| Issue |
Aquat. Living Resour.
Volume 39, 2026
|
|
|---|---|---|
| Article Number | 11 | |
| Number of page(s) | 30 | |
| DOI | https://doi.org/10.1051/alr/2026004 | |
| Published online | 28 April 2026 | |
Research Article
Climate-driven habitat shifts of the vulnerable sea pen Funiculina quadrangularis in the Mediterranean Sea
1
MARBEC, University of Montpellier, CNRS, Ifremer, IRD, Sète, France
2
Institute for Marine Biological Resources and Biotechnology of the National Research Council (IRBIM CNR), Mazara del Vallo, Italy
3
Italian Institute for Environmental Protection and Research (ISPRA), Palermo, Italy
4
Hellenic Centre for Marine Research, Institute of Marine Biological Resources and Inland Waters, Greece
5
Institut de Ciències del Mar, Consejo Superior de Investigaciones Científicas (ICM-CSIC), Passeig Marítim de la Barceloneta 37-49, 16 08003 Barcelona, Spain
6
Fisheries Research Institute (INALE), Hellenic Agricultural Organization - DIMITRA (ELGO - DIMITRA), Nea Peramos, Kavala 18 64007, Greece
7
Department of Environment, Faculty of Environment, Ionian University, Zakynthos, Greece
8
Department of Biosciences, Biotechnology and Environment, University of Bari Aldo Moro, Bari, Italy
9
National Inter-University Consortium for Marine Sciences (CoNISMa), Rome, Italy
10
Department of Life and Environmental Sciences, Polytechnic University of Marche, Via Brecce Bianche 60131 Ancona, Italy
11
Department of Biology, University of Malta, Msida MSD2080, Malta
12
Department of Biological, Geological and Environmental Sciences (BiGeA), Laboratory of Marine Biology and Fisheries, University of Bologna, 61032 Fano, Italy
13
Department of Aquatic Resources, Institute of Marine Research, Swedish University of Agricultural Sciences, 45330 Lysekil, Sweden
14
Ifremer Centre de Méditerranée, Département Océanographie et Dynamique des Ecosystèmes, COAST, 83500 La Seyne-sur-Mer, France
15
Fondazione COISPA, Bari, Italy
16
Institute for Marine Biological Resources and Biotechnology of the National Research Council (IRBIM CNR), Messina, Italy
17
Centre Oceanogràfic de les Balears (COB-IEO), CSIC, Moll de Ponent s/n, 07015 Palma, Illes Balears, Spain
18
Consorzio per il Centro Interuniversitario di Biologia Marina ed Ecologia Applicata G. Bacci, 57128 Livorno, Italy
19
Institute of Marine Biology, University of Montenegro, 85330 Kotor, Montenegro
20
Aquatic Resources Malta, Fort San Lucjan, Triq il-Qajjenza, Marsaxlokk BBG1283, Malta
21
Department of Fisheries and Marine Research, Nicosia, Cyprus
22
Agricultural University of Tirana, Albania
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
7
May
2025
Accepted:
30
March
2026
Abstract
The tall sea pen Funiculina quadrangularis (Pallas, 1766), a widespread octocoral species found in Mediterranean soft-bottom habitats, is recognized by the Food and Agriculture Organization of the United Nations as a vulnerable marine ecosystem indicator due to its sensitivity to bottom trawling. Adapted to stable environments, its distribution could be affected by the combined impacts of climate change, particularly warming bottom waters, and trawling activities. Records from the Mediterranean International Trawl Survey program and other sources were used in an ensemble species distribution modelling approach to predict current habitat suitability and project potential habitat shifts under the intermediate IPCC climate scenario SSP2–4.5 for 2050 and 2100. Results indicated a widespread distribution across the Mediterranean continental shelf and slope, with the species typically found between 100 and 750 m depth (occasionally as shallow as 20 m) and tolerating temperatures between 12°C and 16°C. By 2050, over 80% of the current habitat was projected to remain suitable. However, only 50% were expected to serve as climate refugia by 2100, likely due to warming bottom waters and increased salinity. Some habitat gains were projected (e.g., Ligurian Sea, Corsican waters, deep Eastern Mediterranean), but uncertainties persist regarding the species’ ability to colonize these new areas. Despite its presence in trawled areas, suggesting a relatively lower fishing pressure sensitivity than other VME taxa, significant habitat shifts were projected under future climate conditions. Therefore, it is essential to prioritize conservation efforts in areas projected to remain suitable over time. The climate refugia identified through this study offer critical guidance for future climate-smart VME management plans, helping to ensure the long-term survival of F. quadrangularis in the Mediterranean Sea.
Key words: Megabenthos / climate refugia / conservation management / soft bottoms / SDM / VME
© J. Millot et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Aggregations of marine habitat-forming species, including seagrasses (Duffy, 2006), corals (Davies & Guinotte, 2011; Maynou & Cartes, 2012; Bourque & Demopoulos, 2018; Carbonara et al., 2020), bivalves (Bateman & Bishop, 2017), sponges (Beazley et al., 2013; Maldonado et al., 2015), and polychaetes (Bolam and Fernandes, 2003; Giangrande et al., 2020), support high levels of biodiversity and key diverse ecological processes by increasing habitat complexity, particularly on soft bottoms. Sea pens are octocorals capable of forming extensive aggregations, known as sea pen fields, which enhance the three-dimensional structure of soft-bottom habitats on continental margins and provide shelter and substrate for associated fauna (Hughes, 1998; Buhl-Mortensen et al., 2010; Baillon et al., 2014; De Clippele et al., 2015). However, these erect megabenthic organisms are highly impacted by bottom-contact fishing (Eigaard et al., 2017; Bastari et al., 2018; Mačić et al., 2022), through direct removal of individuals, damage or dislodgement of the colonies, and smothering caused by sediment resuspension (Kaiser et al., 2002; Chimienti et al., 2018b).
The negative impacts of bottom-contact fisheries on marine ecosystems have been recognized by the United Nations General Assembly Resolution (61/105) (UNGA, 2006), which calls on Member States and Regional Fisheries Management Organizations (RFMOs) to identify vulnerable marine ecosystems (VMEs) and to adopt measures for their protection. In response, the Food and Agriculture Organization (FAO) developed guidelines to support the identification of VME indicator taxa (FAO, 2009). Among several coral taxa, the Northwest Atlantic Fisheries Organization (NAFO) has listed sea pens as VME indicator taxa. While no official VME list has yet been established in the Mediterranean, the General Fisheries Commission for the Mediterranean has identified beds of Funiculina quadrangularis (Pallas, 1766) as an indicator of sensitive habitat (GFCM, SAC, 2009). Local fisheries restricted areas (FRAs) have been designated by the GFCM to protect VMEs and essential fish habitats (EFHs), including a general ban on the use of bottom-contacting gears (towed dredges and bottom trawl nets) below 1000 m depth (Rec. GFCM/29/2005/1) (Rodríguez-Rodríguez et al., 2016). This ban is currently being revised to apply below 800 m depth in French (Arrêté du 21 Octobre 2024), Italian, and Spanish waters. While the positive influence of fishing closures on epibenthic communities has been observed locally (Farriols et al., 2022; Martinelli et al., 2023), their implementation remains limited in the Mediterranean Sea, likely due to the involvement of multiple countries in the exploitation of fishable areas and the limited scientific knowledge on the distribution and environmental preferences of benthic species (Aguilar et al., 2017). The tall sea pen F. quadrangularis can form dense fields on muddy sediments, called “soft mud facies with a fluid surface” (Pérès and Picard, 1964; Augier, 1982), at depths between 20 and 1000 m (Greathead et al., 2007; Bastari et al., 2018; Pierdomenico et al., 2018). This octocoral is distributed in the North Atlantic (Greathead et al., 2007; Downie et al., 2021), in the Mediterranean (Fabri et al., 2014; Bastari et al., 2018; Pierdomenico et al., 2018; Martinelli et al., 2023), and in the Pacific (Fujita and Ohta, 1988). Funiculina quadrangularis can reach up to 2 m in height and is characterized by a flexible calcareous axial rod with a four-lobed cross-section along its length (Greathead et al., 2007; Greeley, 2022). A recent analysis of sea pen skeletons estimated that, although F. quadrangularis exhibits relatively fast linear growth (around 3.8 cm per year), its radial growth rate is among the slowest reported for sea pens (around 0.08 mm per year) (Greeley, 2022). This suggested that F. quadrangularis can grow tall quickly while remaining very thin and therefore fragile to physical damage. Although some colonies were estimated to be between 7 and 18 years old, accurate age determination remains difficult because growth rates seem not constant over the lifespan of the species (Greeley, 2022). While its erect structure enhances the complexity of otherwise homogeneous soft bottoms (Buhl-Mortensen et al., 2010; De Clippele et al., 2015; Bastari et al., 2018), this sea pen species is unable to withdraw into the sediment, increasing its vulnerability to fishing gears (Hughes, 1998; Greathead et al., 2007). As F. quadrangularis co-occurs with some of the most important target species of bottom trawling, such as the European hake (Merluccius merluccius (Linnaeus, 1758)), Norway lobster (Nephrops norvegicus (Linnaeus, 1758)) and deep-water rose shrimp (Parapenaeus longirostris (Lucas, 1846)) (Greathead et al., 2007; Downie et al., 2021; Martinelli et al., 2023), it makes this species highly vulnerable to fishing. The negative impact of bottom trawling on F. quadrangularis has been reported in several areas of the Northeast Atlantic (Greathead et al., 2007) and of the Mediterranean (Fabri et al., 2014; Martinelli et al., 2023), with significant density declines in areas of intense bottom trawling (Tudela, 2004; Ardizzone, 2006). Indeed, it is estimated that the F. quadrangularis population in the Mediterranean has shrunk by 40% during the last 20 years, classifying the species as vulnerable in the IUCN Red List of Threatened Species (Bo et al., 2017). Nevertheless, it is important to note the persistence of this species in areas of moderate trawling activities, showing a certain resistance, possibly due to its flexibility and unbranched structure (Lauria et al., 2017; Pierdomenico et al., 2018).
Despite most published research having focused on the impact of fishing activities on this species, its habitat selection and potential response to climate change remain largely unknown. Along continental margins, benthic communities are typically associated with relatively stable abiotic conditions, suggesting limited capacity for adaptation to environmental changes (Danovaro et al., 2018). Ongoing climate change is expected to further reduce their suitable habitat in the future (Sweetman et al., 2017; Morato et al., 2020). Studies have already demonstrated changes in deep-sea environmental conditions, including warming of deep waters (Purkey and Johnson, 2010; Morato et al., 2020), deoxygenation (Keeling et al., 2010), acidification (Byrne et al., 2010), and increased stratification of the water column, which can alter the flux of organic matter to the seafloor (Smith et al., 2013). These effects are expected to be particularly intense in the Mediterranean, a semi-enclosed basin characterized by relatively stable environmental conditions with relatively high bottom temperature (13–14°C below 200 m) with high salinity (38–39 PSU) and oxygen levels (Cartes et al., 2004) and where climate change is progressing at a more accelerated rate than in other regions of the global ocean (Lejeusne et al., 2010; Danovaro, 2018; Pastor et al., 2020). Considering the ecological importance of the VMEs characterized by the presence of F. quadrangularis, it is necessary to predict its suitable habitat under current environmental conditions and project how this could be affected by climate change.
Species distribution modelling (SDM) (Elith and Leathwick, 2009), linked to the “ecological niche” concept, correlates species occurrences with environmental factors to predict the suitable habitat of species across space and time (Leroy et al., 2014; Schickele et al., 2020; Downie et al., 2021). SDMs are known to produce valuable insights for different marine conservation needs such as fisheries management, species protection, and marine spatial planning (Karp et al., 2025). Applying an ensemble approach that combines multiple models fitted using different algorithms has been recognized as particularly efficient to produce robust predictions and future projections (Robert et al., 2016; Georgian et al., 2019). This is especially valuable for deep-sea species, which remain under-sampled despite advances in systematic scientific surveys (Beazley et al., 2018; Murillo et al., 2018; Lauria et al., 2021; Carbonara et al., 2022). Projecting habitat shifts of vulnerable deep-sea species has been a key focus for identifying potential future climate refugia and informing on priority areas for conservation (Morato et al., 2020; Georges et al., 2024; Millot et al., 2024). While occurrences of F. quadrangularis have already been locally described in the Mediterranean (Fabri and Pedel, 2020; Fabri et al., 2014; Pierdomenico et al., 2018; Mačić et al., 2022; Martinelli et al., 2023), with several studies focusing on its distribution at regional scales (Lauria et al., 2017; Bastari et al., 2018), information on its habitat preferences and widespread distribution at the Mediterranean scale still remains unknown.
In this study, we modeled the potential suitable habitat of F. quadrangularis in the Mediterranean Sea using an SDM framework based on the approach developed by Millot et al. (2024). This framework incorporated a robust ensemble of modeling methodologies along with a transparent assessment of modeling uncertainties. Predictions were based on a comprehensive presence–absence dataset from the Mediterranean International Trawl Survey program (MEDITS) (MEDITS, 2017), ROV observations, and records from the literature. To evaluate the potential impact of climate change, the habitat of F. quadrangularis was projected following the “middle of the road” SSP2–4.5 climate scenario of the Intergovernmental Panel on Climate Change (IPCC) for the years 2050 and 2100. Projecting habitat shifts of the species in the near future (by 2050) can inform urgent decision-making on the prioritization of conservation areas for VMEs in the Mediterranean, with long-term projections useful to anticipate the more drastic impacts that are expected to affect the Mediterranean seafloor by the end of the century.
2 Material and methods
2.1 Study area
The study was performed focusing on soft bottom habitats of the Mediterranean basin, from the continental shelf down to the mesobathyal plains (Emig, 1997) (<1000 m). The study area (Fig. 1) covered all the GFCM GSAs (geographical sub-areas) defined in the Mediterranean, excluding those of the Marmara, Black, and Azov seas (Fig. A1 in the Appendix A).
 |
Fig. 1 Map of the study area showing the raw presence (A) and absence (B) records of Funiculina quadrangularis; records are color-coded according to the data source. Blue-shaded regions represent areas where depth is <1000 m. |
2.2 Data compilation
2.2.1 Biological data
Presence and absence records of F. quadrangularis were obtained from bottom trawl surveys, and additional presence records were collated using data coming from Remotely Operated Vehicle (ROV) surveys, open data platforms (OBIS, GBIF), and the scientific literature. The major part of the data was provided by the MEDITS program, which is mandatory for all Mediterranean EU member states. Annual surveys were generally carried out between late spring and mid-summer (the period could vary due to national constraints) and were designed to assess stocks of exploited demersal species. Trawl hauls were located in five bathymetric strata (10–50, 51–100, 101–200, 201–500, and 501–800 m), sampled at the same position over the years according to a stratified random sampling design. Trawl duration was approximately 30 min on shallower bottoms (<200 m) and about 60 min in deeper areas at a constant 3.5 kt speed using a 10 mm mesh size net in the cod end (MEDITS, 2017). All demersal species collected were sorted, weighed, and counted following the latest version of the MEDITS Handbook (MEDITS, 2017). Although recommended since 2012 and currently carried out in most GSAs, sampling of invertebrate benthic species remains optional in the MEDITS protocol. Consequently, data availability and accuracy are dependent on the taxonomic expertise of each team. In order to have a complete database for the analysis, additional data were integrated from local bottom trawl surveys, including GOLDYS (Vaz et al., 2023), EPIBENGOL (Vaz, 2018), NOURMED (Vaz, 2018) over the coast and shelf of the Gulf of Lion (GSA7), and SOLEMON (AdriaMed, 2011). Video data were also incorporated from the MEDSEACAN and CORSEACAN surveys (2009-2010) conducted in French waters (GSAs 7 and 8), from 180 to 700 m depth (Fabri and Pedel, 2020). Further presence points were obtained from the literature to address data gaps in the Aegean Sea (Salomidi et al., 2022; Smith et al., 2022). Details on the data used in this study are provided in Table B1 in the Appendix B.
To ensure that the absence of the species was true and not due to a non-sampling of benthic taxa, only stations from trawl/ROV surveys where F. quadrangularis was observed at least once were included. Absence data from the Aegean Sea (GSAs 22 and 23) were excluded due to non-exhaustive benthic sampling across the area, except for the Argosaronikos subregion. Additionally, potential false presences due to onboard net contamination were excluded when abundance data were available. In particular, hauls with very low yields of F. quadrangularis (g/km² <5th percentile) were removed if they followed a haul with very high yields (g/km² >95th percentile).
2.2.2 Generation of pseudo-absences in presence-only areas
In the Aegean Sea, although the identification of the species was reliable, the lack of exhaustive sorting of the catches did not ensure that absence data were such. In this case, MEDITS absences were not deemed sufficiently reliable due to incomplete benthic sampling, and pseudo-absences were generated using a convex hull approach, following the methodology of Hattab et al. (2017). The environmental space of each area was reduced to three dimensions by applying principal component analysis (PCA) and selecting the first three axes. A convex hull was used to model the “observed environmental space”, encompassing all presence and absence points, while the “realized environmental space” was defined around presence points only. Pseudo-absences were then randomly sampled within the “observed environmental space” but outside the “realized environmental space”. The number of pseudo-absences was set to match the prevalence (presence/absence ratio) typically observed in MEDITS data, the primary source of records, ensuring a realistic proportion of presences to absences in the Aegean Sea relative to the rest of the basin.
2.2.3 Environmental data
Key environmental drivers for benthic communities were compiled from various sources. Depth data were obtained from the EMODnet portal (https://emodnet.ec.europa.eu/en) at a resolution of 0.001°, and seabed slope and roughness (i.e., irregularity of the seabed calculated as the difference between maximum and minimum values of the surrounding cells) were derived with the terra::terrain function in R. The European broad-scale seabed habitat map (Vasquez et al., 2023) was used to generate a layer of sediment average grain size (Millot and Vaz, 2024).
Additionally, a comprehensive set of environmental predictors was collected from the latest Bio-Oracle update (http://www.bio-oracle.org) (Assis et al., 2024). These included bottom seawater temperature, salinity, current speed, pH, dissolved oxygen, primary productivity, and chlorophyll-a concentrations at a resolution of 0.05°. For each predictor, the monthly mean, monthly range, and long-term averages of maximum and minimum values per year were extracted for the period 2010–2020. Further details on the sources and characteristics of each environmental predictor are provided in Table B2 in the Appendix B.
Projections of environmental layers up to 2100 under several shared socioeconomic pathway (SSP) scenarios of the IPCC were extracted from the platform Bio-Oracle (http://www.bio-oracle.org) (Assis et al., 2024). These layers were obtained from monthly data of a large set of Earth system models provided by the CMIP Phase 6. Projections under the optimistic SSP1–2.6, intermediate SSP2–4.5, and pessimistic SSP5–8.5 climate scenarios were generated for two time periods, 2050 and 2100 (Tab. B3 in the Appendix B). Only the projections from the intermediate scenario were compared in the study, while the results from the other scenarios were provided in the Appendix (see the “Results” section).
Environmental data were rescaled at a resolution of 0.05° (approximately 4 to 5 km, i.e., close to the resolution of MEDITS data, which varies between 3.2 and 6.5 km depending on haul duration) and were extracted at each location of presence–absence records of F. quadrangularis. A reduced set of environmental predictors was selected according to ecological relevance and by avoiding predictors correlated higher than 0.7 of the Spearman coefficients (Dormann et al., 2013) (Fig. A2 in the Appendix A). More details on the correlation calculation are available in Millot et al. (2024). Outliers, defined as stations with environmental values beyond the 0.001 and 0.999 quantiles, were removed to prevent overfitting in regression models.
2.3 Species distribution modelling framework
2.3.1 Spatial autocorrelation treatment and variable selection
Spatial autocorrelation occurs when values from nearby locations are more similar to each other than those from distant locations (Cliff and Ord, 1970; Dormann et al., 2007). Reducing spatial autocorrelation in the residuals of the model is necessary to avoid overfitting of the model. Therefore, following the methodology proposed by Millot et al. (2024), we first fitted a generalized additive model (GAM) using the initial selection of environmental predictors. All combinations of these predictors were then evaluated, and the optimal set was selected based on the Bayesian information criterion (BIC), which was chosen for its tendency to favor more parsimonious models and its suitability for large sample sizes. Next, we assessed spatial autocorrelation in model residuals by fitting a variogram and determining an appropriate minimum distance. Using this threshold, we resampled 30 independent datasets to reduce spatial autocorrelation between stations. The final selection of environmental predictors, consistent across all datasets, was determined by fitting a GAM on one resampled dataset and applying the BIC criterion to identify the optimal set.
2.3.2 Model fitting and performance assessment
Functions of the Biomod2 v4.2-4 package (Thuiller et al., 2023) were used in the R v4.2.2 environment to facilitate the implementation of ensemble SDMs and obtain projections under present conditions and future climate scenarios.
Each resampled dataset was split into five spatially independent folds using the blockCV package in R, resulting in two main datasets: calibration and testing. This method ensures robust cross-validation by assessing the models’ ability to extrapolate in unknown environmental conditions (Fourcade et al., 2018). The block sizes were determined based on the spatial extent of the GFCM FAO – GSAs.
Different algorithms were used to develop SDMs on the calibration datasets, in particular, two regression algorithms, Generalized Linear Model (GLM; McCullagh, 1984) and GAM (Hastie and Tibshirani, 1986), and two machine learning algorithms, Random Forest (RF; Breiman, 2001) and Gradient Boosted Model (GBM; Elith et al., 2008). Predictor contributions were evaluated by sequentially shuffling the values of each predictor and comparing the model output when all predictors were included. Response curves for each predictor and run were generated to visualize the probability of the presence of the species along each predictor gradient.
The performance of each model (five runs × four algorithms × 30 datasets) was assessed using standard evaluation metrics: (i) area under the curve (AUC), (ii) specificity (rate of correctly predicted absences), (iii) sensitivity (rate of correctly predicted presences), and (iv) true skill statistic (TSS), which integrates sensitivity and specificity but is not affected by prevalence (Allouche et al., 2006). Only models with a good AUC (>0.7; Fawcett, 2006) and a moderate TSS (>0.3; Allouche et al., 2006) were selected. These thresholds were set to balance model performance with the representativity of the four algorithms in the individual models.
The best-performing models were considered to predict species presence probability across the entire study area under current conditions and to make projections into the future (2050 and 2100). These outputs were combined into an ensemble model, in which individual outputs were averaged and weighted according to their TSS values. Binary habitat suitability maps were derived by converting probabilities into presence (1) and absence (0) values using an optimal threshold determined by the function biomod2::bm_FindOptimStat, ensuring the best trade-off between sensitivity and specificity. Finally, present predictions and future projections were compared with identify climate refugia (defined as areas of suitable habitats in both present and future environmental conditions), habitat gain (unsuitable areas becoming suitable in the future), and habitat loss (suitable areas becoming unsuitable in future environmental conditions).
2.3.3 Assessment of modelling uncertainties
Model residuals were estimated as the difference between observed values (0 for absence and1 for presence) and predicted probabilities of presence (ranging continuously from 0 to 1). Agreement among individual models within the ensemble was assessed by calculating the standard deviation of their predicted presence probabilities. Moreover, to identify the main sources of variation around the ensemble predictions, we performed a three-way Analysis of Variance (ANOVA) without replication for each cell (Legendre and Legendre, 1998), following the approach of Diniz-Filho et al. (2009). Species probability of presence was used as the response variable, while dataset, spatial block cross-validation, and modeling algorithm were included as explanatory factors. We quantified the sum of squares attributed to each of these sources and estimated their relative contribution to overall variability by expressing the associated sum of squares as a proportion of the total sum of squares. Additionally, present-day predictions from the selected models were averaged per algorithm, and the resulting mean predictions were mapped to facilitate interpretation of regions with model disagreement.
Levels of uncertainty associated with environmental extrapolation (i.e., predictions and projections made in environmental conditions not represented in the model's calibrating data) were assessed by the Extrapolation Detection tool (Exdet) based on Euclidean and Mahalanobis distances (Mesgaran et al., 2014). Cells classified as “univariate extrapolation” represent environmental conditions that fall outside the range of individual predictors. Conversely, “combinatorial extrapolation” indicates conditions that lie within the range of individual predictors but form novel combinations not observed in the calibration data. Cells with environmental conditions already present in the calibrating dataset are classified as “analogous conditions”.
3 Results
3.1 Filtering of presence–absence records
From the raw presence–absence dataset of F. quadrangularis (10,881 records: 1837 presences, 9044 absences), 51 presences were removed due to potential onboard net contamination (from hauls with less than 20 g/km² following hauls with more than 27,000 g/km²). Additionally, 956 records, including 61 presences, were excluded due to unavailable environmental data. Finally, the estimated autocorrelation distance used for resampling the presence–absence dataset was set to 0.20° (i.e., about 20 km). The final resampled datasets comprised 1070 records, including 407 presences.
3.2 Environmental predictor selection
Of the 32 environmental predictors tested, only four were ecologically relevant and selected through the correlation analysis and BIC-based model selection: depth (m), mean bottom temperature (°C), long-term maximum salinity (PSU), and long-term minimum oxygen (mmol/m) (Figs. A3 and A4 in the Appendix A). Other predictors related to the geomorphology of the seabed and primary production (chl-a, productivity) were strongly correlated with depth (Fig. A2 in the Appendix A). The long-term minimum pH was also found to significantly contribute to the GAM predictions. However, this predictor was excluded due to the limited environmental gradient of pH observed in the Mediterranean (ranging from 8 to 8.1), which was insufficient to model and interpret pH preferences of the species.
3.3 Model performance assessment and environmental predictors’ contributions
Among the 600 individual models produced, 105 models presenting an AUC > 0.7 and a TSS > 0.3 were selected, including 51 GBMs, 35 RFs, 11 GLMs, and 8 GAMs (Fig. 2A). GBM and RF models exhibited higher sensitivity and specificity (above 0.7) compared with regression models (between 0.6 and 0.7) (Fig. 2B). While the average AUC across selected models was similar across the four algorithms (0.73–0.75), machine learning algorithms exhibited, on average, slightly higher TSS (0.38–0.39) values than regression models (0.34) (Tab. 1).
The contribution of environmental predictors varied slightly among models and algorithms (Fig. 2C). On average, long-term maximum salinity had the highest contribution in RFs and GBMs, ranging between 0.4 and 0.5. In these models, long-term minimum oxygen contributed at levels comparable to depth and mean temperature (averaging between 0.3 and 0.4), except for mean temperature, with a contribution in GBMs lower than 0.3. For GLMs and GAMs, long-term minimum oxygen ranked as the main contributor (between 0.3 and 0.4 on average), closely followed by depth and by long-term maximum salinity (between 0.2 and 0.3 on average). Mean temperature had a slightly lower influence in these regression models, averaging between 0.1 and 0.2 (Fig. 2C).
Averaged AUC and TSS per algorithm across the selected models.
 |
Fig. 2 Model performance evaluation with (A) Area Under the Curve (AUC) and True Skill Statistic (TSS) scores, with the selected models highlighted by bold circles; (B) specificity and sensitivity scores; and (C) contributions of environmental predictors. Models are color-coded by algorithm. 'Salinity_ltmax refers to the long-term maximum salinity, 'temp_mean' to the mean bottom temperature, and 'o2_ltmin' to the long-term minimum oxygen level. |
3.4 Response curves to environmental predictors
Our models suggested that the species occupies a wide depth range in the Mediterranean Sea, occurring generally from 100 to 750 m depth. While GAMs and GLMs predicted shallower waters as suitable for the species, RF and GBM indicated a clear decrease in probability of presence shallower than 100 m (Fig. 3A). These two machine learning algorithms identified an optimal thermal range between 14°C and 16°C, although no clear upper thermal limit was depicted, with relatively high presence probability persisting above 16°C. In contrast, GLMs and GAMs showed a clear decline in habitat suitability beyond 16°C and a gradual decline in presence probability below 14°C (Fig. 3B). Regarding the salinity, only RFs indicated a clear preference for long-term maximum salinity between 38.3 and 39 PSU. The probability of presence decreased abruptly after around 39 or 39.5 PSU depending on the model, the four models strongly differed in identifying the lower limit. RFs indicated a decline in presence probability below 38 PSU, GLMs and GAMs below 37.5 PSU, and GBMs showed a good tolerance down to 37 PSU (Fig. 3C). Finally, the four algorithms demonstrate a similar preference for long-term minimum oxygen concentration between 150 and 225 mmol/m (Fig. 3D).
 |
Fig. 3 Response curves of the probability of presence of F. quadrangularis as a function of the different environmental predictors selected, coloured by algorithm: (A) depth, (B) mean bottom temperature, (C) long-term maximum bottom salinity, and (D) long-term minimum bottom oxygen. The solid line represents the mean response curve across individual models of each algorithm, with shaded areas indicating the 95% confidence interval. |
3.5 Distribution predictions under present conditions and projections in the future
According to the optimization diagnostic, the optimal threshold for converting probability into presence or absence was set at 0.52. The probability of presence reached 0.8 in the present-day and future time periods, with the highest values observed in the Gulf of Lion, in French waters.
Under present-day environmental conditions, F. quadrangularis appeared widely distributed across the continental shelf and upper slopes of Western Mediterranean and the Central Basin. In contrast, suitable areas in the Adriatic Sea and the Eastern Mediterranean Sea were restricted in comparison with the Western and Central Mediterranean. The Levantine basin and the shallow continental shelf of the Gulf of Gabès within Tunisian waters appeared largely unsuitable (Figs. 4A–4B). By 2050, under the intermediate IPCC SSP2–4.5 scenario, the distribution of the species remained unchanged in the Western Mediterranean and in the Central Basin. New suitable zones may appear in the central Adriatic Sea and in the deeper regions of the Cretan Sea, while in the Northern Aegean, suitable areas were expected to become less extensive than they were at present (Figs. 4C–4D). However, by 2100, under the same intermediate IPCC SSP2–4.5 scenario, only the western basin was projected to maintain a wide, suitable habitat for the species. Deeper zones of the Eastern Mediterranean Sea became suitable habitat, while large areas of the Central Basin and the rest of the eastern and southern Mediterranean continental shelves and slopes became unsuitable (Figs. 4E–4F).
 |
Fig. 4 Habitat suitability (left) converted to binary presence–absence values (right) by an optimal threshold of 0.52, illustrating the potential areas that are suitable for F. quadrangularis in the Mediterranean basin for (A–B) the present-day conditions and under the IPCC scenario SSP2–4.5 (C–D) by 2050 and (E–F) by 2100. |
3.6 Identification of climate refugia, habitat gain, and loss
Under the intermediate IPCC SSP2–4.5 scenario, the distribution of F. quadrangularis was projected to remain largely unchanged by 2050 but showed a drastic reduction by 2100. In total, 84% of the areas currently suitable for the species were identified as climate refugia by 2050, whereas only 49% remained as such by the end of the 21st century. In both periods, the western basin retained the majority of the areas identified as climate refugia. The Central Basin, on the other hand, maintained suitable areas only until 2050, with a drastic reduction of the suitable habitat by 2100. Climate refugia were detected in the Northern Adriatic and Northern Aegean Seas by 2050 but were lost based on the projections made for the end of the century (Figs. 5A–5B). Significant habitat gains were also projected in several locations, with expansions of 32% and 47% by 2050 and 2100, respectively, compared with the suitable habitat of the species predicted under present conditions. These new suitable areas were mainly located in the Ligurian Sea, along eastern Corsica, and in northern Tunisian waters, as well as in deep waters of the Eastern Mediterranean Sea (Figs. 5A–5B).
 |
Fig. 5 Areas projected as climate refugia, habitat gain, habitat loss, or unsuitable for F. quadrangularis under the IPCC SSP2–4.5 scenario by (A) 2050 and (B) 2100. Climate refugia are defined as areas that remain suitable under both present and future conditions; habitat gain refers to currently unsuitable areas that become suitable in the future, and habitat loss to suitable areas becoming unsuitable. |
3.7 Model agreement and model residuals
Individual models of the four algorithms showed relatively high agreement across the Mediterranean basin, with a standard deviation ranging from 0.05 to 0.20. Regions associated with the highest variability among models included the Catalan Sea, the Ligurian Sea, the Northern Adriatic, and the Central and Southern Aegean Sea (Fig. 6A). Among the three sources of variations tested, the algorithm explained the most part of the total sum of squares between individual predictions, compared with dataset and spatial block cross-validation (Fig. A5 in the Appendix A). A disagreement between regression models and machine learning models was particularly highlighted in the Northern Adriatic Sea and Catalan Sea, where GLMs and GAMs identified a higher probability of presence under present-day conditions than RFs and GBMs, which predicted a more restricted habitat suitability for the species (Fig. A6).
Most residuals, both negative (i.e., high predicted presence probability despite observed absence) and positive (i.e., low predicted presence probability despite observed presence), showed moderate values ranging between −0.4 and 0.4, accounting for 71% of all residuals (Fig. 6B). Strongly negative residuals (< −0.4; i.e., false presences) represented 23% of the observations and were mainly identified across the Mediterranean basin, particularly in the western region along the Sicilian and western Italian coasts, as well as in the Central Adriatic (Fig. 6B). In contrast, only a small proportion of residuals (6%) were strongly positive (>0.4; i.e., false absences).
 |
Fig. 6 Prediction uncertainties are represented by (A) the standard deviation (sd) among individual model predictions combined in the ensemble model and (B) the ensemble model residuals, with negative residuals shown in red and positive residuals in blue. |
3.8 Environmental extrapolation assessment
Almost the entire Mediterranean basin presented analogous conditions (i.e., similar to conditions known in calibrating datasets), both for present-day (Fig. 7A) and future conditions (Figs. 7B and 7C). Univariate environmental extrapolation concerned a relatively small proportion of pixels, accounting for 11% of the study area under present-day conditions, 13% under the intermediate scenario by 2050, and 16% under the intermediate scenario by 2100. The same areas were identified across the three periods (Figs. 7A–7C), but at a higher level in the future, particularly by 2100 (Fig. 7C). These areas were restricted to the shallow continental shelf of the Gulf of Gabès, the Libyan coast, and the Southern Levantine Sea. They correspond to areas where no MEDITS data were available. No areas of combinatorial extrapolation (i.e., combinations of environmental predictors never observed) were identified under the three periods.
Probabilities of presence and climate refugia projections under the more optimistic low-emissions (SSP1–2.6) and pessimistic high-emissions (SSP5–8.5) scenarios are provided in Appendix A (Figs. A7, A8, and A9), and their uncertainty assessment is in Appendix A (Fig. A10). By 2050, projections under the two extreme climate scenarios were very similar to those of the intermediate SSP2–4.5. However, while the optimistic SSP1–2.6 projected fewer losses in the Central Basin compared with SSP2–4.5, the pessimistic SSP5–8.5 projected even wider losses in both the Central Basin and the Northern Adriatic.
 |
Fig. 7 Environmental extrapolations associated with (A) predictions under present-day conditions, (B) projections under the IPCC SSP2–4.5 scenario by 2050, and (C) by 2100. For each period, univariate extrapolations (i.e., predictions/projections outside the range of the environmental space of calibrating data for at least one predictor) are mapped in orange, and analogous conditions (i.e., predictions/projections in environmental conditions already present in calibrating data) are shown in green. Areas with no sampled data are shown with faded shading, and predictions/projections in such areas must be interpreted with caution, being associated with higher uncertainties. |
4 Discussion
This study used a species distribution modeling approach to assess habitat suitability for the octocoral Funiculina quadrangularis on soft-bottom habitats down to 1000 m depth in the Mediterranean basin. Climate-driven habitat shifts have been projected for the years 2050 and 2100, based on IPCC projections of future seawater conditions. Under present-day environmental conditions, the ensemble model predicted a broad potential distribution, indicating that the species can occupy a wide bathymetric range from shelf habitats to the lower continental slope (100–750 m). This finding was consistent with previous observations across the Mediterranean (Greathead et al., 2015; Bastari et al., 2018; Martinelli et al., 2023).
4.1 Potential suitable habitat of the species
Model responses indicated a relatively broad environmental range for F. quadrangularis in terms of temperature (12–16°C), salinity (37–39 PSU), and dissolved oxygen concentrations (150–225 mmol/m³). These suitable ranges must be interpreted in the context of the Mediterranean basin, where environmental conditions are quite constant below 200 m (Cartes et al., 2004). This pattern contrasts with other VME indicator species, such as the crinoid Leptometra phalangium (Müller, 1841), more restricted to shelf break areas associated with moderate bottom currents (Colloca et al., 2004; Grinyó et al., 2018; Millot et al., 2024) or the bamboo coral I. elongata rarely observed above 600 m depth (Georges et al., 2024). These environmental optima corresponded to the observed environmental niche, which is constrained by the available sampling and may therefore underestimate the full physiological tolerance of the species. Indeed, F. quadrangularis has been reported in much shallower waters (20–50 m) in the Northern Adriatic Sea (Salvalaggio et al., 2016). In addition, the upper thermal limit of the species remained uncertain, as model responses diverged, with machine-learning algorithms predicting relatively high habitat suitability at temperatures exceeding 16°C. The lack of observations from shallow environments and from the southern Mediterranean and Levantine Sea, where seawater temperature is higher than in the rest of the Mediterranean basin, prevented a reliable assessment of the full environmental tolerance of the species.
The wide distribution of F. quadrangularis, still found on highly trawlable bottoms, may also suggest that this species is more resistant to the impacts of bottom trawling than other VME indicator taxa inhabiting soft bottoms, such as the bamboo coral I. elongata, which has been largely eradicated on the continental shelf (Lauria et al., 2017; Georges et al., 2024). Unlike scleractinians, which have a hard calcareous skeleton, the flexibility of this octocoral may partially enable it to evade direct removal, reducing the likelihood of being captured as bycatch (Pierdemonico et al., 2018). Furthermore, experimental studies have shown that the species can reinsert itself into the sediment and position itself right after being uprooted, after only a few days (Eno, 2001). The relatively fast growth rate of the species could help to regenerate rapidly after disturbance (Greeley, 2022). Moreover, high genetic diversity and gene flow among populations of F. quadrangularis, locally demonstrated in Scottish waters, revealed the potential resilience of the species to human activities through its capacity to recolonize areas already damaged by fisheries (Wright et al., 2015). However, significant declines in the species’ abundance have been reported in areas subjected to intense trawling pressure (Pierdomenico et al., 2018; Martinelli et al., 2023), clearly demonstrating its vulnerability to anthropogenic disturbance, which is aggravated by its erect structure (it can exceed 1 m in height) and its inability to withdraw into the sediment unlike other pennatulaceans found in the same habitats (Bastari et al., 2018; Chimienti et al., 2018a; Ambroso et al., 2013, 2021).
4.2 Short-term versus long-term climate projections
While environmental changes could lead to a moderate habitat loss of 16% for the species by the year 2050 under the intermediate SSP2–4.5 scenario, projections for 2100 indicated that around 50% of its currently suitable habitat could be lost. Climate-driven habitat losses have already been predicted for other vulnerable benthic species, such as the crinoid L. phalangium (Millot et al., 2024), the bamboo coral I. elongata (Georges et al., 2024), and several cold-water corals (Morato et al., 2020). The relatively stable physical conditions under which F. quadrangularis currently lives were predicted to be altered by ongoing warming of Mediterranean waters, potentially compromising its capacity to occupy large areas of the continental shelf and slope toward the end of the century (Sweetman et al., 2017; Danovaro et al., 2018). Consistently, the ensemble model indicated that a 1°C increase by 2050 should not strongly reduce suitable habitat, whereas a 2°C increase by 2100 could result in important habitat loss (i.e., the Sicily Channel and the Northern Aegean Sea). An increase in the salinization of Mediterranean bottom waters toward the end of the century (Skliris et al., 2018; Margirier et al., 2020) may further explain habitat losses despite suitable temperatures (i.e., Eastern Ionian Sea).
However, as for predictions under present-day conditions, future projections remained strongly constrained by sampling limitations, particularly by the lack of observation in the warmest waters of the Mediterranean. It is therefore not excluded that F. quadrangularis, tolerates a higher upper thermal limit than those suggested by the model, leading to a potential overestimation of habitat loss. Moreover, disentangling the drivers of these habitat shifts remains challenging as SDMs investigate correlations among environmental predictors and species distributions rather than identifying causal relationships (Austin, 2002) and might fail to determine the cumulative effects of multiple stressors.
In contrast, significant habitat gains, representing 32% and 47% of the suitable habitat of the species under present conditions, were also projected by 2050 and 2100, respectively (e.g., Ligurian Sea, Southern Tyrrhenian Sea, and deep waters of the Eastern Mediterranean). The factor limiting its current distribution in these areas remained unclear, as salinity and oxygen levels were suitable under both current and future conditions. These gained regions were, however, associated with seawater temperatures below 14°C, which were predicted as unsuitable under present-day conditions by the numerous RF models selected in the ensemble but were projected to become suitable under the warming of waters in the future. Nonetheless, this lower thermal limit may be questioned, as regression models (GLMs and GAMs) maintained a high probability of presence below 14°C, and F. quadrangularis has been observed in the cold waters of the North Atlantic and North Sea (Greathead et al., 2007; Downie et al., 2021). In any case, uncertainties remain regarding the ability of the species to colonize new suitable areas in the future. Low genetic differentiation among isolated patches of F. quadrangularis in northwest Scotland suggested a high rate of gene flow between populations during the pelagic stages of its life cycle (Wright et al., 2015), although this still needs to be verified in the Mediterranean Sea. Moreover, a fast growth rate has recently been quantified for this species (around 3.8 cm per year) (Greeley, 2022) as well as a relative high fecundity (Edwards and Moore, 2009). Although only a small proportion of oocytes are spawned annually, this is compensated for by the large size of its colonies (Edwards and Moore, 2009). These characteristics may favor the colonization of new areas if they become suitable in response to climate change. Nevertheless, successful colonization at a pace compatible with the rate of climate change would not be possible if populations are destroyed by continuous bottom trawling. Further experimental studies on growth and reproductive capacities of the species, combined with the development of biophysical models to better understand larval dispersal and connectivity (Matos et al., 2024), would provide valuable insights into how feasible the expansion of the species would be.
4.3 Regional areas of higher uncertainty
Both present-day predictions and future projections revealed areas of elevated uncertainty in the southern and eastern Mediterranean (e.g., Tunisian and Libyan waters and the Levantine Sea). These regions were not sampled by the MEDITS survey and were characterized by environmental conditions that were novel relative to those used to calibrate the models, particularly warmer seawater temperatures. Although an earlier survey conducted in Tunisian waters in 1949 using dredge and trawl sampling did not record the species (Roger et al., 2024), its true absence in these areas must be confirmed. Model outputs in the southern and eastern basins should therefore be interpreted with caution. Nevertheless, producing predictions in these uncertain regions is valuable, as it highlights critical data gaps and the need for improved sampling coverage to enhance basin-scale predictions.
In contrast, while environmental extrapolation was generally low in the northern basin, several regional hotspots exhibited higher model disagreement, notably in the Catalan Sea, Ligurian Sea, Northern Adriatic Sea, and Central Aegean Sea. Prediction variability between individual models was mainly attributed to algorithmic differences (Fig. A5 in the Appendix A). These regions, mostly associated with absence records, were generally predicted as unsuitable by machine learning algorithms but suitable by regression models (Fig. A6 in the Appendix A). This disagreement appears to be linked to a difference in depth response: machine learning models predicted a sharp decline in probability of presence above 50 m depth, whereas regression models maintained high probabilities in shallow waters. These regions of model disagreement are shallower than 50 m and were therefore predicted as unsuitable by the ensemble model, reflecting the predominance of machine learning models, which were selected more frequently due to their slightly higher TSS. Nevertheless, model disagreement remained moderate across the basin, with a standard deviation below 0.2.
4.4 Niche truncation issues
Models fitted using a truncated representation of a species’ full ecological niche can lead to inaccurate extrapolations, not only under present-day environmental conditions but especially when transferring such models to future climatic scenarios (Owens et al., 2013; Peterson et al., 2018). To minimize the risk of niche truncation, occurrence data must, within the limits of available data, adequately cover the study area. Sparse occurrence datasets available for deep-sea species in the Mediterranean from public repositories (e.g., OBIS, GBIF) are insufficient to support robust species distribution modeling. However, compiling records from unconnected populations is questionable, as they might not conserve the same environmental limits (Pellissier et al., 2013). This may artificially increase the suitable habitat in areas where at least some of the species’ populations would not be able to adapt at the rate at which climate change may occur.
Including F. quadrangularis records from the Northeast Atlantic into our models could have captured a broader part of the species' ecological niche. However, it is unclear whether Mediterranean Sea and Atlantic Ocean individuals belong to a single, well-connected population or whether they are genetically isolated and locally adapted to their environment. The high gene flow observed between F. quadrangularis colonies at nearby locations along the northwest coast of Scotland (Wright et al., 2015) does not imply that distant populations across the Atlantic and Mediterranean are equally well connected. Recent studies have investigated genetic diversity and inter-population connectivity of cold-water corals across Mediterranean regions. While weak connectivity was reported among populations of Desmophyllum pertusum (Linnaeus, 1758) between Mediterranean ecoregions (Matos et al., 2024), persistent genetic pathways were observed for I. elongata (Lauria et al., 2025). These contrasting patterns highlight that connectivity is species-specific among cold-water corals, and additional genetic studies on F. quadrangularis at large scale are required.
4.5 Influence of the selection of environmental predictors
It is widely accepted that the choice of environmental predictors has a significant impact on SDM outcomes (Araújo and Guisan, 2006). The selection process remains challenging despite the progress in the development of statistical methods, such as correlation analysis, BIC criteria, and bootstrapping. In particular, choosing between two highly correlated predictors can significantly impact future projections, as they may not change similarly under climate change (Braunisch et al., 2013). For this reason, environmental predictor selection cannot be a fully automated process and requires a case-by-case analysis of their ecological relevance. This remains particularly challenging when limited data on the species’ physiology is available, as it is the case for F. quadrangularis. For instance, it remains unclear if the suitable habitat of sea pens, which have a poorly calcified axial rod, will be impacted by acidification as it has been predicted for scleractinian corals (Gori et al., 2016; Danovaro et al., 2018). Including pH as a predictor led to very pessimistic future projections due to the narrow range of currently observed pH values (between 8.0 and 8.1) in the Mediterranean Sea, compared with the projected 0.4 increase by 2050 under IPCC scenarios. Hence, pH was ultimately not included in our model to avoid an overestimation of climate change impacts, especially taking into consideration that F. quadrangularis is found in the Atlantic Ocean under much lower pH conditions than in the Mediterranean Sea (Greathead et al., 2007; Downie et al., 2021). In this regard, experimental studies under controlled conditions would provide better clues on the effects that increases in pH may have on the survivorship of this species and would allow such pessimistic projections to be confirmed or ruled out.
Furthermore, an indirect limiting factor for the survival of the species could be related to food availability. F. quadrangularis, a passive suspension feeder, depends on the presence of phyto-zooplankton and particulate organic matter in its surrounding waters, which could be significantly impacted by climate change (e.g., acidification, temperature, and lower river inputs) (Macias et al., 2025). This aspect could be particularly important in an oligotrophic context, such as in some areas of the Mediterranean basin (Salgado-Hernanz et al., 2022).
4.6 Data limitations to predict benthic species distribution
MEDITS surveys, conducted by a consortium of eight countries, provide, to our knowledge, the most extensive coverage of benthic records across Mediterranean trawlable soft bottoms (Terribile et al., 2016). However, MEDITS sampling presents several limitations that constrained the representation of the complete environmental niche of a benthic species as the sea pen F. quadrangularis. First of all, the MEDITS bottom trawl is designed to target demersal species and therefore presents limitations on the catchability of smaller-sized flexible benthic species (Chimienti et al., 2018). However, although abundance may be underestimated, presence data might be less biased due to the high densities at which the species is generally found, ensuring the catch of at least one individual. MEDITS surveys are conducted in late spring and summer, primarily to align with fisheries objectives, such as monitoring the recruitment of key commercial species like hake. This season does not coincide with the spawning period of F. quadrangularis, which occurs in midwinter (Edwards and Moore, 2009). Nonetheless, a seasonal survey in the Gulf of Lion showed that the species’ distribution remains stable throughout the year (Vaz et al., 2023). Additionally, the wide size range of F. quadrangularis individuals captured during MEDITS indicates that sampling is not restricted to a specific life stage, suggesting a minimal seasonal bias (Certain et al., 2025).
Despite the use of a standardized MEDITS sampling protocol across countries, the exhaustive sampling and taxonomic identification of benthic fauna, although recommended since 2012, is not mandatory (MEDITS, 2017). Consequently, data quality depends on the level of taxonomic expertise within each scientific team. Particular caution is required in the Aegean Sea, where several teams are carrying out the survey, leading to heterogeneous levels of taxonomic identification and, particularly, a gap of species-level data in the Northern Aegean Sea. Although pseudo-absences were generated in this area to limit bias, presence data may still be underestimated. Moreover, because MEDITS surveys are conducted by EU member states, important spatial gaps of data remain in the southern and eastern Mediterranean (e.g., Tunisia, Libya, and the Levantine basin), where environmental extrapolations are associated with high uncertainties. To fill some data gaps, the MEDITS dataset has been complemented with additional sources of data, including ROV observations, local trawl surveys, and presence records derived from the literature. While the compilation of multiple datasets can raise standardization issues for abundance data due to differences in sampling protocols, presence data are less affected by such limitations. In all cases, the entire dataset was standardized using a resampling grid to avoid spatial autocorrelation and ensure a consistent spatial resolution across the study area. Moreover, the partition of variance associated with different sources of uncertainty confirmed that dataset subsampling and spatial block cross-validations were not the main sources of prediction uncertainties when compared with algorithm disagreement, thus confirming the reliability of the information contained in the datasets at different spatial scales.
Ten-year climatologies of environmental parameters were used to generate species distribution predictions, which may smooth inter- and intra-annual variability. However, given the relatively stable temperature, salinity, and oxygen conditions characterizing Mediterranean continental margins (Cartes et al., 2004), seasonal and short-term variability are expected to be limited within the depth range occupied by F. quadrangularis. We therefore consider that the use of multi-year climatologies was sufficient to represent the potential suitable habitat of this benthic species but acknowledge that the present work does not elucidate processes at play at lower temporal resolutions.
Furthermore, it should be highlighted that the recent data used in this study (mostly 2012–2023) reflect Mediterranean soft-bottom communities under decades of intense bottom trawling (Russo et al., 2020; Marsaglia et al., 2024), and the response curves produced by the models may have been influenced by changes in the potential distribution of the species due to human activities.
4.7 Interest in conservation planning
Projected regions where habitat is expected to remain suitable for the species by the end of the century, despite climate change, are crucial to prioritize conservation actions (Keppel et al., 2015; Carter et al., 2020; Anderson et al., 2022; Chollett et al., 2022). Large areas in the western Mediterranean basin appeared to offer stable habitat conditions under both near- and long-term climate projections and were associated with relatively low uncertainty, reinforcing the importance of implementing additional protected and fishery-restricted areas. SDM outputs can provide valuable inputs for systematic conservation planning, which aims to identify priority areas of conservation while balancing ecological goals and socio-economic constraints (Margules and Presley, 2000). The prioritization of climate refugia identified in this study, as well as from other VME distribution studies in the Mediterranean (Millot et al., 2024; Georges et al., 2024), should represent a key conservation objective. Other crucial principles should be considered to design an efficient and persistent conservation network for VMEs in the Mediterranean, including the connectivity among protected areas and the socio-economic impact of spatial closures (Manea et al., 2020; Combes et al., 2021).
5 Conclusion
This study revealed a wide potential distribution of the VME indicator taxa F. quadrangularis on trawlable bottoms across the Mediterranean basin from the shelf toward the continental slope (100–750 m). The species appeared relatively ubiquitous, with a distribution that did not seem to be strongly structured by environmental factors. While its habitat was projected to remain suitable in the near future, long-term climate projections indicated drastic habitat shifts by 2100, with a potential loss of half of its current habitat. New suitable habitats were also projected in the Western Mediterranean and the deep waters of the Eastern basin, but the factors limiting the current distribution of the species in these areas and the species’ ability to colonize them still need to be clarified. These projections highlight the importance of identifying priority conservation areas for F. quadrangularis, particularly in the Western Mediterranean, which is expected to maintain extensive climate refugia. These insights could be integrated into systematic conservation planning exercises to support effective conservation policies. It may also pave the way toward the achievement of the 30 × 30 target of the EU Biodiversity Strategy, which aims to protect 30% of European seas by 2030.
Acknowledgments
Special thanks go to the scientific teams from the eight participating countries (Croatia, Cyprus, France, Greece, Italy, Malta, Slovenia, and Spain) for their essential roles in coordinating and conducting the MEDITS surveys
Funding
We acknowledge DG MARE (Commission’s Directorate-General for Maritime Affairs and Fisheries) for granting open access to the MEDITS database. This study was partially supported by a PhD studentship funded by Ifremer and by the European Union through the Horizon Europe project REDRESS (Restoration of Deep-Sea Habitats to Rebuild European Seas, https://redress-project.eu/), Grant No. 101135492. CDC acknowledges the grant CEX2024-001494-S funded by AEI 10.13039/501100011033.
Data availability statement
The research data associated with this article are included in the article.
References
- AdriaMed. 2011. Report of the Technical meeting on SoleMon survey activities, May 2011. GCP/RER/010/ITA/SR-01. (No. 01), AdriaMed Scientific Reports. FAO, Rome. [Google Scholar]
- Aguilar R, Perry AL, López J, Conservation and management of vulnerable marine benthic ecosystems, in: Rossi, S., Bramanti, L., Gori, A., Orejas, C. (Eds.), Marine Animal Forests: The Ecology of Benthic Biodiversity Hotspots. Springer International Publishing, Cham, 2017, pp. 1–43. https://doi.org/10.1007/978-3-319-17001-5_34-1 [Google Scholar]
- Allouche O, Tsoar A, Kadmon R. 2006. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J Appl Ecol 43: 1223–1232. [Google Scholar]
- Ambroso S, Dominguez-Carrió C, Grinyó J, López-González PJ, Gili JM, Purroy A, Requena S, Madurell T. 2013. In situ observations on withdrawal behaviour of the sea pen Virgularia mirabilis. Mar Biodivers 43: 257–258. [Google Scholar]
- Anderson OF, Stephenson F, Behrens E, Rowden AA. 2022. Predicting the effects of climate change on deep-water coral distribution around New Zealand–will there be suitable refuges for protection at the end of the 21st century? Glob Change Biol 28: 6556–6576. [Google Scholar]
- Araújo MB, Guisan A. 2006. Five (or so) challenges for species distribution modelling. J Biogeogr 33: 1677–1688. [Google Scholar]
- Arrêté du 21 octobre 2024. https://www.legifrance.gouv.fr/jorf/id/JORFTEXT000050397265 [Google Scholar]
- Ardizzone GD. 2006. Sensitive and Essential Fish Habitats in the Mediterranean Sea. Working document to the STECF/SGMED-06-01 sub-group meeting on sensitive and essential fish habitats in the Mediterranean. https://stecf.jrc.ec.europa.eu/documents/d/stecf/plen_06-01 [Google Scholar]
- Assis J, Fernández Bejarano SJ, Salazar VW, Schepers L, Gouvêa L, Fragkopoulou E, Leclercq F, Vanhoorne B, Tyberghein L, Serrão EA, Verbruggen H, De Clerck O. 2024. Bio-ORACLE v3.0: pushing marine data layers to the CMIP6 Earth system models of climate change research. Glob Ecol Biogeogr 33: e13813. [Google Scholar]
- Augier H. Inventory and classification of marine benthic biocenoses of the Mediterranean, in: Council of Europe (Ed.), Nature and Environment Series, Vol. 25, Council of Europe, Strasbourg: Croton, N.Y., 1982 [Google Scholar]
- Austin MP. 2002. Spatial prediction of species distribution: an interface between ecological theory and statistical modelling. Ecol Model 157: 101–118. [Google Scholar]
- Baillon S, Hamel JF, Mercier A. 2014. Diversity, distribution and nature of faunal associations with deep-sea pennatulacean corals in the northwest Atlantic. PLoS One 9: e111519. [Google Scholar]
- Bastari A, Pica D, Ferretti F, Micheli F, Cerrano C. 2018. Sea pens in the Mediterranean Sea: habitat suitability and opportunities for ecosystem recovery. ICES J Mar Sci 75: 1722–1732. [Google Scholar]
- Bateman DC, Bishop MJ. 2017. The environmental context and traits of habitat-forming bivalves influence the magnitude of their ecosystem engineering. Mar Ecol Prog Ser 563: 95–110. https://doi.org/10.3354/meps11959 [Google Scholar]
- Beazley L, Wang Z, Kenchington E, Yashayaev I, Rapp HT, Xavier JR, Murillo FJ, Fenton D, Fuller S. 2018. Predicted distribution of the glass sponge Vazella pourtalesi on the Scotian Shelf and its persistence in the face of climatic variability. PLoS One 13: e0205505. https://doi.org/10.1371/journal.pone.0205505 [Google Scholar]
- Beazley LI, Kenchington EL, Murillo FJ, Sacau MM. 2013. Deep-sea sponge grounds enhance diversity and abundance of epibenthic megafauna in the northwest Atlantic. ICES J Mar Sci 70: 1471–1490. https://doi.org/10.1093/icesjms/fst124 [Google Scholar]
- Bo M, Otero M. del M, Numa C. 2017. Overview of the conservation status of Mediterranean anthozoa. IUCN. https://doi.org/10.2305/IUCN.CH.2017.RA.2.en [Google Scholar]
- Bolam SG, Fernandes TF. 2003. Dense aggregations of Pygospio elegans: effect on macrofaunal community structure and sediments. J Sea Res 49: 171–185. https://doi.org/10.1016/S1385-1101(03)00007-8 [Google Scholar]
- Bourque JR, Demopoulos AWJ. 2018. The influence of different deep-sea coral habitats on sediment macrofaunal community structure and function. PeerJ 6: e5276. https://doi.org/10.7717/peerj.5276 [Google Scholar]
- Braunisch V, Coppes J, Arlettaz R, Suchant R, Schmid H, Bollmann K. 2013. Selecting from correlated climate variables: a major source of uncertainty for predicting species distributions under climate change. Ecography 36: 971–983. https://doi.org/10.1111/j.1600-0587.2013.00138.x [Google Scholar]
- Breiman L. 2001. Random Forests. Mach Learn 45: 5–32. https://doi.org/10.1023/A:1010933404324 [Google Scholar]
- Buhl-Mortensen L, Vanreusel A, Gooday AJ, Levin LA, Priede IG, Buhl-Mortensen P, Gheerardyn H, King NJ, Raes M. 2010. Biological structures as a source of habitat heterogeneity and biodiversity on the deep ocean margins. Mar Ecol 31: 21–50. https://doi.org/10.1111/j.1439-0485.2010.00359.x [Google Scholar]
- Byrne RH, Mecking S, Feely RA, Liu X. 2010. Direct observations of basin-wide acidification of the North Pacific Ocean. Geophys Res Lett 37: L02601. https://doi.org/10.1029/2009GL040999 [Google Scholar]
- Carbonara P, Zupa W, Follesa MC, Cau A, Capezzuto F, Chimienti G, DOnghia G, Lembo G, Pesci P, Porcu C, Bitetto I, Spedicato MT, Maiorano P. 2020. Exploring a deep-sea vulnerable marine ecosystem: Isidella elongata species assemblages in the western and central Mediterranean. Deep Sea Res Part I Oceanogr Res Pap 166: 103406. https://doi.org/10.1016/j.dsr.2020.103406 [Google Scholar]
- Carbonara P, Zupa W, Follesa MC, Cau A, Donnaloia M, Alfonso S, Casciaro L, Spedicato MT, Maiorano P. 2022. Spatio-temporal distribution of Isidella elongata in the southern Adriatic Sea. Hydrobiologia 849: 4837–4855. https://doi.org/10.1007/s10750-022-05022-4 [Google Scholar]
- Carter AL, Wilson AMW, Bello M, Hoyos-Padilla EM, Inall ME, Ketchum JT, Schurer A, Tudhope AW. 2020. Assessing opportunities to support coral reef climate change refugia in MPAs. Mar Policy 112: 103769. https://doi.org/10.1016/j.marpol.2019.103769 [Google Scholar]
- Cartes JE, Maynou F, Sardà F, Company JB, Lloris D, Tudela S. 2004. Mediterranean deep-sea ecosystems: an overview of their diversity, structure, functioning and anthropogenic impacts, in: The Mediterranean Deep-Sea Ecosystems: An Overview of Their Diversity, Structure, Functioning and Anthropogenic Impacts, with a Proposal for Conservation, IUCN, Málaga and WWF, Rome, 2004, pp. 9–38. [Google Scholar]
- Certain G, Jadaud A, Vaz S, Metral L, Cheret I, Billet N, Schull, Tessier E, Cornou AS, Souplet A, 2025. MEDITS catch data: bentho-demersal assemblages of the Gulf of Lion and eastern Corsica (North-western Mediterranean) from 2016 to present. SEANOE. https://doi.org/10.17882/99052 [Google Scholar]
- Chimienti G, Angeletti L, Mastrototaro F. 2018a. Withdrawal behaviour of the red sea pen Pennatula rubra. Eur Zool J 85: 64–70. https://doi.org/10.1080/24750263.2018.1438530 [Google Scholar]
- Chimienti G, Angeletti L, Rizzo L, Tursi A, Mastrototaro F. 2018b. ROV vs trawling approaches in the study of benthic communities. J Mar Biol Assoc UK 98: 1859–1869. https://doi.org/10.1017/S0025315418000851 [Google Scholar]
- Chollett I, Escovar-Fadul X, Schill SR, Croquer A, Dixon AM, Beger M, Shaver E, Pietsch McNulty V, Wolff NH. 2022. Planning for resilience: incorporating uncertainty when prioritizing climate refugia. Glob Change Biol 28: 4054–4068. https://doi.org/10.1111/gcb.16167 [Google Scholar]
- Cliff AD, Ord K. 1970. Spatial autocorrelation: a review of existing and new measures with applications. Econ Geogr 46: 269–292. https://doi.org/10.2307/143144 [Google Scholar]
- Colloca F, Carpentieri P, Balestri E, Ardizzone GD. 2004. A critical habitat for Mediterranean fish resources. Mar Biol 145: 1129–1142. https://doi.org/10.1007/s00227-004-1405-8 [Google Scholar]
- Combes M, Vaz S, Grehan A, Morato T, Arnaud-Haond S, Dominguez-Carrió C, Fox A, González-Irusta JM, Johnson D, Callery O, Davies A, Fauconnet L, Kenchington E, Orejas C, Roberts JM, Taranto G, Menot L. 2021. Systematic conservation planning at an ocean basin scale. Front Mar Sci 8. https://doi.org/10.3389/fmars.2021.611358 [Google Scholar]
- Danovaro R. 2018. Climate change impacts on the biota of the deep Mediterranean Sea. Rend Lincei Sci Fis Nat 29: 525–541. https://doi.org/10.1007/s12210-018-0725-4 [Google Scholar]
- Davies AJ, Guinotte JM. 2011. Global habitat suitability for framework-forming cold-water corals. PLoS One 6: e18483. https://doi.org/10.1371/journal.pone.0018483 [Google Scholar]
- De Clippele LH, Buhl-Mortensen P, Buhl-Mortensen L. 2015. Fauna associated with cold-water gorgonians and sea pens. Cont Shelf Res 105: 67–78. https://doi.org/10.1016/j.csr.2015. 06.007 [Google Scholar]
- Diniz-Filho JAF, Bini LM, Rangel TF, Loyola RD, Hof C, Nogués-Bravo D, Araújo MB. 2009. Partitioning and mapping uncertainties in ensembles of forecasts. Ecography 32: 897–906. https://doi.org/10.1111/j.1600-0587.2009.06196.x [Google Scholar]
- Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McClean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S. 2013. Collinearity: a review of methods to deal with it. Ecography 36: 27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x [Google Scholar]
- Dormann CF, McPherson JM, Araújo MB, Bivand R, Bolliger J, Carl G, Davies RG, Hirzel A, Jetz W, Kissling WD, Kühn I, Ohlemüller R, Peres-Neto PR, Reineking B, Schröder B, Schurr FM, Wilson R. 2007. Methods to account for spatial autocorrelation in species distribution data. Ecography 30: 609–628. https://doi.org/10.1111/j.2007.0906-7590.05171.x [Google Scholar]
- Downie AL, Vieira RP, Hogg OT, Darby C. 2021. Distribution of vulnerable marine ecosystems at the South Sandwich Islands: results from the Blue Belt Discovery Expedition 99 deep-water camera surveys. Front Mar Sci 8: 662285. https://doi.org/10.3389/fmars.2021.662285 [Google Scholar]
- Duffy JE. 2006. Biodiversity and the functioning of seagrass ecosystems. Mar Ecol Prog Ser 311: 233–250. https://doi.org/10.3354/meps311233 [Google Scholar]
- Edwards DCB, Moore CG. 2009. Reproduction in the sea pen Funiculina quadrangularis (Anthozoa: Pennatulacea) from the west coast of Scotland. Estuar Coast Shelf Sci 82: 161–168. https://doi.org/10.1016/j.ecss.2009.01.006 [Google Scholar]
- Eigaard OR, Bastardie F, Hintzen NT, Buhl-Mortensen L, Buhl-Mortensen P, Catarino R, Dinesen GE, Egekvist J, Fock HO, Geitner K, Gerritsen HD, González MM, Jonsson P, Kavadas S, Laffargue P, Lundy M, Gonzalez-Mirelis G, Nielsen JR, Papadopoulou N, Posen PE, Pulcinella J, Russo T, Sala A, Silva C, Smith CJ, Vanelslander B, Rijnsdorp AD. 2017. The footprint of bottom trawling in European waters: distribution, intensity, and seabed integrity. ICES J Mar Sci 74: 847–865. https://doi.org/10.1093/icesjms/fsw194 [Google Scholar]
- Elith J, Leathwick JR. 2009. Species distribution models: ecological explanation and prediction across space and time. Annu Rev Ecol Evol Syst 40: 677–697. https://doi.org/10.1146/annurev.ecolsys.110308.120159 [Google Scholar]
- Elith J, Leathwick JR, Hastie T. 2008. A working guide to boosted regression trees. J Anim Ecol 77: 802–813. https://doi.org/10.1111/j.1365-2656.2008.01390.x [Google Scholar]
- Emig CC. 1997. Bathyal zones of the Mediterranean continental slope: an attempt. Publ Espec Inst Esp Oceanogr 23: 23–33. [Google Scholar]
- Eno N. 2001. Effects of crustacean traps on benthic fauna. ICES J Mar Sci 58: 11–20. https://doi.org/10.1006/jmsc.2000.0984 [Google Scholar]
- Fabri M-C, Pedel L, Beuck L, Galgani F, Hebbeln D, Freiwald A. 2014. Megafauna of vulnerable marine ecosystems in French Mediterranean submarine canyons: spatial distribution and anthropogenic impacts. Deep Sea Res Part II Top Stud Oceanogr 104: 184–207. https://doi.org/10.1016/j.dsr2.2013.06.016 [Google Scholar]
- Fabri MC, Pedel L. 2020. Funiculina quadrangularis (Pennatulacea) presence and absence extracted from video, from Fabri et al 2014. Sextant. https://doi.org/10.1016/j.dsr2.2013.06.016 [Google Scholar]
- FAO. 2009. International Guidelines for the Management of Deep-sea Fisheries in the High Seas/Directives internationales sur la gestion de la pêche profonde en haute mer/ Directrices Internacionales para la Ordenación de las Pesquerías de Aguas Profundas en Alta Mar. FAO, Rome 14: 20. https://www.fao.org/documents/card/en/c/b02fc35e-a0c4-545a-86fb-4fc340e13b52 [Google Scholar]
- Farriols MT, Irlinger C, Ordines F, Palomino D, Marco-Herrero E, Soto-Navarro J, Jordà G, Mallol S, Díaz D, Martínez-Carreño N, Díaz JA, Fernandez-Arcaya U, Joher S, Ramírez-Amaro S, R de la Ballina N, Vázquez JT, Massutí E. 2022. Recovery signals of rhodolith beds since bottom trawling ban in the SCI Menorca Channel (Western Mediterranean). Diversity 14: 20. https://doi.org/10.3390/d14010020 [Google Scholar]
- Fawcett T. 2006. An introduction to ROC analysis. Pattern Recognit Lett 27: 861–874. https://doi.org/10.1016/j.patrec.2005.10.010 [CrossRef] [Google Scholar]
- Fourcade Y, Besnard AG, Secondi J. 2018. Paintings predict the distribution of species, or the challenge of selecting environmental predictors and evaluation statistics. Glob Ecol Biogeogr 27: 245–256. https://doi.org/10.1111/geb.12684 [Google Scholar]
- Fujita T, Ohta S. 1988. Photographic observations of the life style of a deep-sea ophiuroid Asteronyx loveni (Echinodermata). Deep Sea Res Part A Oceanogr Res Pap 35: 2029–2043. https://doi.org/10.1016/0198-0149(88)90123-9 [Google Scholar]
- Fourt M, Goujard A, Perez T, Chevaldonné P. 2017. Guide de la faune profonde de la mer Méditerranée. Exploration des roches et canyons sous-marins des côtes françaises. Muséum national d’Histoire naturelle. Publications scientifiques, Paris. http://sciencepress.mnhn.fr/fr/collections/patrimoines-naturels/guide-de-la-faune-profonde-de-la-mer-mediterranee [Google Scholar]
- Georges V, Vaz S, Carbonara P, Fabri MC, Fanelli E, Follesa MC, Garofalo G, Gerovasileiou V, Jadaud A, Maiorano P, Marin P, Mytilineou C, Orejas C, Del Mar Otero M, Smith CJ, Thasitis I, Lauria V. 2024. Mapping the habitat refugia of Isidella elongata under climate change and trawling impacts to preserve vulnerable marine ecosystems in the Mediterranean. Sci Rep 14: 6246. https://doi.org/10.1038/s41598-024-56338-1 [Google Scholar]
- Georgian SE, Anderson OF, Rowden AA. 2019. Ensemble habitat suitability modeling of vulnerable marine ecosystem indicator taxa to inform deep-sea fisheries management in the South Pacific Ocean. Fish Res 211: 256–274. https://doi.org/10.1016/j.fishres.2018.11.020 [Google Scholar]
- GFCM SAC, 2009. Criteria for the identification of sensitive habitats of relevance for the management of priority species. General Fisheries Commission for the Mediterranean (GFCM), Scientific Advisory Committee (SAC), Sub-Committee on Marine Environment and Ecosystems, Malaga. [Google Scholar]
- Giangrande A, Gambi MC, Gravina MF. 2020. Polychaetes as habitat former: structure and function, in: Rossi S, Bramanti L (Eds.), Perspectives on the Marine Animal Forests of the World. Springer, Cham, 2020, pp. 219–237. https://doi.org/10.1007/978-3-030-57054-5_8 [Google Scholar]
- Gori A, Ferrier-Pagès C, Hennige SJ, Murray F, Rottier C, Wicks LC, Roberts JM. 2016. Physiological response of the cold-water coral Desmophyllum dianthus to thermal stress and ocean acidification. Peer J 4: e1606. https://doi.org/10.7717/peerj.1606 [Google Scholar]
- Greathead CF, Donnan DW, Mair JM, Saunders GR. 2007. The sea pens Virgularia mirabilis, Pennatula phosphorea and Funiculina quadrangularis: distribution and conservation issues in Scottish waters. J Mar Biol Assoc UK 87: 1095–1103. https://doi.org/10.1017/S0025315407056238 [Google Scholar]
- Greathead C, González-Irusta JM, Clarke J, Boulcott P, Blackadder L, Weetman A, Wright PJ. 2015. Environmental requirements for three sea pen species: relevance to distribution and conservation. ICES J Mar Sci 72: 576–586. https://doi.org/10.1093/icesjms/fsu129 [Google Scholar]
- Greeley K. 2022. Skeletal growth ring microanalysis and growth rates in sea pens from the Laurentian Channel MPA, Newfoundland. Memorial Univ Newfoundland. https://doi.org/10.48336/t646-sy04 [Google Scholar]
- Grinyó J, Gori A, Greenacre M, Requena S, Canepa A, Lo Iacono C, Ambroso S, Purroy A, Gili JM. 2018. Megabenthic assemblages in the continental shelf edge and upper slope of the Menorca Channel, Western Mediterranean Sea. Prog Oceanogr 162: 40–51. https://doi.org/10.1016/j.pocean.2018.02.002 [Google Scholar]
- Hastie T, Tibshirani R. 1986. Generalized additive models. Stat Sci 1: 297–310. https://doi.org/10.1214/ss/1177013604 [Google Scholar]
- Hattab T, Garzón-López CX, Ewald M, Skowronek S, Aerts R, Horen H, Brasseur B, Gallet-Moron E, Spicher F, Decocq G, Feilhauer H, Honnay O, Kempeneers P, Schmidtlein S, Somers B, Van De Kerchove R, Rocchini D, Lenoir J. 2017. A unified framework to model the potential and realized distributions of invasive species within the invaded range. Divers Distrib 23: 806–819. https://doi.org/10.1111/ddi.12566 [Google Scholar]
- Hughes DJ. 1998. Sea pens and burrowing megafauna. An overview of dynamics and sensitivity characteristics for conservation management of marine SACs. Scottish Assoc Mar Sci (UK Marine SACs Project) 105. http://ukmpa.marinebiodiversity.org/uk_sacs/pdfs/seapens.pdf [Google Scholar]
- Kaiser MJ, Collie JS, Hall SJ, Jennings S, Poiner IR. 2002. Modification of marine habitats by trawling activities: prognosis and solutions. Fish Fish 3: 114–136. https://doi.org/10.1046/j.1467-2979.2002.00079.x [Google Scholar]
- Karp MA, Cimino M, Craig JK, Crear DP, Haak C, Hazen EL, Kaplan I, Kobayashi DR, Moustahfid H, Muhling B, Pinsky ML, Smith LA, Thorson JT, Woodworth-Jefcoats PA. 2025. Applications of species distribution modeling and future needs to support marine resource management. ICES J Mar Sci 82: fsaf024. https://doi.org/10.1093/icesjms/fsaf024 [Google Scholar]
- Keeling RF, Körtzinger A, Gruber N. 2010. Ocean deoxygenation in a warming world. Annu Rev Mar Sci 2: 199–229. https://doi.org/10.1146/annurev.marine.010908.163855 [Google Scholar]
- Keppel G, Mokany K, Wardell-Johnson GW, Phillips BL, Welbergen JA, Reside AE. 2015. The capacity of refugia for conservation planning under climate change. Front Ecol Environ 13: 106–112. https://doi.org/10.1890/140055 [Google Scholar]
- Lauria V, Garofalo G, Fiorentino F, Massi D, Milisenda G, Piraino S, Russo T, Gristina M. 2017. Species distribution models of two critically endangered deep-sea octocorals reveal fishing impacts on vulnerable marine ecosystems in central Mediterranean Sea. Sci Rep 7: 8049. https://doi.org/10.1038/s41598-017-08386-z [Google Scholar]
- Lauria V, Massi D, Fiorentino F, Milisenda G, Cillari T. 2021. Habitat suitability mapping of the black coral Leiopathes glaberrima to support conservation of vulnerable marine ecosystems. Sci Rep 11: 15661. https://doi.org/10.1038/s41598-021-95256-4 [Google Scholar]
- Lauria V, Gargano F, Arculeo M, Bono G, Fiorentino F, Flot JF, Collard O, Follesa MC, Garofalo G, Georges V, Maiorano P, Massi D, Stamouli C, Vaz S, Vitale S, Vecchioni L. 2025. Genetic structure and connectivity of the critically endangered deep-sea octocoral Isidella elongata: Implications for marine biodiversity conservation in the Mediterranean Sea. Medit Mar Sci 26: 1050–1067. https://doi.org/10.12681/mms.40883 [Google Scholar]
- Legendre P,& Legendre L. Numerical Ecology, 3rd ed, Elsevier, 1998. [Google Scholar]
- Lejeusne C, Chevaldonné P, Pergent-Martini C, Boudouresque CF, Pérez T. 2010. Climate change effects on a miniature ocean: the highly diverse, highly impacted Mediterranean Sea. Trends Ecol Evol 25: 250–260. https://doi.org/10.1016/j.tree.2009.10.009 [Google Scholar]
- Leroy B, Bellard C, Dubos N, Colliot A, Vasseur M, Courtial C, Bakkenes M, Canard A, Ysnel F. 2014. Forecasted climate and land use changes, and protected areas: the contrasting case of spiders. Divers Distrib 20: 686–697. https://doi.org/10.1111/ddi.12191 [Google Scholar]
- Macias D, Bisselink B, Carmona-Moreno C, Druon JN, Duteil O, Garcia-Gorriz E, Grizzetti B, Guillen J, Miladinova S, Pistocchi A, Piroddi C, Polimene L, Serpetti N, Stips A, Trichakis I, Udias A, Vigiak O. 2025. The overlooked impacts of freshwater scarcity on oceans as evidenced by the Mediterranean Sea. Nat Commun 16: 998. https://doi.org/10.1038/s41467-024-54979-4 [Google Scholar]
- Mačić V, Đorđević N, Đurović M, Petović S, Russo T. 2022. Improving knowledge of Funiculina quadrangularis and vulnerable marine ecosystems in the south Adriatic. Medit Mar Sci 23: 805–816. https://doi.org/10.12681/mms.29898 [Google Scholar]
- Manea E, Bianchelli S, Fanelli E, Danovaro R, Gissi E. 2020. Towards an ecosystem-based marine spatial planning in the deep Mediterranean Sea. Sci Total Environ 715: 136884. https://doi.org/10.1016/j.scitotenv.2020.136884 [Google Scholar]
- Maldonado M, Aguilar R, Bannister RJ, Bell JJ, Conway KW, Dayton PK, Díaz C, Gutt J, Kelly M, Kenchington ELR, Leys SP, Pomponi SA, Rapp HT, Rützler K, Tendal OS, Vacelet J, Young CM. Sponge grounds as key marine habitats: A synthetic review of types, structure, functional roles, and conservation concerns, in: Rossi S, Bramanti L, Gori A, Orejas Saco del Valle C (Eds.), Marine Animal Forests: The Ecology of Benthic Biodiversity Hotspots. Springer, Cham, 2015, pp. 1–39. https://doi.org/10.1007/978-3-319-17001-5_24-1 [Google Scholar]
- Margirier F, Testor P, Heslop E, Mallil K, Bosse A, Houpert L, Mortier L, Bouin MN, Coppola L, D’Ortenzio F, Durrieu de Madron X, Mourre B, Prieur L, Raimbault P, Taillandier V. 2020. Abrupt warming and salinification of intermediate waters interplays with decline of deep convection in the Northwestern Mediterranean Sea. Sci Rep 10: 20923. https://doi.org/10.1038/s41598-020-77859-5 [Google Scholar]
- Margules CR, Pressey RL, 2000. Systematic conservation planning. Nature 405, 243–253. https://doi.org/10.1038/35012251 [CrossRef] [PubMed] [Google Scholar]
- Marsaglia L, Parisi A, Libralato S, Miller NA, Davis P, Paolo FS, Fiorentino F, Garofalo G, Costantini M, Russo T. 2024. Shedding light on trawl fishing activity in the Mediterranean Sea with remote sensing data. ICES J Mar Sci 82: fsae153. https://doi.org/10.1093/icesjms/fsae153 [Google Scholar]
- Martinelli M, Zacchetti L, Belardinelli A, Domenichetti F, Scarpini P, Penna P, Medvešek D, Isajlović I, Vrgoč N. 2023. Changes in abundance and distribution of the sea pen Funiculina quadrangularis in the central Adriatic Sea (Mediterranean Basin) in response to variations in trawling intensity. Fishes 8: 347. https://doi.org/10.3390/fishes8070347 [Google Scholar]
- Matos FL, Aguzzi J, Company JB, Cunha MR. 2024. Gone with the stream: Functional connectivity of a cold-water coral at basin scale. Limnol Oceanogr 69: 217–231. https://doi.org/10.1002/lno.12444 [Google Scholar]
- Maynou F, Cartes JE. 2012. Effects of trawling on fish and invertebrates from deep-sea coral facies of Isidella elongata in the western Mediterranean. J Mar Biol Assoc UK 92: 1501–1507. https://doi.org/10.1017/S0025315411001603 [Google Scholar]
- McCullagh P. 1984. Generalized linear models. Eur J Oper Res 16: 285–292. https://doi.org/10.1016/0377-2217(84)90282-0 [Google Scholar]
- MEDITS Working Group. 2017. MEDITS Handbook. Version 9, 106. https://www.sibm.it/MEDITS%202011/docs/Medits_Handbook_2017_version_9_5-60417r.pdf [Google Scholar]
- Mesgaran MB, Cousens RD, Webber BL. 2014. Here be dragons: a tool for quantifying novelty due to covariate range and correlation change when projecting species distribution models. Divers Distrib 20: 1147–1159. https://doi.org/10.1111/ddi.12209 [Google Scholar]
- Millot J, Georges V, Lauria V, Hattab T, Dominguez-Carrió C, Gerovasileiou V, Smith CJ, Mytilineou C, Teresa Farriols M, Fabri MC, Carbonara P, Massi D, Rinelli P, Profeta A, Chimienti G, Jadaud A, Thasitis I, Camilleri K, Mifsud J, Vaz S. 2024. Habitat shifts of the vulnerable crinoid Leptometra phalangium under climate change scenarios. Prog Oceanogr 229: 103355. https://doi.org/10.1016/j.pocean.2024.103355 [Google Scholar]
- Millot J, Vaz S. 2024. Modelled seabed sediment average grain size in the European North-East Atlantic and Mediterranean waters. Sextant. https://doi.org/10.12770/79050557-6f8a-48cc-9e60-4112cae9fd18 [Google Scholar]
- Morato T, González-Irusta JM, Dominguez-Carrió C, Wei CL, Davies A, Sweetman AK, Taranto GH, Beazley L, García-Alegre A, Grehan A, Laffargue P, Murillo FJ, Sacau M, Vaz S, Kenchington E, Arnaud-Haond S, Callery O, Chimienti G, Cordes E, Egilsdottir H, Freiwald A, Gasbarro R, Gutiérrez-Zárate C, Gianni M, Gilkinson K, Wareham Hayes VE, Hebbeln D, Hedges K, Henry LA, Johnson D, Koen-Alonso M, Lirette C, Mastrototaro F, Menot L, Molodtsova T, Durán Muñoz P, Orejas C, Pennino MG, Puerta P, Ragnarsson SÁ, Ramiro-Sánchez B, Rice J, Rivera J, Roberts JM, Ross SW, Rueda JL, Sampaio Í, Snelgrove P, Stirling D, Treble MA, Urra J, Vad J, van Oevelen D, Watling L, Walkusz W, Wienberg C, Woillez M, Levin LA, Carreiro-Silva M. 2020. Climate-induced changes in the suitable habitat of cold-water corals and commercially important deep-sea fishes in the North Atlantic. Glob Change Biol 26: 2181–2202. https://doi.org/10.1111/gcb.14996 [Google Scholar]
- Murillo F, Kenchington E, Tompkins G, Beazley L, Baker E, Knudby A, Walkusz W. 2018. Sponge assemblages and predicted archetypes in the eastern Canadian Arctic. Mar Ecol Prog Ser 597: 115–135. https://doi.org/10.3354/meps12589 [Google Scholar]
- Owens HL, Campbell LP, Dornak LL, Saupe EE, Barve N, Soberón J, Ingenloff K, Lira-Noriega A, Hensz CM, Myers CE, Peterson AT. 2013. Constraints on interpretation of ecological niche models by limited environmental ranges on calibration areas. Ecol Model 263: 10–18. https://doi.org/10.1016/j.ecolmodel.2013.04.011 [Google Scholar]
- Pastor F, Valiente JA, Khodayar S. 2020. A warming Mediterranean: 38 years of increasing sea surface temperature. Remote Sens 12: 2687. https://doi.org/10.3390/rs12172687 [Google Scholar]
- Pellissier L, Bråthen KA, Vittoz P, Yoccoz NG, Dubuis A, Meier ES, Zimmermann NE, Randin CF, Thuiller W, Garraud L, Van Es J, Guisan A. 2013. Thermal niches are more conserved at cold than warm limits in arctic-alpine plant species. Glob Ecol Biogeogr 22: 933–941. https://doi.org/10.1111/geb.12057 [Google Scholar]
- Pérès JM, Picard J. Nouveau Manuel de bionomie benthique de la mer Méditerranée, in: Recueil Des Travaux de La Station Marine d’Endoume, 1964, p. 137. [Google Scholar]
- Peterson AT, Cobos ME, Jiménez-García D. 2018. Major challenges for correlational ecological niche model projections to future climate conditions. Ann NY Acad Sci 1429: 66–77. https://doi.org/10.1111/nyas.13873 [Google Scholar]
- Pierdomenico M, Russo T, Ambroso S, Gori A, Martorelli E, D’Andrea L, Gili JM, Chiocci FL. 2018. Effects of trawling activity on the bamboo-coral Isidella elongata and the sea pen Funiculina quadrangularis along the Gioia Canyon (Western Mediterranean, southern Tyrrhenian Sea). Prog Oceanogr 169: 214–226. https://doi.org/10.1016/j.pocean.2018.02.019 [Google Scholar]
- Purkey SG, Johnson GC. 2010. Warming of global abyssal and deep Southern Ocean waters between the 1990s and 2000s: Contributions to global heat and sea level rise budgets. J Clim 23: 6336–6351. https://doi.org/10.1175/2010JCLI3682.1 [Google Scholar]
- Robert K, Jones DOB, Roberts JM, Huvenne VAI. 2016. Improving predictive mapping of deep-water habitats: Considering multiple model outputs and ensemble techniques. Deep Sea Res Part I Oceanogr Res Pap 113: 80–89. https://doi.org/10.1016/j.dsr.2016.04.008 [Google Scholar]
- Rodríguez-Rodríguez D, Rodríguez J, Abdul Malak D, Nastasi A, Hernández P. 2016. Marine protected areas and fisheries restricted areas in the Mediterranean: assessing “actual” marine biodiversity protection coverage at multiple scales. Mar Policy 64: 24–30. https://doi.org/10.1016/j.marpol.2015.11.006 [Google Scholar]
- Roger S, Vaz S, Tessier E, Cheret I, Verin Y, Ancellin J, Peres JM. 2024. Catch data from a 1949 trawl survey in Tunisia (South-West Mediterranean). SEANOE. https://doi.org/10.17882/99868 [Google Scholar]
- Russo E, Monti MA, Mangano MC, Raffaetà A, Sarà G, Silvestri C, Pranovi F. 2020. Temporal and spatial patterns of trawl fishing activities in the Adriatic Sea (Central Mediterranean Sea, GSA17). Ocean Coast Manage 192: 105231. https://doi.org/10.1016/j.ocecoaman.2020.105231 [Google Scholar]
- S, Vaz S, Tessier E, Cheret I, Verin Y, Ancellin J, Peres JM. 2024. Catch data from a 1949 trawl survey in Tunisia (South-West Mediterranean). SEANOE. https://doi.org/10.17882/99868 [Google Scholar]
- Salgado-Hernanz PM, Regaudie-de-Gioux A, Antoine D, Basterretxea G. 2022. Pelagic primary production in the coastal Mediterranean Sea: variability, trends, and contribution to basin-scale budgets. Biogeosciences 19: 47–69. https://doi.org/10.5194/bg-19-47-2022 [Google Scholar]
- Salomidi M, Gerovasileiou V, Stamouli C, Drakopoulou V, Otero MM, Jimenez C, Kiparissis S, Mytilineou Ch, Papadopoulou N, Smith CJ, Thasitis I, Anastasiadou Ch, Lefkaditou E, Makovsky Y, Schüler M. Deep-sea vulnerable benthic fauna, in: Otero M, Mytilineou C (Eds.), Deep-Sea Atlas of the Eastern Mediterranean Sea. IUCN-HCMR DeepEastMed Project, IUCN, Gland and Malaga, 2022, pp 123–144. [Google Scholar]
- Salvalaggio V, Fabi G, Punzo E. 2016. Distribution of the sea pens Virgularia mirabilis and Funiculina quadrangularis (Cnidaria Anthozoa) in the northern and central Adriatic Sea. Rapport Du 41e Congres De La CIESM [Google Scholar]
- Schickele A, Leroy B, Beaugrand G, Goberville E, Hattab T, Francour P, Raybaud V. 2020. Modelling European small pelagic fish distribution: Methodological insights. Ecol Model 416: 108902. https://doi.org/10.1016/j.ecolmodel.2019.108902 [Google Scholar]
- Skliris N, Zika JD, Herold L, Josey SA, Marsh R. 2018. Mediterranean sea water budget long-term trend inferred from salinity observations. Clim Dyn 51: 2857–2876. https://doi.org/10.1007/s00382-017-4053-7 [Google Scholar]
- Smith CJ, Gerovasileiou V, Mytilineou Ch, Jimenez C, Papadopoulou K, Salomidi M, Sakellariou D, Drakopoulou V, Otero M. 2022. Revisiting underwater surveys to uncover sites of conservation interest, in: Otero M, Mytilineou C (Eds.), Deep-Sea Atlas of the Eastern Mediterranean Sea. IUCN-HCMR DeepEastMed Project, IUCN, Gland and Malaga, pp. 146–171. [Google Scholar]
- Smith KL, Ruhl HA, Kahru M, Huffard CL, Sherman AD. 2013. Deep ocean communities impacted by changing climate over 24 y in the abyssal northeast Pacific Ocean. Proc Natl Acad Sci USA 110: 19838–19841. https://doi.org/10.1073/pnas. 1315447110 [Google Scholar]
- Sweetman AK, Thurber AR, Smith CR, Levin LA, Mora C, Wei CL, Gooday AJ, Jones DOB, Rex M, Yasuhara M, Ingels J, Ruhl HA, Frieder CA, Danovaro R, Würzberg L, Baco A, Grupe BM, Pasulka A, Meyer KS, Dunlop KM, Henry LA, Roberts JM. 2017. Major impacts of climate change on deep-sea benthic ecosystems. Elem Sci Anthropocene 5: 4. https://doi.org/10.1525/elementa.203 [Google Scholar]
- Terribile K, Evans J, Knittweis L, Schembri PJ. 2016. Maximising MEDITS: Using data collected from trawl surveys to characterize the benthic and demersal assemblages of the circalittoral and deeper waters around the Maltese Islands (Central Mediterranean). Reg Stud Mar Sci 3:163–175. https://doi.org/10.1016/j.rsma.2015.07.006 [Google Scholar]
- Thuiller W, Georges D, Engler R, Breiner F. 2023. biomod2: ensemble platform for species distribution modeling. R package ver 4.2-4 [Google Scholar]
- Tudela S. 2004. Ecosystem effects of fishing in the Mediterranean: An analysis of the major threats of fishing gear and practices to biodiversity and marine habitats. Gen Fish Counc Medit Stud Rev 74. FAO, Rome, 44 pp. [Google Scholar]
- UNGA. 2006. Resolution 61/105 adopted by the General Assembly on Sustainable fisheries, including through the 1995 Agreement for the Implementation of the Provisions of the United Nations Convention on the Law of the Sea of 10 December 1982 relating to the Conservation and Management of Straddling Fish Stocks and Highly Migratory Fish Stocks. UN Gen Assem Res 61/105. https://doi.org/10.18356/9789210021753 [Google Scholar]
- Vasquez M, Ségeat B, Cordingley A, Tilby E, Wikström S, Ehrnsten E, Al Hamdani Z, Agnesi S, Andersen MS, Annunziatellis A, Askew N, Bekkby T, Bentes L, Daniels E, Doncheva V, Drakopoulou V, Ernstsen VB, Gonçalves J, Karvinen V, Laamanen-Nicolas L, Lillis H, Loukaidi V, Manca E, McGrath F, Mo G, Monteiro P, Muresan M, Nygard H, O’Keeffe E, Pelembe T, Radicioli M, Sakellariou D, Teaca A, Todorova V, Tunesi L, Woods H. 2023. EUSeaMap 2023, A European broad-scale seabed habitat map. https://doi.org/10.13155/97116 [Google Scholar]
- Vaz S. 2018a. EPIBENGOL 2018 cruise, RV L’Europe. https://doi.org/10.17600/18000589 [Google Scholar]
- Vaz S. 2018b. NOURMED. https://doi.org/10.18142/296 [Google Scholar]
- Vaz S, Certain G, Hattab T, Jadaud A, Villeneuve R, Raphalen E, Metral L, Cheret I, Bourdeix JH, Tessier E. 2023. Suivi spatio-temporel des espèces démersales et benthiques: Rapport final de l’action 3 du Projet GOLDYS. https://doi.org/10.13155/96823 [Google Scholar]
- Wang S, Murillo FJ, Kenchington E. 2022. Climate-change refugia for the bubblegum coral Paragorgia arborea in the Northwest Atlantic. Front Mar Sci 9: 863693. https://doi.org/10.3389/fmars.2022.863693 [Google Scholar]
- Wright EP, Kemp K, Rogers AD, Yesson C. 2015. Genetic structure of the tall sea pen Funiculina quadrangularis in NW Scottish sea lochs. Mar Ecol 36: 659–667. https://doi.org/10.1111/maec.12174 [Google Scholar]
Cite this article as: Millot J, Lauria V, Georges V, Smith CJ, Dominguez-Carrió C, Evangelopoulos A, Gerovasileiou V, Chimienti G, Fanelli E, Evans J, Casini M, Fabri M-C, Carbonara, P, Rinelli P, Massi D, Profeta A, Jadaud A, Manfredi C, Stamouli C, Mytilineou C, Farriols MT, Musumeci C, Sbrana M, Petović S, Pisani L, Agius D, Kamidis N, Thasitis I, Kolitari J, Vaz S. 2026. Climate-driven habitat shifts of the vulnerable sea pen Funiculina quadrangularis in the Mediterranean Sea. Aquat. Living Resour. 39: 11. https://doi.org/10.1051/alr/2026004
Appendix
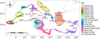 |
Fig. A1 Map of the GSAs (geographical sub-areas) defined by the General Fisheries Commission for the Mediterranean (GFCM), colored according to their respective sea/region within the Mediterranean basin. |
 |
Fig. A2 Correlation matrix among all available environmental predictors based on Spearman’s rank coefficient. Positive correlations are colored in blue and negative correlations in red. Coefficients higher than 0.7 are crossed. See Appendix B, Table B2 for the meaning of the abbreviations. |
 |
Fig. A3 Environmental range of a resampled presence–absence dataset of Funiculina quadrangularis records in the Mediterranean basin. |
 |
Fig. A4 Maps of the four environmental predictors used in the models to predict the potential distribution of Funiculina quadrangularis across the Mediterranean basin (A) under present conditions and under the SSP2–4.5 scenario of the IPCC (B) by 2050 and (C) by 2100. |
 |
Fig. A5 Proportion of total variance explained by (A) dataset, (B) spatial block cross-validation, and (C) algorithm. |
 |
Fig. A6 Mean present-day predictions of the selected models for each algorithm: (A) GLM, (B) GAM, (C) RF, and (D) GBM. |
 |
Fig. A7 Probabilities of presence (left) converted into binary presence–absence data (right) using a threshold of 0.52, illustrating the distribution of the potential habitat of F. quadrangularis in the Mediterranean basin under the IPCC scenario SSP1–2.6 (A–B) by 2050 and (C–D) by 2100. |
 |
Fig. A8 Probabilities of presence (left) converted into binary presence–absence data (right) using a threshold of 0.52, illustrating the distribution of the potential habitat of F. quadrangularis in the Mediterranean basin under the IPCC scenario SSP5–8.5, are (A–B) by 2050 and (C–D) by 2100. |
 |
Fig. A9 Areas predicted as climate refugia, habitat gain, habitat loss, or unsuitable for F. quadrangularis under the IPCC SSP1–2.6 scenario (left) and the IPCC SSP5–8.5 scenario (right) are (A–B) by 2050 and (C–D) by 2100. Climate refugia are defined as areas that remain suitable under both present and future conditions; habitat gain refers to currently unsuitable areas that become suitable in the future, and habitat loss are suitable areas becoming unsuitable. |
 |
Fig. A10 Environmental extrapolations associated with the predictions under the IPCC scenario SSP1–2.6 (left) and the IPCC SSP5–8.5 scenario (right) are (A–B) by 2050 and (C–D) by 2100. For each period, univariate extrapolations (i.e., projections outside the range of the environmental space of calibrating data for at least one predictor) are shown in orange, combinatorial extrapolations (i.e., projections in unknown combinations of predictors) in purple, and analogue conditions (i.e., projections in environmental conditions already known by calibrating data) in green. Areas with no sampled data are shown with faded shading, and projections in such areas must be interpreted with caution because of higher uncertainties. |
Appendix
Metadata of raw presence and absence records of Funiculina quadrangularis before any filtering process.
Metadata of candidate environmental predictors explored for this study. Variables selected for the final ensemble modeling process are highlighted in grey. Abbr. = Abbreviation; Native res. = Native resolution; Ecol. sel. = Ecological selection; Cor. sel. = Correlation selection; and BIC GAM = BIC selection on GAM. Native resolution given in decimal degrees. Mean = Long-term average of monthly bottom values for the period 2010-2020. Long-term minimum = Long-term average of the minimum value of the bottom variable per year for the period 2010–2020. Long-term maximum = Long-term average of the maximum value of the bottom variable per year for the period 2010–2020. Range = Range of the bottom variable for the period 2010–2020 (i.e., average of the absolute difference between the minimum and maximum value of the variable per year).
Metadata of environmental layers projected in future conditions under IPCC climate scenario used in the study.
All Tables
Metadata of raw presence and absence records of Funiculina quadrangularis before any filtering process.
Metadata of candidate environmental predictors explored for this study. Variables selected for the final ensemble modeling process are highlighted in grey. Abbr. = Abbreviation; Native res. = Native resolution; Ecol. sel. = Ecological selection; Cor. sel. = Correlation selection; and BIC GAM = BIC selection on GAM. Native resolution given in decimal degrees. Mean = Long-term average of monthly bottom values for the period 2010-2020. Long-term minimum = Long-term average of the minimum value of the bottom variable per year for the period 2010–2020. Long-term maximum = Long-term average of the maximum value of the bottom variable per year for the period 2010–2020. Range = Range of the bottom variable for the period 2010–2020 (i.e., average of the absolute difference between the minimum and maximum value of the variable per year).
Metadata of environmental layers projected in future conditions under IPCC climate scenario used in the study.
All Figures
 |
Fig. 1 Map of the study area showing the raw presence (A) and absence (B) records of Funiculina quadrangularis; records are color-coded according to the data source. Blue-shaded regions represent areas where depth is <1000 m. |
| In the text | |
 |
Fig. 2 Model performance evaluation with (A) Area Under the Curve (AUC) and True Skill Statistic (TSS) scores, with the selected models highlighted by bold circles; (B) specificity and sensitivity scores; and (C) contributions of environmental predictors. Models are color-coded by algorithm. 'Salinity_ltmax refers to the long-term maximum salinity, 'temp_mean' to the mean bottom temperature, and 'o2_ltmin' to the long-term minimum oxygen level. |
| In the text | |
 |
Fig. 3 Response curves of the probability of presence of F. quadrangularis as a function of the different environmental predictors selected, coloured by algorithm: (A) depth, (B) mean bottom temperature, (C) long-term maximum bottom salinity, and (D) long-term minimum bottom oxygen. The solid line represents the mean response curve across individual models of each algorithm, with shaded areas indicating the 95% confidence interval. |
| In the text | |
 |
Fig. 4 Habitat suitability (left) converted to binary presence–absence values (right) by an optimal threshold of 0.52, illustrating the potential areas that are suitable for F. quadrangularis in the Mediterranean basin for (A–B) the present-day conditions and under the IPCC scenario SSP2–4.5 (C–D) by 2050 and (E–F) by 2100. |
| In the text | |
 |
Fig. 5 Areas projected as climate refugia, habitat gain, habitat loss, or unsuitable for F. quadrangularis under the IPCC SSP2–4.5 scenario by (A) 2050 and (B) 2100. Climate refugia are defined as areas that remain suitable under both present and future conditions; habitat gain refers to currently unsuitable areas that become suitable in the future, and habitat loss to suitable areas becoming unsuitable. |
| In the text | |
 |
Fig. 6 Prediction uncertainties are represented by (A) the standard deviation (sd) among individual model predictions combined in the ensemble model and (B) the ensemble model residuals, with negative residuals shown in red and positive residuals in blue. |
| In the text | |
 |
Fig. 7 Environmental extrapolations associated with (A) predictions under present-day conditions, (B) projections under the IPCC SSP2–4.5 scenario by 2050, and (C) by 2100. For each period, univariate extrapolations (i.e., predictions/projections outside the range of the environmental space of calibrating data for at least one predictor) are mapped in orange, and analogous conditions (i.e., predictions/projections in environmental conditions already present in calibrating data) are shown in green. Areas with no sampled data are shown with faded shading, and predictions/projections in such areas must be interpreted with caution, being associated with higher uncertainties. |
| In the text | |
 |
Fig. A1 Map of the GSAs (geographical sub-areas) defined by the General Fisheries Commission for the Mediterranean (GFCM), colored according to their respective sea/region within the Mediterranean basin. |
| In the text | |
 |
Fig. A2 Correlation matrix among all available environmental predictors based on Spearman’s rank coefficient. Positive correlations are colored in blue and negative correlations in red. Coefficients higher than 0.7 are crossed. See Appendix B, Table B2 for the meaning of the abbreviations. |
| In the text | |
 |
Fig. A3 Environmental range of a resampled presence–absence dataset of Funiculina quadrangularis records in the Mediterranean basin. |
| In the text | |
 |
Fig. A4 Maps of the four environmental predictors used in the models to predict the potential distribution of Funiculina quadrangularis across the Mediterranean basin (A) under present conditions and under the SSP2–4.5 scenario of the IPCC (B) by 2050 and (C) by 2100. |
| In the text | |
 |
Fig. A5 Proportion of total variance explained by (A) dataset, (B) spatial block cross-validation, and (C) algorithm. |
| In the text | |
 |
Fig. A6 Mean present-day predictions of the selected models for each algorithm: (A) GLM, (B) GAM, (C) RF, and (D) GBM. |
| In the text | |
 |
Fig. A7 Probabilities of presence (left) converted into binary presence–absence data (right) using a threshold of 0.52, illustrating the distribution of the potential habitat of F. quadrangularis in the Mediterranean basin under the IPCC scenario SSP1–2.6 (A–B) by 2050 and (C–D) by 2100. |
| In the text | |
 |
Fig. A8 Probabilities of presence (left) converted into binary presence–absence data (right) using a threshold of 0.52, illustrating the distribution of the potential habitat of F. quadrangularis in the Mediterranean basin under the IPCC scenario SSP5–8.5, are (A–B) by 2050 and (C–D) by 2100. |
| In the text | |
 |
Fig. A9 Areas predicted as climate refugia, habitat gain, habitat loss, or unsuitable for F. quadrangularis under the IPCC SSP1–2.6 scenario (left) and the IPCC SSP5–8.5 scenario (right) are (A–B) by 2050 and (C–D) by 2100. Climate refugia are defined as areas that remain suitable under both present and future conditions; habitat gain refers to currently unsuitable areas that become suitable in the future, and habitat loss are suitable areas becoming unsuitable. |
| In the text | |
 |
Fig. A10 Environmental extrapolations associated with the predictions under the IPCC scenario SSP1–2.6 (left) and the IPCC SSP5–8.5 scenario (right) are (A–B) by 2050 and (C–D) by 2100. For each period, univariate extrapolations (i.e., projections outside the range of the environmental space of calibrating data for at least one predictor) are shown in orange, combinatorial extrapolations (i.e., projections in unknown combinations of predictors) in purple, and analogue conditions (i.e., projections in environmental conditions already known by calibrating data) in green. Areas with no sampled data are shown with faded shading, and projections in such areas must be interpreted with caution because of higher uncertainties. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


