show the umbo in 6 days (of 140.5 ± 1.1; Fig. 4B), and all of them were umbonate 8 to 11 days after release (size range 167.1 ± 1.6 µm to 231.6 ± 4.5 µm; Fig. 4C–D). During days 13 to 15th of life, larvae progressively increased in size until the pediveliger stage was reached (from 266.6 ± 4.7 to 302.8 ± 3.2 µm; Fig. 4E–F). At this size, some of the larvae started to show an “eyespot”, the umbo leaned asymmetrically towards one side of the shell (Fig. 4F), and at times displayed the foot as they swam. Once in the settlement cylinders, individuals under the live diet increased their size from ca. 0.3 to 4.9–5.5 mm during the two first months (early September to early November) (Fig. 5A; Fig. 6A), with a rate of settlement success consistently above 70% (Fig. 5B). In contrast, for individuals under the Shellfish Diet 1800®, no survival was observed after 10 days in the settlement cylinders. During the following two months (December to February), the seed under the live diet increased in size by 2.6-fold (Fig. 5A), and individuals started to attain juvenile traits such as a pattern of superimposed dark and clear bands on the right flat valve (Fig. 6B). Seed abundance counted on the 22nd of December 2020 yielded an estimate of 20,593 individuals. On the 19th of March 2021 individuals measured an average of 17.5 ± 0.7 mm and the estimated number was 15,900 individuals, ca. 16% of the initial seed that were allowed to settle.
Individuals were introduced to a site close to IRTA facilities in the northern coast of the Alfacs Bay in March 2021 (3 days after biometric measures) but died ca. 2 months after. The site was chosen for logistic reasons but showed excess sedimentation over the concrete blocks that resulted on burial and anoxia. Surviving individuals during those two months (23rd of May and 26th of June 2021) yielded sizes of 18.2 ± 0.2 mm (N = 100) and 20.1 ± 0.2 mm (N = 31), respectively. Some additional animals (ca. 200 individuals) were, however, kept under vivarium conditions and random measures conducted on November the 11th, 2021 (N = 39 individuals), yielded sizes 21.1 ± 0.6 mm. The survival rate of these individuals was 88.5% by February 2022.
The resulting Schnute-Richards growth equation between the measured shell length of individuals across days of life until November 2021 was:  . This results in age estimates of ca. 1.6 years to reach 30 mm, 3.5 years to reach 40 mm and up to 9.6 years for the theoretical maximum of 45 mm reported for the species.
. This results in age estimates of ca. 1.6 years to reach 30 mm, 3.5 years to reach 40 mm and up to 9.6 years for the theoretical maximum of 45 mm reported for the species.
 |
Fig. 4 Complete larval development of O. stentina raised with the live diet. (A) Veliger larvae with 4 days of life; (B) Larvae with 6 days of life starting to show the umbo (um); (C and D) umbonate larvae of 8 to 11 days of life; (E) early pediveliger (13 days of life); and (F) pediveliger stage, with some of the larvae showing an eyespot (ey) and the umbo leaning asymmetrically towards one side of the shell. |
 |
Fig. 5 (A) Seed size of O. stentina raised with the live diet in the three settlement cylinders. The settlement period is indicated in gray and corresponds to the time frame indicated in panel 5B. Larvae in cylinder 3 were set to settle 1 week later than those in cylinders 1–2. (B) Rates of settlement during the first 23 days of benthic growth. The 70% success is indicated with a dashed line. Error bars are mean standard error. |
 |
Fig. 6 (A) Seed of ca. 5 mm size in early November and (B) seed of ca. 14 mm size in early January already showing some juvenile traits such as a banded dark and clear pattern on the right flat valve. |
3.4 Histological examination of produced seed
Light microscopy of the seed showed that young individuals of only 6 months of age were already sexually reproductive. Examined individuals showed hermaphroditism with alternating sex at the moment of sampling in February 2021. These individuals featured an apparent primary female development, with the ripe female gonad showing mainly vitellogenic oocytes attached to the follicle wall; at the same time, a developing male gonad was represented by growing follicles filled with germinative cells and spermatogonia present around its periphery (Fig. 7A and B). The adjacent digestive tissue showed a well-developed epithelium, indicative of an adequate feeding condition necessary for active growth and maturation (Fig. 7C). Overall, no evidence of a disease condition (i.e., presence of pathogens, and/or inflammatory responses) was observed. In fact, histological sections revealed an optimum development of the first line of immune defense constituted by mucous and granular cells in the mantle epithelium (Fig. 7D).
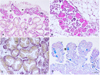 |
Fig. 7 Histology of different tissue of O. stentina: (A and B) visible hermaphroditic gonad with ripe female phase and developing male phase; vitellogenic ovocytes (Ov) and spermatogonia (Spg) are indicated; (C) digestive gland (DG) show a thick adsorbing epithelium sign of an active feeding; (D) Details of mantle epithelium (E) showing well developed mucous cells stained with acid alcian Blue Pas (black arrow) and eosinophilic granular cells (white arrow) part of the oyster first line of defense. CT: connective tissue. |
4 Discussion
Our results confirm the presence of Ostrea stentina – according to the genetic classification of Hu et al. (2019) – in the Ebro Delta (Alfacs Bay). This finding contrast with results for dwarf oysters in the Mar Menor, the closest location (ca. 400 km) for which there is genetic information, which were classified as the new species Ostrea neostentina sp. nov. (Hu et al., 2019). This mismatch highlights the lack of knowledge on genetically verified geographic distributions and possible specific habitat requirements of these two Mediterranean species included within the O. stentina complex (Hu et al., 2019). According to current information, the O. stentina species (group 4 from Hu et al., 2019) has been found in the Western (Alfacs Bay, Spain) and Southern Mediterranean Sea (Bizerte Lagoon, Tunisia), as well as in the Eastern Atlantic (from Dakhla Bay in Morocco to Sado and Mira estuaries in Portugal, and Avilés in the Bay of Biscay in Northern Spain). Yet, the finding of O. stentina in the Cantabrian Sea has been pointed as a non-indigenous species arrival associated with intense maritime traffic and disturbed environmental conditions (Pejovic et al., 2016; Rech et al., 2018). In former genetic research by Lapègue et al. (2006), authors suggested historical native presence range along the African Atlantic and Southern Mediterranean coasts but also include the northeastern coast of the Iberian Peninsula. Other works in the Catalonian and Balearic region based only on morphological descriptions (Aguilar-Amat, 1935; Altimira et al., 1981; De Porta, 1987; Cuerda et al., 1989; Bonnin and Rodrígez-Babío, 1990, Tarruella and López, 2006) also reported the presence of the species in the area, which is now genetically confirmed by our study. Regarding environmental conditions, genetically verified natural sites with O. stentina include sheltered bays with slightly brackish to marine salinity ranges and steady to strongly seasonal fluctuations in seawater temperature (El Asri et al., 2019, this study); and Atlantic estuaries with large freshwater supply and strong spatial gradients in salinity (Ré, 1996), thus suggesting a wide plasticity in the physiological requirements. Even so, the species has not been yet genetically confirmed from hypersaline lagoons such as the Mar Menor.
Larvae obtained from broodstock attached to collected pen shell specimens were swarmed at an initial size of 132 ± 1.1 µm, which is in the lower range of that reported for other species in the genus, including O. permollis (108–127 µm), O. edulis (180–190 µm), O. lurida (169–185 µm), and O. chilensis (416–521 µm) (Buroker, 1985) and might be partly due to variability in the incubation period (from 3–9 days to 8 weeks, depending on the species; see Gray et al., 2019 and references therein). From that starting point, significant differences in the growth and survival of larvae fed live microalgae vs. those fed the Shellfish Diet 1800® diet become apparent only 3 days after the start of the experiment. The total duration of the larval period ranged from only 15 to 22 days (299.2 ± 4 µm) using the live diet to 36 days (280 ± 7.2 µm) using the commercial diet, with an overall mortality of 72.3 vs. 99.3% by the end of the hatchery period, respectively for each diet. Although the robustness of our results is limited due to the lack of adequate replication, the conditions of both tanks were almost identical: they were positioned next to each other, got the water from the same source, and they were manipulated by the same person all the time. Therefore, it would be highly improbable that a potential pathogen would have developed in one tank and not in the other, although the possibility cannot be fully discarded. This makes the different diet provided to each tank the most reasonable explanation for the observed differences in growth and survival between tanks. Our findings contrast with those reported in other hatchery studies with bivalve larvae and suggest that the efficiency of the product could be partly dependent on the target species. For instance, Southgate et al. (2016) reported successful hatchery production of the winged pearl oyster, Pteria penguin, using Instant Algae® (“Isochrysis 1800®” and “Pavlova 1800®”) with ca. 4.6% of the larvae surviving to fixation (a total of 700,000 eyed pediveligers obtained), although no simultaneous comparisons were conducted with a traditional diet based on live microalgae. In another study, larvae of Tridacna noae were shown to ingest microalgae species from commercial concentrates using epifluorescence microscopy and displayed subsequent growth (Southgate et al., 2017). Authors also observed that veliger larvae displayed a distinctive temporal digestion of microalgae species, but this effect should not be responsible fot the observed hatchery failure with O. stentina, since the product used (Shellfish Diet 1800®) consists of a mix of up to six different species. An alternative explanation might be the lack of a preliminary adjustment of the feeding ration for the species, as indicated for P. penguin larvae (Wassnig and Southgate, 2016). The diet was supplied following the manufacturer instructions, which in turn were based on FAO protocols for C. gigas and has been also successfully applied in C. virginica (Rikard and Walton, 2012). In fact, there is an abundant literature supporting the efficiency of Shellfish Diet 1800® in C. virginica in hatchery and nursery arenas as well as in adult conditioning (e.g., Rolton et al., 2016; Kroll et al., 2016; Pruett et al., 2021, among others). Conversely, other species of bivalves have been shown to experience deficient physiological performance with commercial microalgae concentrates that support our present results. For instance, in recent experiments with the Manila clam conducted at IRTA with the same experimental diets and enhanced levels of replication (N = 3 and repeated three times) similar mortalities and decreased growth rates were obtained with the Shellfish Diet 1800® (unpublished results). Hernandis et al., (submitted), compared scope for growth (SFG) in juvenile Pinna rudis Linnaeus, 1758 fed with Shellfish Diet 1800®, live cultured Isochrysis galbana Parke, 1949, and a 50/50% mix diet and found a negative energy balance (–5.67 J g h–1), indicative of highly stressful conditions, that suggest lower acceptability and digestibility. Similarly, Prado et al. (2020b, 2021b) used different species microalgae concentrates (Easy Reefs, Fitoplancton Marino) to feed juvenile P. nobilis and found 85% lower growth than in controls based on live microalgae, along with almost double mortality rate associated to severe nutritional impairment and depletion of internal reserves. The reasons behind the failure of commercial concentrates in some bivalve species are unclear. Hernandis et al., (submitted) performed analyses on cultured and commercial microalgal cells and showed that both had a similar organic content, but ash content was higher in commercial (19%) than in laboratory cultured microalgae (5.3%) which might result in decreased digestibility. It is also possible that the compounds used in commercial products to keep low bacterial loads and trigger flocculation (Knuckey et al., 2006; Camacho-Rodríguez et al., 2016) are not well tolerated by all bivalve species, thus causing detrimental side effects on the assimilation efficiency that might cascade to reduced growth and enhanced mortality. The very few individuals from the Shellfish Diet 1800® treatment that survived to reach a size of ca. 280 µm (ca. 0.7%) and were transferred to fixation in settlement cylinders showed no survival after 10 days.
When live phytoplankton was used for hatchery production, larvae took 15 to 21 days in reaching the pediveliger stage for benthic settlement. This planktonic period appears to be highly variable across the Ostrea genus: 0–2 days in O. chilensis, 6–14 days in O. edulis, 10–23 days in O. lurida, and 30–33 in O. permollis (see review by Buroker, 1985) and might integrate species differences in growth rate and initial larval size, and the effect of environmental conditions. Larval survival with the investigated live diet (27.71%) could be considered low compared with studies showing values from ca. 50 to >90% in O. edulis (e.g., Araya et al., 2012; Robert et al., 2017; González-Araya and Robert, 2018). In particular, the highest survival rates of up to 98% were obtained for larvae fed with a mixed diet of C. neogracilis and T. lutea (1:1 cell volume) and maintained throughout the planktonic life at 25 °C using a flow-through rearing system (Robert et al., 2017). The progressive development of hatchery production techniques for this species since the late 1930s (Colsoul et al., 2021) suggest that the culture of O. stentina could also be improved using other diet formulations, daily rations, water temperature and/ or rearing system. In addition, since broodstock released larvae only two days after field collection, the potential effects of parental nutrition in progeny are unknown and could have limited larval development (González-Araya and Robert, 2018). By the end of the planktonic period, a total of 97,000 late pediveliger larvae were allowed to settle in early and mid-September, with a mean of ca. 84% experiencing settlement possibly favored by temperature conditions (Robert et al., 2017) or by the presence to some unknown natural inducer such as bacterial biofilms or chemical cues (Mesías-Gansbiller et al., 2013). Yet only ca. 16% of the late pediveliger larvae reached a juvenile stage and were still alive by mid-March 2021, evidencing that nursery production was also below values observed in other studies with O. edulis (ca. 28–40% post-larval yields using mixed diets; Helm, 1977; Araya et al., 2012). The obtained seed showed, however, a healthy condition, as evidenced by the presence of well-developed digestive tissue in histological examinations. Bacterial and protozoan diseases are common bottlenecks in bivalve hatcheries and in wild populations (see review by Paillard et al., 2004), so their absence from the culture opens the door to their potential safe translocation to the natural media. Unfortunately, experimental introduction of juveniles cemented to concrete blocks was not successful due to excess organic matter and siltation from nearby agricultural drainage channels, causing observable burial on anoxic sediment which may lead to infiltration of exogenous bacteria and subsequent mortality (Coffin et al., 2021). Yet, the survival of some individuals for two months coupled with the maintenance of ca. 200 in nursery facilities allowed for fitting to a Schnute-Richards growth equation. Although growth rates might be slightly different in wild populations, this yield estimates of ca. 1.6 years of age for animals of 30 mm, 3.5 years for those of 40 mm, and up to 10 years to attain maximum sizes around 45 mm (Rosique et al., 1995; this study). This estimate of maximum age is coherent with that indicated for O. edulis, which may range from 10 to 14 years and with other species also reaching an age of 15 years or more, depending on preferential harvesting of larger animals (Richardson et al., 1993; Lockwood and Mann, 2019). The obtained growth equation was also similar in shape to that found for O. edulis, with the phase of asymptotic reached at ca. 6 years of age (Richardson et al., 1993, this study). In fact, histological sections showed that individuals of about 20 mm featured hermaphroditism with alternating sex, which is consistent with observations reporting that O. edulis may produce eggs even in the first year of life (see review by Cole, 1942). Our results also showed a primary female development in the examined individuals, which is contrary to the protandric hermaphroditism often described for O. edulis (e.g., Kamphausen et al., 2011; Joyce et al., 2013) and for O. stentina (Salah et al., 2012). Yet, distinctive groups of adult spawners, one first acting as females with considerably larger brood strength, and other acting first as males and producing a lower number of ripened eggs due to partially spent reserves have been also indicated for O. edulis (Cole, 1942). Besides, Maneiro et al. (2017, 2020) observed the presence of simultaneous female and male gametes in the follicles at different maturation stages, suggesting that maturation is not a homogeneous process and that might be influenced by multiple factors including temperature, photoperiod, and nutrition. Hence, the tentative nature of this observations is stressed, since the evaluation of a larger number of individuals appears to be necessary to assess patterns of sex-change in flat oysters, particularly when extrapolating hatchery results to field patterns.
To conclude, our results reveal the presence of O. stentina in Alfacs Bay, which represents the only genetically confirmed location in the Western Mediterranean Sea so far. Individuals found in association with pen shells appear to be an adequate source of broostock, although given the endangered status of this species (García-March et al., 2020) the finding of individuals might become a limiting factor for restoration (Brumbaugh and Coen, 2009). Yet, we were able to grow larvae and obtain healthy spat, thus opening the door to the possible culture of the species for wild release, the best strategy for oyster conservation (Colsoul et al., 2021). Our results point that Shellfish Diet 1800® is not an adequate diet for the hatchery and nursery production of O. stentina despite positive results in other species (Rikard and Walton, 2012; Southgate et al., 2016, 2017, Wassnig and Southgate, 2016). Further research is, however, needed to develop reliable protocols for production, particularly in terms of feeding regime (microalgae species and food ration), water temperature, and the implementation of flowthrough larval rearing systems to enhance growth and survival of larvae and spat to ca. 70% higher rates reported in other species of flat oyster (Robert et al., 2017). The northern coast of Alfacs Bay does not seem to be appropiate for benthic reintroduction given the proximity to agricultural discharge channels, but the southern coast were the broodstock were obtained features a much lower influence of discharges (Prado, 2018), and could provide better results. Since O. stentina does not have a commercial interest, its wild reintroduction could constitute a new paradigm for assessing the efficiency of previous ‘restoration’ efforts (Coen and Luckenbach, 2000). Besides, an enhanced resilience of O. stentina to the disease caused by the protozoan parasite Bonamia exitiosa Hine, Cochennac and Berthe, 2001, has been reported (Hill et al., 2010), which might be an additional trait favouring the effectivity of this oyster species for ecological restoration and the recovery of ecosystem functions and services (Colsoul et al., 2021).
Acknowledgements
Authors are grateful to the Biodiversity Foundation of the Ministry for Ecological Transition and the Demographic Challenge for supporting the Pinna Spat and Recupera Pinna projects in the 2019 and 2020 calls for proposals. P. Prado was contracted under the INIA-CCAA cooperative research program for postdoctoral incorporation from the Spanish National Institute for Agricultural and Food Research and Technology (INIA). Authors would like to thank the students and teachers at the Institute of Professional Aquaculture and Environmental Studies of Catalonia (IEPAAC) for their help cementing juvenile oysters to concrete blocks and assistance with transportation into the field.
References
- Altimira CM, Huelin F, Ros JD. 1981. Mol·luscs bentònics de les Illes Medes (Girona). I. Sistemàtica. Butll Inst Cat Hist Nat 47: 69–75. [Google Scholar]
- Aguilar-Amat JB. 1935. Observacions Malacològiues.XXIII. Més dades malacològiques tarragonines. Butll Inst Cat Hist Nat 35: 77–80. [Google Scholar]
- Aranda-Burgos JA, Da Costa F, Nóvoa S, Ojea J, Martínez-Patiño D. 2014. Embryonic and larval development of Ruditapes decussatus (Bivalvia: Veneridae): a study of the shell differentiation process. J Mollusc Stud 80: 8–16. [CrossRef] [Google Scholar]
- Araya, R G, Mingant, C, Petton, B, Robert, R. 2012. Influence of diet assemblage on Ostrea edulis broodstock conditioning and subsequent larval development. Aquaculture 364: 272–280. [CrossRef] [Google Scholar]
- Beck MW, Brumbaugh RD, Airoldi L, Carranza A, Coen LD, Crawford C, Defeo O, Edgar GJ, Hancock B, Kay MC. 2011. Oyster reefs at risk and recommendations for conservation, restoration, and management. Bioscience 61: 107–116. [CrossRef] [Google Scholar]
- Bonnin J, Rodrígez-Babío C. 1990. Catálogo provisional de los moluscos bivalvos marinos de la plataforma continental de las costas mediterráneas de la Península Ibérica y de las Islas Baleares. Iberus 9: 97–110. [Google Scholar]
- Brenner M, Fraser D, Van Nieuwenhove K, O'Beirn, F, Buck BH, Mazurié J, Thorarinsdottir G, Dolmer P, Sanchez-Mata A, Strand O, Flimlin G, Miossec L, Kamermans P. 2014. Bivalve aquaculture transfers in Atlantic Europe. Part B: environmental impacts of transfer activities. Ocean Coast Manag 89: 139–146. [CrossRef] [Google Scholar]
- Brunet J., Capdevila M. 2005. Atlas malacològic del Delta de l’Ebre. Privately published. pp. 198 [Google Scholar]
- Brumbaugh R.D., Coen L.D. 2009. Contemporary approaches for small-scale oyster reef restoration to address substrate versus recruitment limitation: a review and comments relevant for the Olympia oyster, Ostrea lurida Carpenter 1864. J Shellfish Res 28: 147–161. [CrossRef] [Google Scholar]
- Buroker N.E. 1985. Evolutionary patterns in the family Ostreidae: larviparity vs. oviparity. J Exp Mar Biol Ecol 90: 233–247. [CrossRef] [Google Scholar]
- Camacho-Rodríguez J., Cerón-García M.C., Macías-Sánchez M.D., Fernández-Sevilla J.M., López-Rosales L., Molina-Grima E. 2016. Long-term preservation of concentrated Nannochloropsis gaditana cultures for use in aquaculture. J Appl Phycol 28: 299–312. [CrossRef] [Google Scholar]
- Carmichael R.H., Walton W., Clark H. 2012. Bivalve enhanced nitrogen removal from coastal estuaries. Can J Fish Aquat Sci 69: 1131–1149. [CrossRef] [Google Scholar]
- Castejón D., Guerao G. 2013. A new record of the American blue crab, Callinectes sapidus Rathbun, 1896 (Decapoda: Brachyura: Portunidae), from the Mediterranean coast of the Iberian Peninsula. BioInvasions Rec 2: 141–143. [CrossRef] [Google Scholar]
- Cerco C., Noel M. 2007. Can oyster restoration reverse cultural eutrophication in Chesapeake Bay? Estuar Coasts 30: 331–343. [CrossRef] [Google Scholar]
- Coen L.D., Luckenbach M.W. 2000. Developing success criteria and goals for evaluating oyster reef restoration: ecological function or resource exploitation? Ecol Eng 15: 323–343. [CrossRef] [Google Scholar]
- Cole H.A. 1942. Primary sex-phases in Ostrea edulis. J Cell Sci 2: 317–356. [CrossRef] [Google Scholar]
- Colsoul B., Boudry P., Pérez‐Parallé M.L., Bratoš Cetinić A., Hugh‐Jones, T., et al. 2021. Sustainable large‐scale production of European flat oyster (Ostrea edulis) seed for ecological restoration and aquaculture: a review. Rev Aquacult 13: 1423–1468. [CrossRef] [Google Scholar]
- Coffin M.R., Clements J.C., Comeau L.A., Guyondet T., Maillet M., et al. 2021. The killer within: Endogenous bacteria accelerate oyster mortality during sustained anoxia. Limnol Oceanogr 2885–2900. DOI: 10.1002/lno.11798 [CrossRef] [Google Scholar]
- Cuerda J., Gracia F., Vicens D. 1989. Dos nuevos yacimientos del Pleistoceno superior marino en Porto Colom (Mallorca). Boll Soc Hist Nat Balears 33: 49–66. [Google Scholar]
- de Porta J. 1987. Los Ostreidae en el contexto de la sedimentación del Mioceno marino de la Depresión Prelitoral Catalana (Noroeste de España). Acta Geol Hisp 21: 549–554. [Google Scholar]
- El Asri F., Martin D., Tamsouri M.N., Errhif A., Maanan M., Idrissi M.M., Zidane H. 2019. Spatial and temporal variability in distribution, diversity, and structure of the polychaete assemblages from Dakhla Bay (Atlantic coast of South Morocco). Mar Biodiv 49: 1271–1281. [CrossRef] [Google Scholar]
- Felsenstein J. 1985. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 39: 783–791. [CrossRef] [PubMed] [Google Scholar]
- García-March J.R., Tena J., Henandis S., Vázquez-Luis M., López D., et al., 2020. Can we save a marine species affected by a highly infective, highly lethal, waterborne disease from extinction? Biol Conserv 243: 108498. [CrossRef] [Google Scholar]
- Goelz T., Vogt B., Hartley T. 2020. Alternative substrates used for oyster reef restoration: a review. J Shellfish Res 39: 1–12. [CrossRef] [Google Scholar]
- González-Wangüemert M., Pérez-Ruzafa A., Rosique M.J., Ortiz A. 2004. Genetic differentiation in two cryptic species of Ostreidae, Ostrea edulis (Linnaeus, 1758) and Ostreola stentina (Payraudeau, 1826) in Mar Menor Lagoon, southwestern Mediterranean Sea. Nautilus 118: 103–111. [Google Scholar]
- González-Araya R, Robert, R. 2018. Larval development and fatty acid composition of Ostrea edulis (L.) fed four different single diets from conditioning to pre–settlement. Aquaculture Research 49: 1768–1781. [CrossRef] [Google Scholar]
- Gray M.W., Chaparro O., Huebert K.B., O'Neill S.P., Couture T., Moreira A., Brady D.C. 2019. Life history traits conferring larval resistance against ocean acidification: the case of brooding oysters of the genus Ostrea. J Shellfish Res 38: 751–761. [CrossRef] [MathSciNet] [Google Scholar]
- Helm M.M., Bourne N., Lovatelli A. 2004. Hatchery culture of bivalves. A Practical Manual., FAO Fisheries Technical Paper 471. FAO Publishing, Rome, Italy. pp. 98 [Google Scholar]
- Helm M.M. 1977. Mixed algal feeding of Ostrea edulis larvae with Isochrysis galbana and Tetraselmis suecica. J Mar Biol Assoc UK 57: 1019–1029. [CrossRef] [Google Scholar]
- Hernandis S., Ibarrola I., Tena J., Vázquez-Luis M., García-March J.R., Prado P., Albentosa M. (submitted) SFG (scope for growth) methodology evidence poor nutritional results of commercial microalgal diets in Pinna rudis. Aquat Sci (Ref.: AQSC-D-22-00037). [Google Scholar]
- Hill K.M., Carnegie R.B., Aloui-Bejaoui N., El Gharsalli R., White D.M., Stokes N.A., Burreson E.M. 2010. Observation of a Bonamia sp. infecting the oyster Ostrea stentina in Tunisia, and a consideration of its phylogenetic affinities. J Invert Pathol 103: 179–185. [CrossRef] [Google Scholar]
- Hu L., Wang H., Zhang Z., Li C., Guo X. 2019. Classification of small flat oysters of Ostrea stentina species complex and a new species Ostrea neostentina sp. nov. (Bivalvia: Ostreidae). J Shellfish Res 38: 295–308. [CrossRef] [MathSciNet] [Google Scholar]
- Hughes A.R., Hanley T.C., Byers J.E., Grabowski J.H., McCrudden T., Piehler M.F., Kimbro D.L. 2019. Genetic diversity and phenotypic variation within hatchery‐produced oyster cohorts predict size and success in the field. Ecol Appl 29: e01940. [CrossRef] [PubMed] [Google Scholar]
- Joyce A., Holthuis T.D., Charrier G., Lindegarth S. 2013. Experimental effects of temperature and photoperiod on synchrony of gametogenesis and sex ratio in the European oyster Ostrea edulis (Linnaeus). J Shellfish Res 32: 447–458. [CrossRef] [Google Scholar]
- Kamphausen L., Jensen A., Hawkins L. 2011. Unusually high proportion of males in a collapsing population of commercially fished oysters (Ostrea edulis) in the Solent, United Kingdom. J Shellfish Res 30: 217–223. [CrossRef] [Google Scholar]
- Knuckey R.M., Brown M.R., Robert R., Frampton D.M. 2006. Production of microalgal concentrates by flocculation and their assessment as aquaculture feeds. Aquacult Eng 35: 300–313. [CrossRef] [Google Scholar]
- Kroll I.R., Poray A.K., Puckett B.J., Eggleston D.B., Fodrie F.J. 2016. Environmental effects on elemental signatures in eastern oyster Crassostrea virginica shells: using geochemical tagging to assess population connectivity. Mar Ecol Progr Ser 543: 173–186. [CrossRef] [Google Scholar]
- Kumar S., Stecher G., Li M., Knyaz C., Tamura, K. 2018. MEGA X: molecular evolutionary genetics analysis across computing platforms. Molec Biol Evol 35: 1547–1549. [CrossRef] [PubMed] [Google Scholar]
- Lapègue S., Salah I.B., Batista F.M., Heurtebise S., Neifar L., Boudry P. 2006. Phylogeographic study of the dwarf oyster, Ostreola stentina, from Morocco, Portugal and Tunisia: evidence of a geographic disjunction with the closely related taxa, Ostrea aupouria and Ostreola equestris. Mar Biol 150: 103–110. [CrossRef] [Google Scholar]
- Lenihan H.S., Micheli F., Shelton S.W., Peterson C.H. 1999. The influence of multiple environmental stressors on susceptibility to parasites: an experimental determination with oysters. Limnol Oceanogr 44: 910–924. [CrossRef] [Google Scholar]
- Lenihan H.S., Peterson C.H. 1998. How habitat degradation through fishery disturbance enhances impacts of hypoxia on oyster reefs. Ecol Appl 8: 128–140. [CrossRef] [Google Scholar]
- Lin J. 1991. Predator-prey interactions between blue crabs and ribbed mussels living in clumps. Estuar Coast Shelf Sci 2: 61–69. [CrossRef] [Google Scholar]
- Lockwood R., Mann R. 2019. A conservation palaeobiological perspective on Chesapeake Bay oysters. Phil Trans Royal Soc B 374: 20190209. [CrossRef] [PubMed] [Google Scholar]
- Lodeiros C., Soria G., Valentich-Scott P., Munguía-Vega A., Cabrera J.S., Cudney-Bueno R., Looe A., Márquez A., Sonnenholzner S. 2016. Spondylids of eastern Pacific Ocean. J Shellfish Res 35: 279–294. [CrossRef] [Google Scholar]
- López V., Rodon J. 2018. Diagnosi i situació actual del Cranc Blau (Callinectes sapidus) al delta de l’Ebre. Direcció General de Pesca i Afers Marítims, Generalitat de Catalunya. [Google Scholar]
- Loor A., Ortega D., Lodeiros C., Sonnenholzner S. 2016. Early life cycle and effects of microalgal diets on larval development of the spiny rock–scallop, Spondylus limbatus (Sowerby I.I., 1847). Aquaculture 450: 328–334. [CrossRef] [Google Scholar]
- Maneiro V., Santos Y., Pazos A.J., Silva A., Torres-Corral Y., Sánchez J.L., Pérez-Parallé M.L. 2020. Effects of food ration, water flow rate and bacteriological levels of broodstock on the reproductive conditioning of the European flat oyster (Ostrea edulis, Linnaeus 1758). Aquacut Rep 18: 100412. [CrossRef] [Google Scholar]
- Maneiro V., Silva A., Pazos A.J., Sánchez J.L., Pérez‐Parallé M.L. 2017. Effects of temperature and photoperiod on the conditioning of the flat oyster (Ostrea edulis L.) in autumn. Aquacult Res 48: 4554–4562. [CrossRef] [Google Scholar]
- Mazzi V. 1977. Manuale di tecniche istologiche e istochimiche. Piccin-Nuova Libraria (Ed.), Padova, Italy. [Google Scholar]
- Mesías-Gansbiller C., Silva A., Maneiro V., Pazos A., Sánchez J.L., Pérez-Parallé M.L. 2013. Effects of chemical cues on larval settlement of the flat oyster (Ostrea edulis L.): a hatchery approach. Aquaculture 376: 85–89. [CrossRef] [Google Scholar]
- Meyer D.L., Townsend E.C., Thayer G.W. 1997. Stabilization and erosion control value of oyster cultch for intertidal marsh. Restor Ecol 5: 93–99. [CrossRef] [Google Scholar]
- Neo M.L., Todd P.A., Teo S.L.M., Chou L.M. 2013. The effects of diet, temperature and salinity on survival of larvae of the fluted giant clam, Tridacna squamosa. J Conch 4: 369–376. [Google Scholar]
- Paillard C., Le Roux F., Borrego J.J. 2004. Bacterial disease in marine bivalves, a review of recent studies: trends and evolution. Aquat Liv Res 17: 477–498. [CrossRef] [EDP Sciences] [Google Scholar]
- Pejovic I., Ardura A., Miralles L., Arias A., Borrell Y.J., García-Vázquez E. 2016. DNA barcoding for assessment of exotic molluscs associated with maritime ports in northern Iberia. Mar Biol Res 12: 168–176. [CrossRef] [Google Scholar]
- Prado P. 2018. Seagrass epiphytic assemblages are strong indicators of agricultural discharge but weak indicators of host features. Estuar Coast Shelf Sci 204: 140–148. [CrossRef] [Google Scholar]
- Prado P., Grau A., Catanese G., Cabanes P., Carella F., Fernández-Tejedor M., Andree K.A., Añón T., Hernandis S., Tena J., García-March J.R. 2021a. Pinna nobilis in suboptimal environments are more tolerant to disease but more vulnerable to severe weather phenomena. Mar Environ Res 163: 105220. [CrossRef] [PubMed] [Google Scholar]
- Prado P., Cabanes P., Hernandis S., García-March J.R., Tena J. 2021b. Stable isotope analyses reveal major nutritional deficiencies in captive vs. field juvenile individuals of Pinna nobilis. Mar Environ Res 168: 105304. [CrossRef] [PubMed] [Google Scholar]
- Prado P., Peñas A., Ibáñez C., Cabanes P., Jornet L., Álvarez N., Caiola N. 2020a. Prey size and species preferences in the invasive blue crab, Callinectes sapidus: Potential effects in marine and freshwater ecosystems. Estuar Coastal Shelf Sci 245: 106997. [CrossRef] [Google Scholar]
- Prado P., Cabanes P., Catanese G., Carella F., Carrasco N., Grau A., Hernandis S., García-March J.R., Tena J., Caiola N., Andree K.A. 2020b. Growth of juvenile Pinna nobilis in captivity conditions: Dietary and pathological constraints. Aquaculture 522: 735167. [CrossRef] [Google Scholar]
- Prado P., Caiola N., Ibáñez C. 2014. Habitat use by a large population of Pinna nobilis in shallow waters. Sci Mar 78: 555–565. [CrossRef] [Google Scholar]
- Pruett J.L., Pandelides A.F., Willett K.L., Gochfeld D.J. 2021. Effects of flood-associated stressors on growth and survival of early life stage oysters (Crassostrea virginica). J Exp Mar Biol Ecol 544: 151615. [CrossRef] [Google Scholar]
- Ré P. 1996. Anchovy spawning in Mira estuary (southwestern Portugal). Sci Mar 60: 141–153. [Google Scholar]
- Rech S., Borrell Pichs Y.J., García-Vazquez E. 2018. Anthropogenic marine litter composition in coastal areas may be a predictor of potentially invasive rafting fauna. PloS one 13: e0191859. [CrossRef] [PubMed] [Google Scholar]
- Richards G.W., Vita-Finzi C. 1982. Marine deposits 35,000–25,000 years old in the Chott el Djerid, southern Tunisia. Nature 295: 54–55. [CrossRef] [Google Scholar]
- Richardson C.A., Collis S.A., Ekaratne K., Dare P., Key D. 1993. The age determination and growth rate of the European flat oyster, Ostrea edulis, in British waters determined from acetate peels of umbo growth lines. ICES J Mar Sci 50: 493–500. [CrossRef] [Google Scholar]
- Rikard F.S., Walton W.C. 2012. Use of microalgae concentrates for rearing oyster larvae, Crassostrea virginica. Mississippi–Alabama Sea Grant Publication No.: MASGP-12: 48. [Google Scholar]
- Robert R., Vignier J., Petton B. 2017. Influence of feeding regime and temperature on development and settlement of oyster Ostrea edulis (Linnaeus, 1758) larvae. Aquacult Res 48: 4756–4773. [CrossRef] [Google Scholar]
- Rolton A., Vignier J., Volety A.K., Pierce R.H., Henry M., Shumway S.E., Bricelj M., Hégaret H., Soudant P. 2016. Effects of field and laboratory exposure to the toxic dinoflagellate Karenia brevis on the reproduction of the eastern oyster, Crassostrea virginica, and subsequent development of offspring. Harmful Algae 57: 13–26. [CrossRef] [PubMed] [Google Scholar]
- Rosique M.J., Garcia-Garcia B., Rosique M. 1995. Primera aproximación a la identificación del comportamiento en cultivo de dos especies de ostreidos del Mar Menor. In: Ministerio de Agricultura, Pesca y Alimentación, editor. Actas del V Congreso Nacional de Acuicultura, Cartagena, Murcia. pp. 106–112. [Google Scholar]
- Salah I.B., Bouain A., Neifar L. 2012. Gonadal cycle of the dwarf oyster Ostreola stentina from the south of the Gulf of Hammamet on the eastern coast of Tunisia. Afr J Mar Sci 34: 537–545. [CrossRef] [Google Scholar]
- Saitou N., Nei M. 1987. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol Biol Evol 4: 406–425. [PubMed] [Google Scholar]
- Schnute J.T., Richards L.J. 1990. A unified approach to the analysis of fish growth, maturity, and survivorship data. Can J Fish Aquat 47: 24–40. [CrossRef] [Google Scholar]
- Southgate P.C., Beer A.C., Ngaluafe P. 2016. Hatchery culture of the winged pearl oyster, Pteria penguin, without living micro-algae. Aquaculture 451: 121–124. [CrossRef] [Google Scholar]
- Southgate P.C., Braley R.D., Militz T.A. 2017. Ingestion and digestion of micro-algae concentrates by veliger larvae of the giant clam, Tridacna noae. Aquaculture 473: 443–448. [CrossRef] [Google Scholar]
- Tamura K., Nei M., Kumar S. 2004. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proceed Nat Acad Sci (USA) 101: 11030–11035. [CrossRef] [PubMed] [Google Scholar]
- Tarruella A., López J. 2006. Moluscos marinos del Baix Camp (Tarragona, NE Península Ibérica). Spira 2: 1–16. [Google Scholar]
- Thompson J.D., Higgins D.G., Gibson T.J. 1994. Clustal w: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific-gap penalties and weight matrix choice. Nucleic Acids Res 22: 4673–4680. [CrossRef] [PubMed] [Google Scholar]
- Trigos S., Vicente N., Prado P., Espinós F.J. 2018. Adult spawning and early larval development of the endangered bivalve Pinna nobilis. Aquaculture 483: 102–110. [CrossRef] [Google Scholar]
- Wassnig M., Southgate P. 2016. The effects of stocking density and ration on survival and growth of winged pearl oyster (Pteria penguin) larvae fed commercially available micro-algae concentrates. Aquacult Rep 4: 17–21. [CrossRef] [Google Scholar]
Cite this article as: Prado P, Fernández M, Cordero D, Saavedra C, Carella F, Alcaraz C, Gairin I. 2022. Molecular identification, life cycle characterization, and hatchery seed production of dwarf oysters from the Ebro Delta (Spain). Aquat. Living Resour. 35: 5
All Tables
Results of two-tailed t-test for independent samples testing for differences between larval growth with the live microalgae diet and the Shellfish Diet 1800® at each experimental date. Significant differences are indicated in bold.
All Figures
 |
Fig. 1 Close up of a P. nobilis shell showing two attached adult individuals of O. stentina used for maturation. |
| In the text | |
 |
Fig. 2 Neighbor joining (NJ) tree based on Maximum Composite Likelihood (MCL) distances showing the phylogenetic affinity of the Ostrea sp. specimens used for this study (Ostrea sp-IRTA - Ebro Delta) to Ostrea stentina. Numbers near nodes give bootstrap support (%). The inclusion in the different groups of dwarf oysters proposed by Hu et al. (2019) is indicated. Groups 1 and 2 are O. equestris and O. aupouria from close but divergent populations from the Americas and Asian Pacific, respectively. Group 3 is a new species Ostrea neostentina sp. nov. found in southeastern Spain (Mar Menor lagoon), eastern Tunisia, Hong Kong, and Japan, and group 4 is O. stentina occurring in northern (Avilés in the Atlantic Ocean) and northeastern Spain (Alfacs Bay in the Mediterranean Sea), Portugal, Morocco, and northern Tunisia. |
| In the text | |
 |
Fig. 3 (A) Larval size (first 15 days of life) of individuals fed with live microalgae vs. those fed the Shellfish Diet 1800®. For the live diet, the size of a group of larvae that remained in the tank while the majority was taken to fixation is also indicated. (B) Temporal changes in the number of larvae in of O. stentina feed with each type of diet. Error bars are mean standard error. |
| In the text | |
 |
Fig. 4 Complete larval development of O. stentina raised with the live diet. (A) Veliger larvae with 4 days of life; (B) Larvae with 6 days of life starting to show the umbo (um); (C and D) umbonate larvae of 8 to 11 days of life; (E) early pediveliger (13 days of life); and (F) pediveliger stage, with some of the larvae showing an eyespot (ey) and the umbo leaning asymmetrically towards one side of the shell. |
| In the text | |
 |
Fig. 5 (A) Seed size of O. stentina raised with the live diet in the three settlement cylinders. The settlement period is indicated in gray and corresponds to the time frame indicated in panel 5B. Larvae in cylinder 3 were set to settle 1 week later than those in cylinders 1–2. (B) Rates of settlement during the first 23 days of benthic growth. The 70% success is indicated with a dashed line. Error bars are mean standard error. |
| In the text | |
 |
Fig. 6 (A) Seed of ca. 5 mm size in early November and (B) seed of ca. 14 mm size in early January already showing some juvenile traits such as a banded dark and clear pattern on the right flat valve. |
| In the text | |
 |
Fig. 7 Histology of different tissue of O. stentina: (A and B) visible hermaphroditic gonad with ripe female phase and developing male phase; vitellogenic ovocytes (Ov) and spermatogonia (Spg) are indicated; (C) digestive gland (DG) show a thick adsorbing epithelium sign of an active feeding; (D) Details of mantle epithelium (E) showing well developed mucous cells stained with acid alcian Blue Pas (black arrow) and eosinophilic granular cells (white arrow) part of the oyster first line of defense. CT: connective tissue. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


