| Issue |
Aquat. Living Resour.
Volume 39, 2026
Special Issue - Interdisciplinary issues on Manila and European clams in their socio-ecosystems
|
|
|---|---|---|
| Article Number | 5 | |
| Number of page(s) | 20 | |
| DOI | https://doi.org/10.1051/alr/2025023 | |
| Published online | 10 February 2026 | |
Research Article
Contrasting responses of Ruditapes philippinarum and Mytilus galloprovincialis to gadolinium and salinity stress
1
Department of Biology, University of Aveiro
Aveiro, 3810-193 Portugal
2
Centre for Environmental and Marine Studies (CESAM), University of Aveiro Aveiro, 3810-193 Portugal
3
REQUIMTE & Department of Chemistry, University of Aveiro Aveiro, 3810-193 Portugal
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
28
September
2025
Accepted:
4
December
2025
Abstract
The widespread use of gadolinium‐based contrast agents (GBCAs) in medical imaging has raised concerns regarding their persistence in aquatic environments and potential ecotoxicological impacts. This study evaluated the effects of gadolinium (Gd) exposure, under environmentally relevant concentrations (10 µg/L), on two ecologically and economically important bivalves from the Ria de Aveiro, Ruditapes philippinarum and Mytilus galloprovincialis, while considering the role of salinity fluctuations (30 and 40) associated with climate change. Organisms were exposed for 14 days and a suite of biochemical markers was analysed, including antioxidant and biotransformation enzymes, energy metabolism, neurotoxicity, oxidative damage, and osmoregulation. Both species bioaccumulated Gd, with clams showing higher tissue concentrations at salinity 30 compared to 40, while mussels accumulated lower levels overall. Multivariate analyses revealed species‐specific biochemical responses. Clams primarily mobilised energy reserves and detoxification pathways under moderate salinity, while at hypersalinity they shifted towards antioxidant defenses (SOD, CAT), indicating oxidative stress management. In contrast, mussels showed an energy‐conserving profile: at salinity 30 Gd depressed SOD and induced CbEs, while at salinity 40 both antioxidant (SOD, CAT) and conjugation (GSTs) activities were reduced. Integrated Biomarker Response Index version 2 (IBRv2) peaked at salinity 40 (with and without Gd), confirming salinity as the dominant driver of response. These findings demonstrate that salinity modulates Gd toxicity and highlight interspecific differences in resilience strategies. Overall, the results underscore the importance of integrating emerging pollutants and climate‐related stressors in ecotoxicological assessments to better predict risks to marine ecosystems.
Key words: Bivalves / gadolinium / oxidative stress / biochemical performance
Handling Editor: Stefania Chiesa
© I. Moreira et al., Published by EDP Sciences 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Marine and coastal systems are threatened by a wide array of pollutants, spanning from traditional contaminants, trace metals and hydrocarbons, to more recently recognised substances known as emerging pollutants (EPs). Emerging pollutants are anthropogenic compounds that are not yet consistently monitored or regulated, yet they possess the potential to cause considerable harm to ecosystems and human health (Li et al., 2024). They frequently arise from industrial activities, pharmaceuticals, personal care products, and cutting‐edge technologies, which introduce by‐products and chemical compounds into the environment (Sauvé and Desrosiers, 2014; Patel et al., 2020). Among EPs, rare earth elements (REEs), such as gadolinium (Gd), stand out for their dual nature. While they are essential for advanced technological and medical applications, they also persist as pollutants in natural ecosystems (Trapasso et al., 2021). Gadolinium exemplifies the environmental challenges posed by EPs. Although naturally present at trace levels, Gd has seen increased anthropogenic input due to its widespread use in gadolinium‐based contrast agents (GBCAs) for magnetic resonance imaging (Rogowska et al.,2018; Brünjes and Hofmann, 2020). These agents, introduced in the 1980s, utilize Gd's paramagnetic properties to enhance diagnostic accuracy. Despite being biologically inert in medical settings, GBCAs are highly stable, resist degradation, and are poorly removed by conventional wastewater treatment plants. As a result, they accumulate in rivers, lakes, and coastal waters, often exceeding natural background levels (Laczovics et al., 2023).
Background Gd concentrations in pristine waters are typically in the low range (1–10 ng/L), while anthropogenically influenced systems, particularly rivers near large hospitals or densely populated areas, can reach 100–1000 ng/L, with hotspots occasionally surpassing 1 µg /L (see for review, Moreira et al., 2025). In estuarine and coastal areas, positive Gd anomalies have been repeatedly reported, confirming its persistence and conservative transport over long distances. Consequently, dissolved Gd concentrations in urban‐impacted estuaries are frequently one to two orders of magnitude higher than natural baselines (see for review Moreira et al., 2025). In marine invertebrates, particularly bivalves and crustaceans, reported Gd concentrations in tissues range from trace levels (<0.01 µg /g) up to values exceeding 0.4 µg /g, with Mytilus galloprovincialis and Scrobicularia plana (Moreira et al., 2025; Rétif et al., 2024a,b) among the most studied taxa. Regional comparisons reveal higher concentrations in organisms from estuaries subjected to intense urban and hospital effluents (Santos et al., 2023), underscoring the role of filter‐feeding invertebrates as sensitive bioindicators. Their high filtration rates, combined with direct exposure to dissolved and particle‐bound Gd, make them especially prone to bioaccumulation, which may translate into oxidative stress, impaired growth, and reproductive disturbances (Hanana et al., 2017; Perrat et al., 2017; Spampinato et al., 2024).
The persistence of Gd in marine and freshwater systems poses notable ecological risks. Laboratory and field studies demonstrate that Gd can bioaccumulate in algae, bivalves, and fish, impairing vital physiological processes (for review, see Moreira et al., 2025). For instance, exposure has been associated with reduced algal photosynthetic efficiency, disruption of calcium‐dependent pathways in fish, and altered filtration and energy metabolism in bivalves (Telgmann et al., 2012; Schmidt et al., 2019). In mussels, Gd exposure has been linked to oxidative stress, increased lipid peroxidation, and induction of antioxidant enzymes, indicating a shift in redox homeostasis (Henriques et al., 2019; Andrade et al., 2022, 2023). Moreover, Gd can compete with essential elements such as Ca2+ and Mg2+, interfering with enzyme activity, ion regulation, and membrane stability in aquatic organisms. Chronic exposure may therefore compromise growth, reproduction, and ecosystem stability (Rogowska et al., 2018).
Pollutants’ behavior, bioavailability, and toxicity might change according to environmental factors, including salinity, which is closely related to ongoing climate changes such as increased temperatures, rising sea levels, and extreme weather events (IPCC, 2023). Increased precipitation, droughts, and sea level rise contribute to abrupt salinity shifts: freshwater inputs lower salinity, while evaporation and reduced river flow induce hypersaline conditions. These changes can exceed the physiological tolerances of many marine species, especially in shallow habitats (IPCC, 2019). Salinity shifts also influence pollutant dynamics by altering solubility, speciation, and bioavailability of metals. Such conditions can enhance contaminant uptake and toxicity in marine organisms, weakening antioxidant defenses and increasing lipid peroxidation, an indicator of cellular damage (Andrade et al., 2022; Chen et al., 2024). These combined stressors reduce resilience and threaten ecosystem services (IPCC, 2023). Addressing these multifactorial threats requires interdisciplinary strategies, robust emission reductions, pollutant regulation, and international cooperation to safeguard marine ecosystems and ensure the sustainability of natural systems and human societies (Gissi et al., 2021; Zitoun et al., 2024).
Salinity perturbations also disrupt species distributions, trophic interactions, and physiological stability. Bivalves respond to osmotic stress by closing their valves, a strategy to limit ion imbalance. However, chronic salinity fluctuations impair feeding, digestion, nutrient absorption, and increase metabolic costs, diverting energy from growth and reproduction. Long‐term exposure is also linked to oxidative stress, enzyme disruption, and cytotoxicity (Carregosa et al., 2014; Rivera‐Ingraham and Lignot, 2017). Therefore, ecotoxicological assessments must integrate climate variability, oceanographic shifts, and species‐specific responses to accurately predict ecological risks. A multidimensional and adaptive governance approach is essential to mitigate the compounded effects of salinity instability, pollution, and climate change on marine ecosystems.
Considering the available literature on ecological risks associated with Gd, this study aimed to: i) investigate, under controlled laboratory conditions, the ecotoxicological effects of Gd on two ecologically and economically important bivalves from the Ria de Aveiro, Ruditapes philippinarum and M. galloprovincialis, with particular attention to species‐specific biochemical responses; ii) examine how climate change‐related stressors, specifically salinity fluctuations, influence the accumulation and toxicity of Gd, thereby providing insight into how environmental variability modulates contaminant impacts in marine bivalves. To enable a true interspecific comparison, mussels and clams were exposed simultaneously under identical Gd and salinity conditions, overcoming the limitations of previous mussel‐only studies performed under different exposure settings.
2 Materials and methods
2.1 Sampling area
Ruditapes philippinarum (length: 37.54 ± 2.64 mm; width: 25.18 ± 1.87 mm), and Mytilus galloprovincialis (length: 31.57 ± 1.48 mm; width: 57.02 ± 2.82 mm) were sampled from Mira Channel, in Ria de Aveiro, a coastal lagoon located in northwestern Portugal. The broad geographic distribution and high population density of bivalves, coupled with their ease of collection and long lifespan, render them particularly valuable and desirable organisms for scientific research (Boening, 1999; Dame, 2012; Zuykov et al., 2013). Salinity levels in the lagoon vary considerably, ranging from values below 5 in inner channels distant from the ocean inlet to between 30 and 34 near the connection with the Atlantic Ocean (Dias et al., 2000; Lopes et al., 2021). However, the lagoon is susceptible to extreme weather conditions, including intense rainfall and prolonged droughts, which induce significant fluctuations in both temperature and salinity (Lopes et al., 2021).
2.2 Depuration and acclimation
For depuration, clams and mussels were kept separately for one week in artificial seawater (prepared with deionized water and commercial Red Sea® salt), under continuous aeration, a natural photoperiod, at controled temperature (approximately 17 ± 1°C), with a pH of 8.0 ± 0.1 and a salinity of 30 ± 1. These conditions were selected based on the salinity measured in the Mira Channel at the time of collection. The water was renewed every two days, and proteins and organic compounds were continuously removed using a protein skimmer. After the first week of depuration, clams and mussels were divided into two groups and kept for an additional week for acclimation to two different salinities (30 and 40). Salinity levels were gradually increased every two days until the target values were reached, while maintaining controled temperature (17 ± 1°C). Throughout this period, organisms were kept under continuous aeration, a natural photoperiod, at a pH of 8.0 ± 0.1, and were fed every two days with a mixture of three algae species (Tetraselmis chuii (Phyto Algae), Isochrysis aff. galbana (Iso Prime), and Phaeodactylum tricornutum (Phyto Algae)) at a concentration of 150,000 cells per bivalve per day. During this period, mortality was checked regularly, clams and mussels were considered dead when their shells remained open and did not close after an external stimulus.
2.3 Experimental setup
For the experimental exposure, Gd concentrations were selected based on available literature reporting its occurrence in marine and coastal waters. Background levels in seawater are typically in the range of 1 to 10 µg/L (Moreira et al., 2025). Considering these findings, and following previous studies conducted with bivalves under similar exposure levels (Hanana et al., 2017; Perrat et al., 2017), a concentration of 10 µg/L Gd was chosen to assess its effects on both species. The Gd was administered as gadolinium(III) chloride (GdCl₃) at the desired concentration.
The salinity values were selected considering records from the Ria de Aveiro, as well as the climatic events predicted in climate change scenarios. In the Mira Channel, salinity can vary between 10 and 38, approximately (Santos et al., 2007). The control salinity (30) was chosen considering the mean salinity values at the sampling site (Henriques et al., 2006; Vaz et al., 2019) and according to the control values used in other similar studies (Velez et al., 2017; Coppola et al., 2020; Freitas et al., 2020). A salinity of 40 was chosen to simulate extreme weather events such as periods of drought or high evaporation, which can lead to increased salinity in coastal and estuarine environments (IPCC, 2023).
After acclimation, similarly sized clams (R. philippinarum) and mussels (M. galloprovincialis) were used to minimize the influence of body size on Gd responses. Separately, they were exposed to selected concentrations of Gd: CTL, aquaria with clams or mussels, both without Gd, under control salinity (30); and salinity 40, Gd aquaria with clams or mussels, both with 10 µg/L of Gd, under control salinity (30) and salinity 40. For each treatment, three replicate aquaria were used, with five clams or mussels per aquarium, each with 3 L of seawater.
The exposure lasted for 14 days, the same exposure period used in other REE studies with bivalves (Cunha et al., 2022). During this period, the organisms were maintained in artificial seawater, under continuous aeration, a natural photoperiod, at a temperature of approximately 17 ± 1°C, and were fed three times per week with the same mixture of algae used in acclimation at a concentration of 150,000 cells per bivalve per day. The water was renewed and re‐spiked with Gd once a week, and mortality was monitored regularly.
At the end of the exposure, all organisms were collected and immediately frozen in liquid nitrogen and stored at ‐80 °C for future analysis. For biochemical analysis, 36 clams (nine per treatment, three per aquarium) and 36 mussels (nine per treatment, three per aquarium) were used. Each organism was removed from the shell, and the whole soft tissue was individually and manually homogenized while still frozen. For this procedure, a mortar and pestle were used under liquid nitrogen. Each homogenized organism was divided into 0.3 g fresh weight (FW) aliquots for biochemical analyzing, using a 1:2 (w/v) ratio of tissue to buffer. The remaining tissue of one organism per aquarium (three per treatment) was used for Gd quantification.
2.4 Quantification of gadolinium in soft tissues
Approximately 200 mg of freeze‐dried tissue samples were weighed into Teflon digestion vessels. To each vessel, 1 mL of 65 % (v/v) nitric acid (HNO₃), 2 mL of 30 % (v/v) hydrogen peroxide (H₂O₂), and 1 mL of ultrapure water were added. The digestion procedure was carried out in a CEM MARS 6 microwave oven, which allows precise temperature control. The temperature inside the vessels was increased to 180 °C over a 25 min ramp time and then maintained at 180 °C for an additional 5 min. After digestion, the vessels were allowed to cool to room temperature, and the digested samples were quantitatively transferred into polyethylene bottles. The volume was adjusted to 25 mL with ultrapure water, and the solutions were stored at room temperature until analysis
For quality control, digested blanks, duplicate samples, and a certified reference material were included. Gadolinium concentration in the digested blank was below the limit of quantification (0.01 µg/L). Duplicate analyzes yielded a coefficient of variation of 4 %, confirming high analytical precision. The accuracy of the method was validated using the certified reference material BCR‐668 (mussels’ tissue), with a certified Gd concentration of 13.0 µg/kg. Recovery for BCR‐668 was 108 %, attesting to the trueness of the quantification.
2.5 Biochemical responses
To investigate the biochemical changes resulting from the exposure of R. philippinarum and M. galloprovincialis to Gd and the combination of Gd with two different salinities, a suite of biomarkers was used to assess key biochemical responses. The biomarkers selected enabled the evaluation of metabolic capacity and energy reserves, through measurements of electron transport system (ETS) activity, total protein (PROT) and carbohydrates (CARB) contents. Antioxidant defense mechanism was assessed by measuring the activities of superoxide dismutase (SOD) and catalase (CAT) enzymes, while biotransformation capacity was evaluated through the activities of carboxylesterases (CbEs) and glutathione S‐transferases (GSTs) enzymes. Additionally, indicators of oxidative damage, such as lipid peroxidation (LPO) and protein carbonylation (PC) levels, were quantified. To further investigate cellular stress responses and osmoregulatory adjustments, proline (PROL) content was measured. Neurotoxic effects were assessed through the determination of acetylcholinesterase (AChE) activity. The selection of biomarkers was based on previous studies conducted with bivalves exposed to environmental contaminants and climate‐related stressors (Cunha et al., 2022; Andrade et al., 2023; Leite et al., 2024).
The buffers employed were as follows: 50 mM potassium phosphate buffer for the analysis of PROT, CARB, SOD, CAT, CbEs, GSTs, PC, and AChE; 0.1 M Tris‐HCl buffer for ETS; 20% (w/v) trichloroacetic acid (TCA) for LPO; and 3% (w/v) sulfosalicylic acid for PROL levels.
Tissue homogenates were sonicated for 90 s at a frequency of 20 1/s using a TissueLyser II (Qiagen) and subsequently centrifuged under different conditions according to the buffer used: 20 min at 10,000 g for phosphate buffer and TCA, 20 min at 3,000 g for Tris‐HCl buffer, and 10 min at 10,000 g for sulfosalicylic acid. Supernatants were collected and transferred into microtubes. Samples that were not immediately analyzed were stored at – 80 °C until further processing.
All biochemical parameters were determined in duplicate to ensure accuracy and reproducibility, with absorbance measurements performed using a microplate reader (Biotek).
2.5.1 Metabolic capacity and energy reserves
To assess metabolic capacity, ETS activity was measured following the method developed by King and Packard (1975) and modified by De Coen and Janssen (1997). Briefly, tissue extracts were incubated with the reagent INT (2‐(p‐iodophenyl)‐3‐(p‐nitrophenyl)‐5‐phenyl tetrazolium chloride), which acts as an artificial electron acceptor. Dehydrogenase enzymes, utilizing NADH and NADPH as co‐factors, reduce INT to form insoluble formazan, a colored compound. Formazan production was read for 10 min at 490 nm with 25 s intervals using the extinction coefficient (ε) of 15.9 mM−1cm−1, and the results were expressed as nmol per min per g FW.
Energy reserves were assessed by measuring CARB and PROT contents, with CARB representing a primary energy source for metabolic demands, while PROT were considered mainly for their functional and structural roles. Carbohydrate content was quantified following the sulfuric acid method described by Dubois et al. (1956). In this procedure, sugars and their derivatives react with phenol under strongly acidic conditions to form a yellow‐orange complex, the intensity of which is proportional to the amount of sugar present. Sulfuric acid hydrolyzes the sugars, enabling their quantification. After incubation for 30 min at room temperature, absorbance was measured at 492 nm, and the CARB concentration in the samples was determined using a calibration curve generated with glucose standards ranging from 0 to 5 mg /mL. The results were expressed as mg per g FW.
The total PROT content was quantified using the Biuret method, originally described by Robinson and Hogden (1940). In this colorimetric assay, the Biuret reagent reacts with peptide bonds under alkaline conditions, forming a violet‐colored complex whose intensity is proportional to the PROT concentration. Absorbance was measured at 540 nm, and the PROT concentration in the samples was determined using a calibration curve generated with bovine serum albumin (BSA) standards ranging from 0 to 40 mg /mL. Results were reported as mg per g FW.
2.5.2 Antioxidant and biotransformation capacity
Among the enzymatic defenses against reactive oxygen species (ROS), SOD and CAT play central roles. Superoxide dismutase catalyzes the dismutation of the highly reactive superoxide anion radical (O₂⁻) into H₂O₂, a less reactive but still potentially harmful molecule. Subsequently, CAT decomposes H₂O₂ into water and molecular oxygen, preventing the accumulation of H₂O₂ and the formation of even more damaging radicals through Fenton reactions (Regoli and Giuliani, 2014).
The activity of SOD was assessed according to the method described by Magnani et al. (2000), which relies on the ability of the enzyme to inhibit pyrogallol autoxidation. Tissue samples were mixed with Tris‐EDTA buffer, and the reaction was initiated by adding pyrogallol dissolved in 0.01 mmol/L HCl. Following a 1‐min incubation at room temperature, the absorbance was recorded at 420 nm. Results were expressed in U per g FW, where one unit (U) of enzyme activity indicates the inhibition of 50 % of pyrogallol autoxidation.
The activity of CAT was determined following the procedure described by Aebi (1984). In this assay, 30 mM H₂O₂ was added to the samples, and the enzymatic decomposition of H₂O₂ was monitored by measuring the decrease in absorbance at 240 nm over 2 min, with readings taken every 15 s. The molar extinction coefficient used was ε = 43.5 M⁻¹cm⁻¹. The results were expressed as µmol per min per g FW.
In addition to the antioxidant enzymes, biotransformation enzymes such as CbEs and GSTs contribute significantly to the detoxification processes. CbEs catalyze the hydrolysis of carboxylic esters into their corresponding alcohol and acid (Solé et al., 2018). The activity of CbEs was determined according to the method of Hosokawa and Satoh (2002), with adaptations introduced by Solé et al. (2018). In this assay, p‐nitrophenyl butyrate (pNPB) was used as a substrate, which upon hydrolysis releases p‐nitrophenol ions, forming a yellow chromophore. The CbEs activity was monitored at 405 nm over 5 min at 15 s intervals. For activity quantification, an extinction coefficient (ε) of 18 mM−1 cm−1 was used, and the results were expressed in nmol per min per g FW.
The activity of GSTs was assessed following the method originally described by Habig et al. (1974), adapted by Carregosa et al. (2014). These enzymes catalyze the conjugation of reduced glutathione with the substrate 1‐chloro‐2,4‐dinitrobenzene (CDNB), resulting in the formation of a thioester bond that can be detected spectrophotometrically at 340 nm for 5 min between 15 s. Thioether formation was quantified using an extinction coefficient (ε) of 9.6 mM−1cm−1. The results were expressed as nmol per min per g FW.
2.5.3 Cellular damage
When antioxidant and biotransformation systems fail to neutralize ROS, oxidative damage accumulates in cells. Excess ROS triggers lipid peroxidation (evaluated by LPO), compromising membrane stability and permeability, and oxidize proteins (assessed by PC), generating carbonyls, indicators of oxidative stress (Regoli and Giuliani, 2014; Freitas et al., 2016). Evaluating both LPO and PC provides a comprehensive overview of oxidative damage and the disturbance of cellular redox homeostasis.
Levels of LPO were determined by quantifying malondialdehyde (MDA), a major aldehyde byproduct generated during the peroxidation of polyunsaturated fatty acids. The method followed was that of Ohkawa et al. (1979), with adaptations introduced by Carregosa et al. (2014). In this assay, MDA reacts with thiobarbituric acid (TBA) under high temperature and acidic conditions to form a pink‐colored adduct known as the thiobarbituric acid reactive substances (TBARS) complex. The intensity of the colored complex was measured spectrophotometrically at 532 nm. The concentration of MDA in the sample was determined using an extinction coefficient (ε) of 156 mM−1 cm−1. Results were expressed in nmol of MDA equivalents formed per g FW.
Levels of PC were assessed following the alkaline DNPH (2,4‐dinitrophenylhydrazine) method described by Mesquita et al. (2014). In this procedure, DNPH reacts specifically with carbonyl groups introduced into protein side chains by oxidative reactions, forming stable hydrazone derivatives. After derivatization, the DNP‐protein hydrazones formed were measured spectrophotometrically at 450 nm. The levels of PC were calculated using the specific extinction coefficient (ε) of 22.308 mM−1 cm−1. Results were expressed in nmol per g FW.
2.5.4 Osmoregulation responses
Proline is a low‐molecular‐weight organic osmolyte that plays a vital role in the osmotic regulation of cells exposed to environmental stressors such as salinity fluctuations. In marine invertebrates, PROL plays a crucial role in maintaining cellular homeostasis by acting as a compatible solute, stabilizing proteins, and membranes under osmotic stress. Additionally, it functions as a scavenger of ROS, contributing to the protection of cellular structures against oxidative damage (Willett and Burton, 2002). Its accumulation is essential for maintaining cellular homeostasis during salinity stress, making it a reliable indicator of osmotic and environmental stress responses (Stączek et al., 2024).
To determine PROL levels, the method of Bates et al. (1973) was adapted for marine invertebrates. Acid ninhydrin and glacial acetic acid were added to the samples, which were then incubated at 100 °C for 45 min. Absorbance was read at 520 nm, and PROL concentration was determined using a D‐proline standard curve (0–0.075 mg/mL). Results were expressed as mg per g FW, providing insight into the organisms’ osmotic adjustment capacity under experimental conditions.
2.5.5 Neurotransmission
Acetylcholinesterase is a key enzyme in vertebrate and invertebrate nervous systems, hydrolyzing acetylcholine to terminate nerve impulses and maintain synaptic function. When AChE is inhibited, acetylcholine accumulates, causing continuous nerve stimulation, impaired muscle function and neurotoxicity. For this reason, AChE activity is widely used as a sensitive biomarker of neurotoxic effects of environmental contaminants, including organophosphates, carbamates and metals (Čolović et al., 2013).
The activity of AChE was determined following the method described by Ellman et al. (1961), with modifications according to Mennillo et al. (2017). In this assay, samples were combined with 0.1 M K₂HPO4 buffer at pH 8.0, followed by the addition of 5,5'‐dithiobis(2‐nitrobenzoic acid) (DTNB) and acetylthiocholine iodide (ATChI). The reaction kinetics were monitored spectrophotometrically at 412 nm for 5 min, with absorbance readings taken every 25 s. For quantification, an extinction coefficient (ε) of 13.6 mM⁻¹cm⁻¹ was used and the results were expressed as nmol per min per g FW.
2.6 Data analysis
2.6.1 Statistical analyses
Data obtained for Gd concentration in soft tissues and biochemical parameters were statistically analyzed using hypothesis testing with permutational multivariate analyzes of variance (PERMANOVA) in PRIMER v6, with PERMANOVA+ add‐on (Anderson et al., 2008). The Pseudo‐F test was determined using unrestricted permutations of the raw data (9999 permutations) based on the Euclidean distance matrix. The null hypotheses were rejected for p‐values < 0.05 (significant differences). When significant differences were observed in the main test, pairwise comparisons were performed. Results are presented as means with standard deviations.
Four null hypotheses were tested for each biochemical parameter separately for each species: (i) no significant differences exist between control groups at different salinities (30 and 40), indicated by lowercase letters in the figures (and p‐values given in supplementary material); (ii) no significant differences exist between Gd‐exposed groups at different salinities, indicated by uppercase letters in the figures (and p‐values given in supplementary material); (iii) no significant differences exist between non‐exposed and Gd‐exposed groups within each salinity, indicated by asterisks in the figures (and p‐values given in supplementary material); (iv) no significant differences exist between species for both non‐exposed and Gd‐exposed groups at each salinity, with p‐values given in the text. For the quantification of Gd in the tissue, a null hypothesis was tested, namely, no significant differences exist among treatments for each species, indicated by lowercase letters.
2.6.2 Multivariate analyzes
Considering the multiple variables assessed in this study, including eleven biochemical parameters, two salinities (30 and 40), and two chemical conditions (presence and absence of Gd), a Principal Coordinates Ordination (PCO) was conducted to provide an integrative overview of the biological responses under different experimental treatments. A Euclidean distance similarity matrix based on all biochemical parameters was computed, and only the biochemical variables with correlation coefficients higher than 0.70 were superimposed as vectors, thereby enabling a clearer interpretation of the biomarkers that most strongly contributed to the differentiation of the biological profiles under the different experimental conditions.
2.6.3 Integrated biomarker response (IBR)
To integrate the biochemical responses of mussels and clams to the different treatment conditions, the Integrated Biomarker Response Index version 2 (IBRv2) was applied, following the methodology described by Beliaeff and Burgeot (2002) and adapted by Sanchez et al. (2013). This approach allowed for an integrated characterization of the biochemical responses in mussels and clams under the stress treatments. The integration process was evaluated by considering the stress treatments, namely Gd exposure at salinities of 30 (Gd30) and 40 (Gd40), as well as the control at salinity 40 (CTL40), which were compared with CTL30, defined as the baseline control condition. Before the calculation of the IBRv2, a logarithmic transformation was performed to reduce data variability, using the formula Yi = log(Xi/X0), where Xi corresponds to the biomarker value for each treated group and X0 represents the mean value from the control group. Subsequently, the transformed data were standardized according to Zi = (Yi ‐ μ) / σ, where μ and σ are the mean and standard deviation of the Yi values, respectively. The biomarker deviation index was then calculated as A = Zi ‐ Z0, establishing a baseline centered on zero to represent deviations relative to the control condition. The absolute values of these deviations (|A|) were then summed across all biomarkers to compute the final IBRv2 value, providing a quantitative measure of the overall biochemical response. Higher IBRv2 values indicate more pronounced biochemical alterations in response to the treatments. To facilitate interpretation, the deviation values for each biomarker were represented graphically, allowing a clear visualization of the intensity of the biochemical responses across different treatment conditions. These values were analyzed according to the overall response pattern established by the final IBRv2 index.
3 Results
3.1 Gadolinium concentrations in bivalves’ tissues
Gadolinium concentrations in clams’ soft tissue, analyzed after exposure, are presented in Table 1. In the clams of the control group, concentrations of 0.025 ± 0.009 µg/g dry weight (DW) were observed at salinity 30 and 0.024 ± 0.006 µg/g DW at salinity 40, very close values that suggest a negligible influence of salinity on basal Gd accumulation. The clams exposed to Gd at both salinities, 30 and 40, presented values of 0.099 ± 0.017 µg/g DW and 0.065 ± 0.013 µg/g DW, respectively, which represent a significant increase in relation to the values of the control groups, reflecting a clear bioaccumulation. Furthermore, the concentration of Gd in the tissue of clams at a salinity of 30 was significantly higher than in those at a salinity of 40.
Gadolinium concentrations in the soft tissue of mussels, analyzed after exposure, are presented in Table 2. In mussels under control conditions, concentrations were 0.015 ± 0.010 µg/g DW at salinity 30 and 0.008 ± 0.002 µg/g DW at salinity 40, indicating not only a lower accumulation compared to clams, but also a possible reduction in concentration with increasing salinity. Mussels exposed to Gd showed significantly higher concentrations than the respective controls, with values of 0.042 ± 0.013 µg/g DW and 0.027 ± 0.002 µg/g DW at salinities of 30 and 40, respectively. Furthermore, no significant differences were observed between salinities for Gd‐exposed mussels.
Gadolinium (Gd) concentration (µg/g dry weight (DW)) in the dried soft tissues of clams after 14 days of non‐exposure and exposure to 10 µg/L of Gd at salinities of 30 or 40.
Gadolinium (Gd) concentration (µg/g dry weight (DW)) in the dried soft tissues of mussels after 14 days of non‐exposure and exposure to 10 µg/L of Gd at salinities of 30 or 40.
3.2 Biochemical responses
3.2.1 Metabolic capacity and energy reserves
In clams, electron transport system (ETS) activity did not vary significantly between salinities in non‐contaminated treatments. Similarly, in Gd‐exposed clams, no significant differences were observed between the tested salinities. When comparing Gd‐exposed and non‐exposed clams at each salinity, mean ETS values tended to be higher in Gd‐exposed clams, although these differences were not statistically significant (Fig. 1A, Tab. S1). In mussels, ETS activity remained similar between non‐contaminated groups exposed to both salinities. Consistently, in the Gd‐exposed groups, no significant differences were observed between salinities. Similarly, no significant differences were found between control and Gd‐exposed mussels, regardless of the salinity tested (Fig. 1A, Tab. S1). No significant differences (p > 0.05) were observed between the two species regardless of the treatment tested.
In clams, the content in carbohydrates (CARB) did not vary significantly between salinities in non‐contaminated treatments. Similarly, no significant differences were observed in Gd‐exposed clams regardless of the tested salinities. When comparing Gd‐exposed and non‐exposed clams at each salinity, CARB levels remained consistent, with no significant differences detected at either salinity (Fig. 1B, Tab. S1). In mussels, no significant differences were observed at salinity 40 compared to salinity 30 in the non‐contaminated groups. Consistently, for the Gd‐exposed groups, CARB levels remained similar between salinities. Comparisons between control and Gd‐exposed mussels showed no significant differences at either salinity (Fig. 1B, Tab. S1). No significant differences (p > 0.05) were observed between the two species, regardless of the treatment.
In clams, in the absence of Gd, the total protein (PROT) showed no significant differences between salinities. No significant differences were observed in Gd‐exposed clams between the two salinities. Comparisons between Gd‐exposed and non‐exposed clams showed no significant changes at either salinity (Fig. 1C, Tab. S1). In mussels, the PROT content remained stable across salinities in both control and Gd‐exposed groups. Likewise, no significant differences were detected between Gd‐exposed and non‐exposed mussels at either salinity (Fig. 1C, Tab. S1). No significant differences (p > 0.05) were observed between the two species, regardless of the treatment.
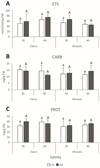 |
Fig. 1 A: Electron transport system (ETS) activity; B: Carbohydrates (CARB) content; C: Protein (PROT) content, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are presented as means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
3.2.2 Antioxidant and biotransformation capacity
In clams, superoxide dismutase (SOD) activity showed no significant differences between salinities in the non‐contaminated groups. No significant differences were observed in Gd‐exposed clams between salinities. When comparing Gd‐exposed and non‐exposed clams at each salinity, SOD levels remained similar, with no statistically significant changes detected (Fig. 2A, Tab. S1). In mussels, SOD activity was significantly lower at salinity 40 compared to salinity 30 in the non‐contaminated groups. In Gd‐exposed mussels, no significant differences were observed between salinities. When comparing control and Gd‐exposed mussels at salinity 30, a significant decrease in SOD activity was observed following Gd exposure, whereas no significant differences were detected at salinity 40 (Fig. 2A, Tab. S1). Differences between the two species were observed in Gd‐exposed groups at both salinities (p = 0.0496 for salinity 30; p = 0.0053 for salinity 40), with higher values in clams.
In clams, catalase (CAT) activity remained similar between salinities for non‐contaminated, while it increased significantly in Gd‐exposed clams at salinity 40 compared with salinity 30. When comparing Gd‐exposed and non‐exposed clams at each salinity, no significant differences were noticed regardless of the salinity (Fig. 2B, Tab. S1). In mussels, CAT activity was significantly lower at salinity 40 than at salinity 30 in the non‐contaminated groups. Similarly, for Gd‐exposed groups, a significant decrease in CAT activity was observed at salinity 40. Comparisons between control and Gd‐exposed mussels showed no significant differences regardless of the salinity tested (Fig. 2B, Tab. S1). Differences between the two species were observed at both salinities in non‐contaminated (p = 0.009 for salinity 30; p = 0.0038 for salinity 40) and Gd‐exposed (p = 0.0159 for salinity 30; p = 0.0001 for salinity 40) groups, with higher values in clams.
In clams, carboxylesterases (CbEs) activity did not vary significantly between salinities in non‐contaminated and Gd‐exposed clams. Comparisons between Gd‐exposed and non‐exposed clams showed no significant differences at either salinity (Fig. 2C, Tab. S1). In mussels, CbEs activity was significantly higher at salinity 40 compared to salinity 30 in the non‐contaminated groups. For the Gd‐exposed groups, a significant difference was also observed, with higher activity at salinity 30. Comparisons between control and Gd‐exposed mussels revealed a significant increase in CbEs activity at salinity 30 in the Gd‐exposed group (Fig. 2C, Tab. S1). Differences between the two species were observed at both salinities in non‐contaminated (p = 0.0001 for salinity 30; p = 0.0007 for salinity 40) and Gd‐exposed (p = 0.0001 for salinity 30; p = 0.0001 for salinity 40) groups, with higher values in mussels.
In clams, glutathione S‐transferases (GSTs) activity did not differ significantly between salinities in the non‐contaminated groups. In contrast, a significant difference was observed between salinities in Gd‐exposed clams, with lower values at salinity 40. At salinity 30, when comparing Gd‐exposed and non‐exposed clams, a significant increase in GSTs activity was found in Gd‐exposed clams, whereas no significant differences were observed between non‐exposed and Gd‐exposed clams at salinity 40 (Fig. 2D, Tab. S1). In mussels, a significant difference in GSTs activity was observed between salinities in the non‐contaminated groups, with lower values at salinity 40. Similarly, a significant difference was observed between salinities in Gd‐exposed mussels, with lower values at salinity 40. When comparing control and Gd‐exposed mussels within each salinity, no significant differences were observed at salinity 30, while a significant decrease in GSTs activity was found in Gd‐exposed mussels at salinity 40 (Fig. 2D, Tab. S1). Differences between the two species were observed at both salinities in non‐contaminated (p = 0.0001 for salinity 30; p = 0.0014 for salinity 40) and Gd‐exposed (p = 0.0001 for salinity 30; p = 0.0004 for salinity 40) groups, with higher values in clams.
 |
Fig. 2 A: Superoxide dismutase (SOD) activity; B: Catalase (CAT) activity; C: Carboxylesterases (CbEs) activity with pNPB substrate; D: Glutathione S‐transferases (GSTs) activity, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are presented as means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
3.2.3 Osmoregulatory responses
In clams, proline (PROL) content did not differ significantly between salinities 30 and 40 in the non‐contaminated and contaminated groups. Comparisons between Gd‐exposed and non‐exposed clams showed no significant differences at both salinities (Fig. 3A, Tab. S1). In mussels, PROL content did not differ significantly between salinities 30 and 40 in the non‐contaminated groups, and similarly, no significant differences were observed in the Gd‐exposed groups. Comparisons between control and Gd‐exposed mussels revealed no significant differences at either salinity (Fig. 3A, Tab. S1). No significant differences were observed between species regardless of the treatment (p>0.05).
 |
Fig. 3 A: Proline (PROL) content; B: Lipid peroxidation (LPO) levels; C: Protein carbonylation (PC) levels, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
3.2.4 Cellular damage
In clams, lipid peroxidation (LPO) levels did not differ significantly in non‐contaminated groups between salinities 30 and 40. Similarly, no significant differences were observed for Gd‐exposed groups between the two salinities. When comparing Gd‐exposed and non‐exposed clams, no significant differences were detected at salinity 30, nor at salinity 40 (Fig. 3B, Tab. S1). In mussels, LPO levels did not differ significantly between salinities for non‐contaminated groups. Also, for Gd‐exposed groups, no significant differences were observed between salinities. Comparisons between control and Gd‐exposed mussels showed no significant differences at either salinity (Fig. 3B, Tab. S1). Differences between the two species were observed in non‐contaminated groups at both salinities (p = 0.0122 for salinity 30; p = 0.0085 for salinity 40) and Gd‐exposed groups at salinity 30 (p = 00086), with higher values in mussels.
In clams, no significant differences in protein carbonylation (PC) levels were observed between salinities in either the non‐contaminated or the Gd‐exposed groups. When comparing Gd‐exposed and non‐exposed clams within each salinity, no significant differences were detected (Fig. 3C, Tab. S1). In mussels, PC levels did not differ significantly between salinities 30 and 40 in non‐contaminated mussels, and similarly, no significant differences were observed for Gd‐exposed mussels between salinities. Comparisons between control and Gd‐exposed mussels within each salinity showed no significant differences (Fig. 3C, Tab. S1). Differences between the two species were only observed at salinity 40 in both non‐contaminated (p = 0.0156) and Gd‐exposed (p = 0.0361) groups, with higher values in mussels.
3.2.5 Neurotransmission
In clams, acetylcholinesterase (AChE) activity did not vary significantly between salinities in non‐contaminated treatments. Similarly, no significant differences were observed in Gd‐exposed clams regardless of the tested salinities. Comparisons between Gd‐exposed and non‐exposed clams showed no significant differences at either salinity (Fig. 4, Tab. S1). In mussels, AChE activity did not differ significantly between salinities 30 and 40 in both non‐contaminated and Gd‐exposed groups. Comparisons between control and Gd‐exposed mussels within each salinity revealed no significant differences at either salinity (Fig. 4, Tab. S1). No significant differences (p>0.05) were observed between the two species regardless of the treatment.
 |
Fig. 4 Acetylcholinesterase (AChE) activity, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
 |
Fig. 5 Centroid ordination diagram of Principal Coordinates Analysis (PCO) based on tested treatments (control, CTL; Gadolinium, Gd; salinities 30 or 40) and biochemical parameters measured in Ruditapes philippinarum (C) and Mytilus galloprovincialis (M). Biochemical parameters are represented with name abbreviations. Correlation vectors (r > 0.70) are superimposed as supplementary variables. |
3.3 Principal coordinates analysis (PCO)
The PCO analysis (Fig. 5) revealed that the first principal coordinate axis (PCO1) accounted for 55% of the total variance, while the second coordinate (PCO2) explained 17.5%, together representing 72.5% of the overall variation in the dataset. Based on the ordination of the PCO analysis, a clear separation between species is observed along the PCO1 axis. All treatments of R. philippinarum (CCTL Sal 30, CCTL Sal 40, CGd Sal 30, CGd Sal 40) are positioned on the positive side of this axis, while all treatments of M. galloprovincialis (MCTL Sal 30, MCTL Sal 40, MGd Sal 30, MGd Sal 40) appear on the negative side, reflecting a clear distinction between the biochemical profiles of the two species. The distribution of the biochemical vectors follows this separation, with CARB, PROT and GSTs projecting on the positive side of PCO1 and associating with R. philippinarum treatments, while CbEs, PC, AChE, and PROL project on the negative side, in proximity to M. galloprovincialis. LPO stands out negatively on PCO2, associated with MCTL Sal 30, while SOD and CAT project negatively along the same axis and are exclusively related to CGd Sal 40.
In the treatments with R. philippinarum, CCTL Sal 30, CCTL Sal 40 and CGd Sal 30 form a compact cluster in the upper right quadrant, close to the CARB, PROT, and GSTs vectors. This proximity suggests a relevant contribution of energy reserves and detoxification enzymes under these treatments, with CGd Sal 30 being close to GSTs, indicating a higher activation of these enzymes in the presence of Gd at salinity 30. CCTL Sal 30 is located at the center‐right of the plot, CCTL Sal 40 slightly above, and CGd Sal 30 between the two all with little dispersion along the PCO2 axis, reinforcing the biochemical similarity among these groups. In contrast, CGd Sal 40 appears separated from the other treatments of the species, positioned in the lower right quadrant, associated with the SOD and CAT vectors, indicating a clear activation of antioxidant defenses. CGd Sal 40 is also the only R. philippinarum treatment with a notable projection along the PCO2 axis, highlighting the role of oxidative stress under combined Gd exposure and high salinity.
In the M. galloprovincialis treatments, MCTL Sal 30 appears close to the LPO vector, suggesting a greater contribution of this biomarker under the control condition at salinity 30. MCTL Sal 40, was slightly above the horizontal axis and farther from the biochemical vectors, suggesting a high influence of the most explanatory biomarkers (PC, CbEs, AChE). Gd‐exposed treatments showed clearer association with specific biomarkers. MGd Sal 30 is located centrally on the negative side of PCO1, near the CbEs, PC, and AChE vectors, indicating a strong contribution of biotransformation and neurotoxicity processes. MGd Sal 40 occupies the upper left quadrant and shows a slight additional proximity to the PROL vector, which may indicate a complementary response linked to osmotic regulation.
The analysis thus demonstrates a clear differentiation in biochemical responses between species, with R. philippinarum showing more clustered patterns dominated by energy metabolism and detoxification, except for CGd Sal 40, where antioxidant activation stands out. Meanwhile, M. galloprovincialis exhibits greater dispersion and association with markers of oxidative damage, osmorregulation, neurotoxicity, and biotransformation, particularly under Gd exposure.
3.4 Integrated biomarker response (IBR)
Table 3 summarizes the IBR values obtained, highlighting the differences in biochemical responses between species and salinity conditions tested. For R. philippinarum, IBRv2 values were similar across treatments, with the highest scores observed under Gd exposure at both salinities (9.0), compared to 8.4 at salinity 40. In M. galloprovincialis, higher IBRv2 values were recorded at salinity 40, both with (13.3) and without (12.7) Gd, compared to Gd exposure at salinity 30 (11.5) (Tab. 3).
Integrated Biomarker Response index version 2 (IBRv2) in Ruditapes philippinarum and Mytilus galloprovincialis after 14 days of exposure to 0 μg/L (CTL) and 10 μg/L of gadolinium (Gd) at different salinity levels (30 and 40).
4 Discussion
The present study aimed to assess the impacts induced by Gd on Ruditapes philippinarum and Mytilus galloprovincialis. Also, the influence of salinity was investigated on the effects caused by Gd.
4.1 Gadolinium concentrations in bivalves’ soft tissues
4.1.1 Concentration in clams
Upon exposure to 10 µg/L of Gd, clams exhibited a significant increase in tissue concentrations, indicating an uptake of Gd under the tested treatments. Furthermore, the Gd concentration was significantly higher in contaminated clams at salinity of 30 than the contaminated ones at salinity of 40. Although both values represent substantial enrichments compared to their respective controls, the difference between salinities suggests that environmental salinity may influence Gd bioavailability or internal retention dynamics. As reported by Rainbow (2002), similar variability in trace metal accumulation patterns was observed across aquatic invertebrate taxa, with bioaccumulation reflecting not only environmental availability but also species‐specific physiological handling and detoxification capacity.
The substantial fold increase between CTL and Gd‐exposed groups, approximately 4‐fold at salinity 30 and 2.7‐fold at salinity 40, indicates an efficient accumulation pathway in R. philippinarum, potentially associated with its filtration rate, infaunal behavior, and continuous interaction with both dissolved and particulate fractions of the contaminant. As reported by Yang et al. (2013), similar patterns of trace element accumulation were observed in R. philippinarum collected from coastal mariculture zones in China, where its filter‐feeding behavior and sedimentary exposure facilitated significant uptake of metals from both water and food sources. These results align with previous studies demonstrating that bivalves, particularly clams, are highly susceptible to accumulating rare earth elements (REEs) in their soft tissues over short‐ to medium‐term exposure periods. The underlying mechanisms may involve passive diffusion across gill epithelia, endocytosis of metal complexes, or adsorption onto mucus‐secreting surfaces followed by internalization (Lachaux et al., 2023).
The influence of salinity on Gd bioaccumulation remains complex. Lower salinities are often linked to enhanced metal uptake, which may result from reduced competition with major cations (e.g., Ca2+, Mg2+), increased permeability of epithelial barriers, or greater speciation of metals into more bioavailable forms. Conversely, higher salinities may promote the complexation of Gd with chloride or sulfate ions, altering its speciation and potentially diminishing its bioavailability (IPCC, 2019). In the present study, the significantly higher Gd accumulation observed at salinity 30 supports the former hypothesis, indicating that osmotic conditions at this salinity favor Gd uptake, possibly through increased epithelial permeability or enhanced membrane transport. Nevertheless, the continued substantial accumulation at salinity 40 highlights that Gd remains bioavailable and biologically active even under marine salinity conditions, which is ecologically significant given the ongoing salinization of estuarine environments driven by climate‐induced drought scenarios.
From a physiological standpoint, the accumulation of Gd in soft tissues may disrupt key ionic balances, particularly due to Gd's chemical mimicry of calcium (Krasznai et al., 2003; Trapasso et al., 2021). Such interference could impair signaling pathways, enzyme activities, and the stability of membrane potentials in target organs such as the gills, digestive gland, and nervous system. The elevated tissue concentrations observed in the present study are consistent with subsequent biochemical alterations, including the slight increase of electron transport system (ETS) activity and oxidative stress, as discussed in later sections. Therefore, Gd accumulation in R. philippinarum represents not only a biomarker of environmental contamination but also a precursor to functional impairment, reinforcing its suitability as a sentinel species for future biomonitoring of REEs in transitional water systems. As highlighted by Sturla Lompré et al. (2025), this species exhibits marked cellular and physiological responses when exposed to environmental stressors, including oxidative damage and metabolic disruption, supporting its relevance for ecotoxicological risk assessment under multi‐stressor conditions.
4.1.2 Concentration in mussels
Upon exposure to 10 µg/L of Gd for 14 days, mussels showed a clear accumulation of Gd in soft tissues, with concentrations significantly increasing compared to CTL. Although these values remain lower than those recorded for clams under equivalent conditions, the fold‐change relative to control levels is still substantial, with approximately a 2.8‐fold increase at salinity 30 and a 3.4‐fold increase at salinity 40. This finding emphasizes that M. galloprovincialis retains a non‐negligible capacity for bioaccumulation under contaminant‐rich conditions, highlighting that even species with reduced baseline uptake can experience physiological burdens when subjected to sustained exposure. As observed by Spampinato et al. (2024), mussels exposed to prolonged metal contamination exhibited tissue accumulation and sublethal effects, supporting the notion that chronic exposure can overwhelm baseline regulatory barriers, leading to physiological stress even in species considered less permeable.
Although subtle, the salinity effect suggests that ionic strength does influence Gd bioaccumulation in M. galloprovincialis, likely through mechanisms such as modifications in metal speciation, competition with major cations, or changes in epithelial transport processes. As reported by Spampinato et al. (2024), REEs uptake in mussels can be modulated by environmental parameters such as salinity and ionic composition, which affect the speciation and bioavailability of dissolved metals, ultimately influencing their accumulation profiles. It is plausible that at lower salinities, reduced concentrations of competing cations (e.g., Ca2+, Na+) facilitate the passive diffusion or channel‐mediated uptake of Gd3+, whereas higher salinities may promote Gd precipitation or complexation with anions such as SO42⁻ or Cl⁻, thereby reducing its free ionic activity and subsequent bioavailability.
Despite the modest Gd concentrations, the detection of accumulation within 14 days indicates a biologically relevant exposure. Importantly, the lower Gd burden in mussels does not imply the absence of toxicological effects. Several biochemical markers were modulated by Gd in this species, demonstrating that even moderate internal contamination levels can trigger stress responses. This suggests that M. galloprovincialis may have a lower threshold for functional impairment induced by Gd or that tissue‐specific partitioning leads to the concentration of the metal in sensitive organs such as the gills or digestive gland, even when whole‐body burdens remain relatively low. Alternatively, the species may employ a more conservative metabolic and detoxification strategy, activating defense pathways early in the exposure process to prevent significant metal accumulation. This hypothesis is consistent with the findings of Spampinato et al. (2024), who reported that REEs exposure in M. galloprovincialis induced oxidative stress and enzymatic changes even in the absence of substantial whole‐body accumulation, suggesting that sublethal effects can emerge from localized or early‐stage bioaccumulation processes.
4.2 Biochemical responses
The Principal Coordinates Ordination (PCO) clearly separates the biochemical responses of R. philippinarum and M. galloprovincialis along the first axis. In R. philippinarum, control and Gd‐exposed groups at salinity 30 (CCTL Sal 30, CGd Sal 30) cluster near carbohidrates (CARB), protein (PROT), and glutathione S‐transferases (GSTs) vectors, indicating a dominance of energy reserves and detoxification processes. At salinity 40, CGd Sal 40 aligns with superoxide dismutase (SOD) and catalase (CAT) vectors along PCO2, evidencing changes in antioxidant defense mechanisms. Despite the different strategy that suggests that under hypersaline Gd exposure clams prioritize oxidative stress management, the IBR value determined in CGd Sal 40 is similar to the IBR observed in CGd Sal 30. As demonstrated by Andrade et al. (2022, 2023), M. galloprovincialis displayed marked activation of antioxidant enzymes under Gd exposure, particularly when combined with environmental stressors, underscoring its ability to upregulate defensive metabolism once physiological thresholds are surpassed and indicating the onset of an oxidative stress response and the activation of protective pathways against Gd‐induced ROS.
For M. galloprovincialis, Gd exposure at salinity 30 (MGd Sal 30) shifts mussels toward carboxylesterases (CbEs), protein carbonylation (PC), and acetylcholinesterase (AChE) vectors, highlighting biotransformation and neurotoxicity pathways. At salinity 40, MGd Sal 40 associates with proline (PROL) and maintains proximity to CbEs and AChE, linked to osmotic regulation and moderate detoxification, showing the highest IBR value. These patterns align with the findings of Andrade et al. (2023), who reported that Gd exposure in M. galloprovincialis leads to significant alterations in detoxification enzymes (CbEs), osmorregulation capacity, and AChE, with modulation dependent on salinity and indicative of cumulative sublethal stress.
Thus, clams primarily responded through enhanced energy mobilization and antioxidant defenses, whereas mussels exhibited early activation of phase I biotransformation, osmorregulation and neuroprotective pathways.
4.2.1 Metabolic capacity and energy reserves
The transition from a moderate salinity of 30 to a hypersaline regime of 40 in R. philippinarum showed no significant variations on ETS activity although a slight increase was observed at the highest salinity. As noted by Coughlan et al. (2009), this species exhibits partial osmotic regulation, which may contribute to its resilience under salinity fluctuations without altering key metabolic pathways. The absence of significant ETS activation in response to Gd exposure indicates that mitochondrial electron flux remains steady and that clams do not increase their potential aerobic capacity under these conditions. Given that ETS activity is a proxy for potential aerobic metabolism, these results suggest that clams rely on existing respiratory capacity and basal detoxification mechanisms to cope with the combined osmotic and toxic challenges without imposing further energetic strain. This interpretation aligns with Mossotto et al. (2025), who reported that Gd exposure did not significantly disturb primary physiological processes in aquatic organisms, suggesting a potentially low impact of Gd on basal metabolic rates in these species.
For CARB, R. philippinarum maintained stable reserves across salinities and treatments, with no significant depletion or accumulation detected. This observation is in line with the work of Coughlan et al. (2009), who described how R. philippinarum may rely on organic osmolytes to maintain cellular homeostasis during salinity fluctuations, preserving energy stores such as CARB. This resilience suggests that the clam preserves its principal carbohydrate store, meeting increased energetic demands via modulation of other metabolic pathways or efficient external nutrient assimilation, rather than by depleting CARB.
Regarding PROT content, R. philippinarum exhibited a slight increase at salinity 40 compared to 30 under non‐contaminated conditions, suggesting a reallocation of nitrogen resources. Since proteins serve as major reservoirs of nitrogen within the organism, this slight increase may reflect an adaptive adjustment to enhance the synthesis of nitrogen‐containing osmolytes, such as free amino acids or compatible solutes, thereby counterbalancing the osmotic stress induced by hypersaline conditions (Buer et al., 2020). As observed by Munari et al. (2011), clams exposed to varying salinity levels maintain stable haemolymph protein content, reinforcing the idea that protein adjustments are not necessarily indicators of cellular stress, but rather strategic physiological responses to environmental salinity fluctuations. However, no significant differences were found in PROT levels between salinities in Gd‐exposed clams, nor between Gd‐exposed and control clams within each salinity, indicating that exposure to Gd at 10 µg/L does not significantly affect PROT dynamics under hypersaline conditions. This stability under Gd exposure is consistent with the findings of Mossotto et al. (2025), who noted that primary consumers like bivalves exhibit a higher tolerance to Gd, with limited disruption to biochemical parameters such as protein metabolism. Such results suggest that clams can maintain protein homeostasis in the presence of Gd, possibly through efficient regulation of nitrogen distribution between basal metabolic demands and defense‐related pathways, without requiring increased protein synthesis.
In M. galloprovincialis, ETS activity remained stable across all salinities and treatments, indicating that the mussels also maintain a conservative respiratory output regardless of osmotic or toxic stress. This consistent pattern, as indicated by Leite et al. (2024), suggests that mussels exhibit a more rigid metabolic strategy, avoiding the energetic costs associated with increased ETS, thereby minimizing oxidative damage and preserving internal energy reserves.
CARB content in M. galloprovincialis also remained unchanged across all salinities and treatments. As demonstrated by Leite et al. (2024), these energy reserves were not significantly mobilized despite exposure to osmotic stress and rare earth elements, suggesting a tight regulation of carbohydrate metabolism. This tight regulation implies that mussels adopt a highly conservative energy management strategy, prioritizing the preservation of internal carbohydrate pools even under environmental stress.
Similarly, PROT content in mussels showed no significant variation across treatments and salinities, reinforcing the notion of a highly conservative metabolic strategy. Leite et al. (2024) similarly observed that PROT content in mussels remained stable under salinity shifts and exposure to rare earth elements, highlighting the prioritization of structural protein conservation for essential physiological functions such as growth and reproduction. The lack of proteomic modulation under both osmotic and Gd stress implies a fixed nitrogen economy in mussels, where structural integrity is preserved at the expense of metabolic plasticity. This conservative strategy allows mussels to avoid unnecessary nitrogen expenditure, even in potentially adverse conditions, contributing to their resilience in fluctuating environments.
4.2.2 Antioxidant and biotransformation capacity
In the absence of Gd, R. philippinarum shows a slightly higher SOD activity at salinity 30 than at salinity 40, which suggests increased oxidative pressure when ion transport is most active. As noted by Munari et al. (2011), temperature and salinity shift significantly influence haemocyte immune responses in R. philippinarum, indicating that physiological stress under altered salinity conditions can modulate antioxidant defenses. In Gd‐exposed clams, SOD activity remains stable between salinities, and no significant differences were detected when comparing exposed and non‐exposed groups at each salinity, although a non‐significant decrease was observed at salinity 30 and an opposite pattern was observed at salinity 40. Thus, Gd exposure does not enhance SOD activity in clams under the tested conditions. These results imply that while baseline SOD activity slightly declines with increasing salinity, Gd exposure does not trigger significant enzymatic upregulation, suggesting that oxidative demand does not exceed the constitutive defense capacity of clams.
Catalase in non-contaminated R. philippinarum although not statistically significant, tended to increase at salinity 40, hinting that hydrogen peroxide removal might be driven by osmotic pressure alone. In Gd‐exposed clams, CAT activity increased significantly at salinity 40 compared to 30 and is also higher than in the non‐exposed group at this salinity. This pattern indicates that combined with salinity 40 and Gd‐exposure elevate peroxidative stress sufficiently to stimulate CAT activity, reinforcing a protective antioxidant response when environmental challenges intensify. As reported by Sturla Lompré et al. (2025), exposure to combined stressors, including contaminants and temperature, can lead to significant modulation of antioxidant enzymes like CAT in R. philippinarum, highlighting their responsiveness under multiple environmental pressures.
R. philippinarum displays stable CbEs activity across salinities in both control and Gd‐exposed groups, with no significant differences observed between treatments. As observed in Zhou et al. (2023), although oxidative stress responses are commonly activated under salinity stress in bivalves, certain detoxification enzymes, such as CbEs, can remain stable, suggesting selective modulation of enzymatic pathways depending on the nature and intensity of the stressor. This stability suggests that CbEs are not modulated by either osmotic stress or Gd exposure under the tested conditions, implying that baseline hydrolytic detoxification capacity suffices and that clams avoid unnecessary metabolic expenditure when the stressor burden does not exceed homeostatic thresholds.
Glutathione S‐transferases activity in R. philippinarum remains unchanged between the two salinities in control. In Gd‐exposed clams, GSTs activity was higher at salinity 30 compared to 40. A significant increase in GSTs activity is also observed at salinity 30 between exposed and non‐exposed clams, whereas at salinity 40 no significant differences are detected. This observation aligns with Velez et al. (2017), who demonstrated that GSTs activity in clams is sensitive to environmental stressors and can be modulated under favorable conditions (such as moderate salinity) to enhance detoxification processes. These patterns suggest that Gd-exposed clams enhance GSTs‐mediated conjugative detoxification under moderate salinity when oxidative and metabolic conditions are more favorable, while the reduced activity at salinity 40 may reflect a limitation in substrate availability or energetic cost control.
In M. galloprovincialis, SOD activity is significantly higher at salinity 30 than at 40 in non‐contaminated groups. As observed in Figueiredo et al. (2022), salinity shifts can impose oxidative stress, influencing antioxidant enzyme activities such as SOD, especially under moderate salinity levels. In Gd‐exposed mussels, no significant differences are observed between salinities, and Gd exposure significantly reduces SOD activity at salinity 30 compared to the control, while no significant effects are seen at salinity 40. These findings suggest that Gd suppresses antioxidant defenses at moderate salinity, but the already reduced basal SOD activity at salinity 40 remains unaffected. The species therefore maintains an energy‐conservative strategy, limiting antioxidant investment as the metabolic rate declines.
Catalase in M. galloprovincialis is significantly higher at salinity 30 than at 40 under control conditions. Similar trends were reported by Freitas et al. (2019), where CAT activity was modulated by salinity in marine mussels, indicating the enzyme's sensitivity to osmotic stress. In Gd‐exposed mussels, CAT activity is slightly lower compared to controls at both salinities, with a decline at salinity 40. This is consistent with Andrade et al. (2022), who demonstrated that exposure to lanthanides can severely inhibit CAT activity, especially under combined pollutant and temperature stress. This suppression reflects a strategy that limits energy investment in peroxide detoxification, instead relying on broader reductions in oxygen consumption to curb oxidative stress. Such behavior fits the profile of an energy‐conserver organism that prioritizes resource preservation under adverse conditions.
Carboxylesterases activity in M. galloprovincialis is significantly higher at salinity 40 than at 30 in non‐contaminated groups. As described in Freitas et al. (2019), CbEs activity increases under salinity stress, likely as a compensatory response to maintain homeostasis in detoxification pathways. For Gd‐exposed mussels, CbEs activity is higher at salinity 30 while no significant differences are observed at salinity 40 between exposed and non‐exposed groups. This outcome supports the findings by Freitas et al. (2019), where biotransformation enzymes exhibited higher inducibility under moderate salinity when combined with metal exposure. This suggests that CbEs induction occurs under milder salinity stress combined with Gd exposure, but is curtailed under hypersaline, low‐metabolism conditions, fitting an energy‐conserver strategy that balances detoxification needs with metabolic constraints.
In M. galloprovincialis, GSTs activity is significantly higher at salinity 30 than at 40 in non‐contaminated groups. This agrees with the observations by Andrade et al. (2022), where GSTs activity was found to decline under high salinity and warming scenarios, indicative of metabolic depression. For Gd‐exposed mussels, GSTs activity also differs significantly between salinities, and a significant reduction is observed at salinity 40 compared to the control. Similar results were noted by Andrade et al. (2022), who reported reduced GSTs activity under combined exposure to REEs and environmental stressors, suggesting an energy‐saving response. These data indicate that conjugative detoxification is favored at moderate salinity but is downregulated under hypersaline stress and Gd exposure, reinforcing the conservation of resources during metabolic depression.
Overall, R. philippinarum exhibits a more selective and condition‐dependent activation of antioxidant and detoxification enzymes, enhancing CAT and GSTs activities only when oxidative and osmotic challenges surpass baseline thresholds, whereas M. galloprovincialis follows a conservative strategy by broadly downregulating antioxidant defenses and limiting biotransformation responses under combined Gd and salinity stress, thereby prioritizing energy conservation over enzymatic adaptation.
4.2.3 Osmoregulatory responses
R. philippinarum shows no significant variation in PROL content between salinities 30 and 40 under control conditions, although higher values were observed at salinity 40. Li et al. (2024) explained the role of PROL as a major osmolyte, with its accumulation being more prominent only under hypersalinity conditions, where the glutamate pathway actively enhances its synthesis to counterbalance osmotic stress. Coughlan et al. (2009) affirmed that R. philippinarum possesses partial osmoregulatory capabilities that allow it to maintain cellular homeostasis under varying salinity conditions, contributing to its physiological resilience. In the presence of Gd, PROL also tended to increase at salinity 40 but less than under control conditions, indicating that Gd exposure affects the osmotic adjustment capacity of clams. This is consistent with the findings of Li et al. (2024), who demonstrated that environmental stressors, such as increased salinity, promote significant changes in the expression and activity of key enzymes, modulating PROL metabolism to maintain osmotic balance under challenging conditions. According to these authors, under hyperosmotic environments, the organism's ability to regulate PROL synthesis becomes more robust, with upregulation of the glutamate pathway enzymes contributing to maintaining homeostasis, even when external stressors are present. These results suggest that Gd alters the regulation of PROL, possibly by interfering with its synthesis or accumulation pathways. Indeed, as discussed by Li et al. (2024), PROL homeostasis under salinity stress is maintained through complex regulation mechanisms involving transcriptional control and feedback inhibition of key enzymes, allowing organisms to fine‐tune osmolyte levels depending on environmental stress intensity. Despite these modifications, proline homeostasis appears to be maintained at salinity 40, likely supported by the clam's broader set of osmolytes and antioxidant defenses.
In M. galloprovincialis, PROL tended to increase at salinity 40 in both non‐contaminated and Gd‐exposed groups. No significant differences were observed between Gd‐exposed and non‐exposed mussels at either salinity, indicating that PROL regulation in mussels is not markedly influenced by Gd exposure. This observation aligns with findings by Arrigo et al. (2024), where M. galloprovincialis maintained proline levels even under combined stress of temperature increase and contaminant exposure, supporting the hypothesis that the species employs stable osmolyte regulation as a protective strategy. This constancy highlights a conservative osmotic adjustment strategy, where mussels maintain osmolyte balance without major resource shifts, complementing their broader energy‐saving response under combined osmotic and metal stress.
Thus, while R. philippinarum displays a flexible osmotic adjustment capacity with PROL levels modulated by salinity and Gd exposure, reflecting an inducible osmoregulatory strategy, M. galloprovincialis maintains stable PROL content in the presence of Gd regardless of the salinity, indicating a more conservative osmoregulatory mechanism that ensures osmotic balance without the need for significant metabolic adjustments.
4.2.4 Cellular damage
R. philippinarum exhibits stable LPO levels at salinities of 30 and 40 under control conditions, with no significant differences between salinities. As is possible to observe in Coughlan et al. (2009), this species shows partial osmotic regulation, maintaining cellular stability under salinity fluctuations, which may contribute to the maintenance of membrane integrity. Similarly, although higher at salinity 40, no significant differences were detected in the presence of Gd, and comparisons between Gd‐exposed and non‐exposed clams at each salinity show no changes. This is consistent with findings by Sturla Lompré et al. (2025), where clams maintained oxidative stability even under combined environmental stressors, highlighting the robustness of their antioxidant defenses. These results suggest that membrane lipid stability in clams is maintained under both osmotic and metal stress conditions, possibly due to efficient antioxidant defenses that prevent the accumulation of oxidative damage in polyunsaturated fatty acids.
Protein carbonylation (PC) in R. philippinarum also remains unchanged between salinities in both non‐contaminated and Gd‐exposed groups, and no significant differences are observed between Gd‐exposed and non‐exposed clams at either salinity. As pointed out by Sturla Lompré et al. (2025), stable protein oxidative markers under multiple stressors suggest that clams possess an effective oxidative balance system capable of mitigating PC even in the presence of contaminants. This constancy indicates that protein oxidation is not significantly impacted by salinity 40 or Gd exposure, suggesting that the oxidative balance and protective mechanisms in clams are sufficient to prevent the formation of carbonyl derivatives under the tested conditions.
In M. galloprovincialis, LPO levels are similar across salinities under both non‐contaminated and Gd‐exposed conditions, with no significant differences detected between exposed and unexposed mussels at either salinity. As it was possible to observe in Queirós et al. (2024), mussels exposed to osmotic stress conditions, such as salinity shifts, maintain stable levels of oxidative biomarkers like LPO, reflecting their high metabolic adaptability to environmental changes. This stability suggests that mussels regulate oxidative metabolism effectively, maintaining membrane integrity under varying salinity and Gd exposure levels. However, species comparisons indicate that mussels present higher LPO levels than clams at both salinities under non‐contaminated conditions and at salinity 30 under Gd exposure, pointing to a baseline difference in oxidative status or susceptibility.
Similarly, PC levels in M. galloprovincialis do not vary significantly between salinities or between Gd‐exposed and non‐exposed mussels, although tended to increase at salinity 40 in non-contaminated ones. This finding is consistent with observations by Queirós et al. (2024), where no significant alterations in PC were reported across different salinity regimes, even under pharmaceutical exposure, suggesting that mussels employ inherent antioxidant and protective mechanisms to stabilize protein structures. These findings imply that mussels maintain protein stability despite osmotic and metal stress, possibly relying on inherent antioxidant defenses and reduced metabolic activity to limit oxidative damage. Moreover, as reported by Queirós et al. (2024), the maintenance of protein integrity under stress conditions supports the hypothesis of an energy‐conserver strategy adopted by M. galloprovincialis, where biochemical stability is favored to minimize the costs associated with environmental fluctuations. The absence of significant modulation further supports the interpretation of an energy‐conserver strategy, where minimal biochemical investment accompanies reduced oxidative stress.
Overall, while R. philippinarum exhibits a robust oxidative balance with minimal lipid and protein damage even under Gd exposure, indicating effective stress mitigation mechanisms. This observation is further supported by Coughlan et al. (2009), who demonstrated that R. philippinarum can sustain key physiological processes, even under osmotic stress, without exhibiting significant cellular damage. M. galloprovincialis maintains cellular stability through an energy‐conserver strategy but shows inherently higher baseline oxidative markers, suggesting a trade‐off between oxidative resilience and metabolic economy.
4.2.5 Neurotransmission
R. philippinarum shows a stable AChE activity under both salinities 30 and 40 in non‐contaminated treatments, with no significant differences observed. Similarly, the addition of 10 µg/L of Gd does not alter AChE activity across salinities or between Gd‐exposed and non‐exposed groups. These results indicate that cholinergic neurotransmission in clams remains unaffected by moderate salinity variations or Gd exposure, suggesting that AChE activity is resilient under these environmental conditions. Such stability aligns with findings from Coughlan et al. (2009), where the Manila clam's capacity for partial osmoregulation was linked to its overall physiological robustness in fluctuating salinity environments. This stability further implies that neuromuscular function is preserved without requiring additional energetic investment, allowing clams to prioritize energy allocation toward antioxidant defenses and osmotic regulation under combined stressors.
In M. galloprovincialis, AChE activity also remains constant between salinities in non‐contaminated groups, and no significant differences were observed under Gd exposure. Comparisons between Gd‐exposed and non‐exposed mussels showed no significant changes at either salinity, indicating that mussels, like clams, maintain cholinergic function under varying salinity and metal stress conditions. This aligns with the observations of Freitas et al. (2019), who suggested that the maintenance or increase of AChE activity under stress could reflect an energy‐conserving strategy, allowing basic neural operations without excessive metabolic cost. The conservation of AChE activity supports the energy‐conserver strategy of mussels, minimizing costly neural adjustments while maintaining basic neuromuscular operations during environmental stress.
Overall, while R. philippinarum sustains cholinergic stability through partial osmoregulation without enhancing neuromuscular activity, M. galloprovincialis exhibits a higher baseline AChE activity, suggesting a more active but energy‐conserving neural strategy under environmental stressors, reflecting distinct adaptive mechanisms in neurotransmission regulation between the two species.
4.3 Comparison between Ruditapes philippinarum and Mytilus galloprovincialis biochemical performance
The contrasting biochemical profiles observed between clams and mussels reflect fundamental physiological differences between the two species and explain the divergence captured by PCO and IBR analyses. R. philippinarum exhibits a more flexible osmoregulatory strategy and maintains active ion transport under salinity stress, which increases mitochondrial respiration and ROS generation. This helps explain the activation of CAT and conjugative detoxification (GSTs) pathways when exposed stressful conditions, enabling clams to mobilize metabolic and protective mechanisms without incurring substantial cellular damage. The stable levels of oxidative stress and metabolism‐related biomarkers, together with comparatively lower IBR values, indicate an adaptive strategy that preserves physiological integrity while still investing energy in defense responses. In contrast, M. galloprovincialis tends to reduce metabolic rate under osmotic perturbation, especially in the presence of Gd, ATP by downregulating energetically expensive antioxidant enzymes and relying predominantly on biotransformation pathways (notably CbEs) to mitigate Gd effects. This metabolic depression strategy contributes to a higher degree of biochemical disturbance, even under lower internal Gd concentrations, and results in elevated oxidative stress signals and higher IBR values. Overall, these patterns reveal distinct adaptive strategies, such as energy investment in clams versus energy conservation in mussels. Collectively, the results highlight the clam's superior ability to buffer osmotic and contaminant pressures at multiple biological levels, emphasizing R. philippinarum as a particularly resistant and ecologically valuable sentinel species in environments impacted by emerging pollutants such as Gd.
From a broader perspective, the superior physiological fitness of R. philippinarum under adverse conditions may confer an ecological advantage, allowing this species to maintain growth and reproduction where more sensitive taxa, such as mussels, exhibit functional decline. This capacity not only strengthens its role in benthic community stability but also enhances its socio‐economic relevance, since clams represent a major aquaculture and fishery resource in many transitional and coastal systems. Their capacity to withstand combined salinity fluctuations and contaminant exposure translates into a more predictable yield and reduced vulnerability to stock collapses, offering clear advantages for food security and the sustainability of local economies dependent on bivalve harvesting. By contrast, the heightened sensitivity of mussels underscores their importance as early‐warning indicators but also signals potential risks for mussel aquaculture under scenarios of increasing chemical and osmotic stress.
4.4 Gadolinium toxicity in a multi‐stressor marine environment
In addition to salinity, other environmental stressors are likely to modulate the biological effects of Gd in marine organisms, and their combined action may amplify ecosystem‐level consequences. Recent experimental studies show that warming can greatly intensify Gd toxicity in bivalves: increased temperature enhanced Gd accumulation, oxidative stress and metabolic depression in M. galloprovincialis (Andrade et al., 2023) and exacerbated Gd‐induced biochemical disturbances in the surf clam Spisula solida under warmer and acidified conditions (Figueiredo et al., 2022). Similar multi‐stressor patterns have been reported for other rare earth elements, whose effects are magnified when combined with elevated temperature or salinity fluctuations in M. galloprovincialis, underscoring the relevance of climate‐related stressors in modulating REEs toxicity (Andrade et al., 2021; Leite et al., 2024). At the ecosystem level, global assessments emphasize that marine organisms are increasingly exposed to interacting drivers such as warming, acidification, hypoxia, altered salinity and metal/contaminant pollution, with combined effects frequently exceeding those predicted from single‐stressor studies (Defeo and Elliott, 2021; IPCC, 2023; Krishna et al., 2025). Therefore, the Gd and salinity responses reported here should be interpreted within a broader multi‐stressor context, and future studies should explicitly address how Gd interacts with other climate‐driven variables (e.g., temperature fluctuations, acidification) and co‐occurring contaminants to improve predictions of long‐term impacts on bivalve populations and the ecosystem services they sustain (Leite et al., 2024; Secco et al., 2024; Revel et al., 2025).
To advance this goal, future research should strengthen the integration of mathematical and computational models into ecotoxicological assessments of emerging pollutants. Progress beyond linear and single‐stressor dose–response assumptions will require models that incorporate species‐specific physiology, bioaccumulation kinetics and the combined influence of climate‐related stressors such as salinity and temperature. Coupling bioaccumulation modules with biomarker‐based toxicity models and adopting machine‐learning or Bayesian frameworks would help to detect non‐linear patterns and quantify uncertainty more effectively, providing more realistic and transferable tools for environmental risk assessment and management.
4.5 Potential human‐health risks linked to Gd‐contaminated bivalves
Recent studies have confirmed that marine bivalves efficiently bioaccumulate Gd in soft tissues, both under controlled exposures and in the field. Experimental work with mussels (M. galloprovincialis) and clams (S. solida, Donax trunculus) has shown dose‐ and time‐dependent Gd accumulation, often accompanied by marked biochemical disturbances under environmentally realistic scenarios (Figueiredo et al., 2022; Andrade et al., 2023; Secco et al., 2024). Field biomonitoring further reports positive Gd anomalies in different bivalve species collected from aquatic systems, demonstrating that anthropogenic Gd is effectively taken up by edible bivalves (Castro et al., 2023). Together with our results, these findings support the view that commercially important clams and mussels can act as vectors of anthropogenic Gd into marine food webs.
From a human‐health perspective, Gd‐based contrast agents (GBCAs), although fundamental for diagnostic imaging, are no longer considered completely risk‐free. Their use has been clearly associated with nephrogenic systemic fibrosis in patients with moderate to severe renal impairment, leading to major changes in clinical guidelines and stricter risk assessment for vulnerable populations (for review, see Agarwal et al., 2009). In addition, accumulating evidence shows that Gd from GBCAs can be retained long‐term in the brain, skin and other tissues, even in individuals with normal renal function and after repeated administrations, raising concerns about cumulative exposure (among others, Gulani et al., 2017; Guo et al., 2018; van der Molen et al., 2024). Although the clinical consequences of this retention are still under debate, these observations underline that Gd is not biologically inert in humans. In this context, the demonstrated capacity of bivalves to bioaccumulate Gd, combined with their high consumption in many coastal regions, highlights the need to better evaluate whether chronic dietary exposure via seafood may contribute, albeit likely at low levels, to overall Gd body burdens in humans.
5 Conclusion
This study demonstrates that Ruditapes philippinarum and Mytilus galloprovincialis adopt contrasting strategies when exposed to gadolinium (Gd) under varying salinity conditions. Clams accumulated substantially more Gd and adopted a “spender” strategy, characterized by strong activation of metabolic and antioxidant defenses at an energetic cost. Conversely, mussels accumulated less Gd and followed a “conserver” strategy, maintaining metabolic stability and limiting antioxidant investment, thereby conserving energy under combined contaminant and salinity stress. These findings reveal a clear species‐dependent divergence in physiological resilience, with clams showing a higher capacity to buffer cellular disruption and mussels showing greater vulnerability.
Beyond advancing fundamental ecotoxicology, the present results hold direct relevance for ecological risk assessment and environmental management. The species‐specific responses observed here demonstrate that the use of a single bioindicator may underestimate or overestimate the impacts of rare earth elements depending on the sentinel species chosen, reinforcing the need to incorporate interspecific variability into ecological risk models. Given the persistence of Gd in coastal ecosystems, its growing anthropogenic input via medical contrast agents, and the intensifying salinity fluctuations linked to climate change, our findings support the inclusion of Gd in coastal biomonitoring programs and seafood‐safety surveillance where bivalves are harvested for human consumption. Ultimately, the mechanistic insights provided here can help guide the development of predictive tools and monitoring strategies aimed at detecting early warning signs of Gd contamination, informing both regulatory frameworks and mitigation actions in estuarine and coastal environments.
Acknowledgments
Marta Cunha benefited from PhD grant (2024.01100.BD) funded by National Funds through the Portuguese Science Foundation (Fundação para a Ciência e a Tecnologia, FCT). This work was funded by national funds through FCT – Fundação para a Ciência e a Tecnologia I.P., under the project CESAM‐Centro de Estudos do Ambiente e do Mar, references UID/50017/2025 (doi.org/10.54499/UID/50017/2025) and LA/P/0094/2020 (doi.org/10.54499/LA/P/0094/2020).
Data availability statement
No new data/codes were created or analyzed in this study.
Supplementary Material
Table S1. p‐values for the comparisons between non‐contaminated and contaminated (CTL30 vs Gd30, CTL40 vs Gd40) organisms at each salinity, as well as between salinities for non‐contaminated (CTL30 vs CTL40) and contaminated organisms (Gd30 vs Gd40). When no significant differences were observed in the main test (p > 0.05), pairwise comparisons were not performed. In bold are represented pairwise comparisons with significant values (p < 0.05). Access Supplementary Material
References
- Aebi H. Catalase in vitro, in: Methods in Enzymology, Academic Press, 1984, Vol. 105, pp. 121–126. [Google Scholar]
- Agarwal R, Brunelli SM, Williams K Mitchell MD, Feldman HI, Umscheid CA. 2009. Gadolinium‐based contrast agents and nephrogenic systemic fibrosis: a systematic review and meta‐analysis. Nephrol Dial Transpl 24: 856–863. [Google Scholar]
- Anderson MJ, Gorley RN, Clarke KR. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods, 2008. www.primer-e.com [Google Scholar]
- Andrade M, Soares AMVM, Solé M, Pereira E, Freitas R. 2023. Gadolinium accumulation and its biochemical effects in Mytilus galloprovincialis under a scenario of global warming. Environ Sci Pollut Res 30: 116120–116133. [Google Scholar]
- Andrade M, Soares AMVM, Solé M, Pereira E, Freitas R. 2022. Will climate changes enhance the impacts of e‐waste in aquatic systems? Chemosphere 288: 132264. [Google Scholar]
- Andrade M, Soares AM, Solé M, Pereira E, Freitas R. 2021. Salinity influences on the response of Mytilus galloprovincialis to the rare‐earth element lanthanum. Sci Total Environ 794: 148512. [Google Scholar]
- Arrigo F, De Marchi L, Meucci V, Piccione G, Soares AMVM, Faggio C, Freitas R. 2024. Mytilus galloprovincialis: a valuable bioindicator species for understanding the effects of diclofenac under warming conditions. Sci Total Environ 946: 173809. [Google Scholar]
- Bates LS, Waldren RP, Teare ID. 1973. Rapid determination of free proline for water‐stress studies. Plant Soil 39: 205–207. [CrossRef] [Google Scholar]
- Bau M, Dulski P. 1996. Anthropogenic origin of positive gadolinium anomalies in river waters. Earth Planet Sci Lett 143(1–4): 245–255. [Google Scholar]
- Bau M, Knappe A, Dulski P. 2006. Anthropogenic gadolinium as a micropollutant in river waters in Pennsylvania and in Lake Erie, northeastern United States. Geochemistry 66: 143–152. [Google Scholar]
- Beliaeff B, Burgeot T. 2002. Integrated biomarker response: a useful tool for ecological risk assessment. Environ Toxicol Chem 21: 1216–1322. [Google Scholar]
- Boening DW. 1999. An evaluation of bivalves as biomonitors of heavy metals pollution in marine waters. Environ Monit Assess 55: 459‐470. [Google Scholar]
- Briant N, Le Monier P, Bruzac S, Sireau T, Araújo DF, Grouhel A. 2021. Rare earth element in bivalves’ soft tissues of French metropolitan coasts: spatial and temporal distribution. Arch Environ Contam Toxicol 81: 600–611. [Google Scholar]
- Brünjes R, Hofmann T. 2020. Anthropogenic gadolinium in freshwater and drinking water systems. Water Res 182: 115966. [Google Scholar]
- Buer AL, Taylor D, Bergström P, Ritzenhofen L, Klemmstein A. 2020. Nitrogen and phosphorous content in blue mussels (Mytilus spp.) across the Baltic Sea. Front Mar Sci 7: 705. [Google Scholar]
- Carregosa V, Velez C, Soares AMVM, Figueira E, Freitas R. 2014. Physiological and biochemical responses of three Veneridae clams exposed to salinity changes. Comp Biochem Physiol Part B: Biochem Mol Biol 177–178: 1–9. [Google Scholar]
- Castro L, Farkas J, Jenssen BM, Piarulli S, Ciesielski TM. 2023. Biomonitoring of rare earth elements in Southern Norway: Distribution, fractionation, and accumulation patterns in the marine bivalves Mytilus spp. and Tapes spp.. Environmental Pollution 335: 122300. [Google Scholar]
- Chen Z, Li X, Gao J, Liu Y, Zhang N, Guo Y, Wang Z, Dong Z. 2024. Effects of salinity on behavior and reproductive toxicity of BPA in adult marine medaka. Chemosphere 357: 142103. [Google Scholar]
- Čolović MB, Krstić DZ, Lazarević‐Pašti TD, Bondžić AM, Vasic VM. 2013. Acetylcholinesterase inhibitors: pharmacology and toxicology. Curr Neuropharmacol 11: 315–335. [Google Scholar]
- Coppola F, Bessa A, Henriques B, Russo T, Soares AMVM, Figueira E, Marques PAAP, Polese G, Di Cosmo A, Pereira E, Freitas R. 2020. Oxidative stress, metabolic and histopathological alterations in mussels exposed to remediated seawater by GO‐PEI after contamination with mercury. Comp Biochem Physiol Part A: Mol Integr Physiol 243: 110674. [Google Scholar]
- Coughlan BM, Moroney GA, Pelt FNAMV, O'Brien NM, Davenport J, O'Halloran J. 2009. The effects of salinity on the Manila clam (Ruditapes philippinarum) using the neutral red retention assay with adapted physiological saline solutions. Mar Pollut Bull 58: 1680–1684. [Google Scholar]
- Cunha M, Louro P, Silva M, Soares AMVM, Pereira E, Freitas R. 2022. Biochemical alterations caused by lanthanum and gadolinium in Mytilus galloprovincialis after exposure and recovery periods. Environ Pollut 307: 119387. [Google Scholar]
- Dame RF. Ecology of Marine Bivalves: An Ecosystem Approach, 2nd edn, CRC Press, 2012, p. 283. [Google Scholar]
- De Coen WM, Janssen CR. 1997. The use of biomarkers in Daphnia magna toxicity testing. IV. Cellular energy allocation: a new methodology to assess the energy budget of toxicant-stressed Daphnia populations J Aquat Ecosyst Stress Recover 6 (Kluwer Academic Publishers). [Google Scholar]
- Defeo O, Elliott M. 2021. The ‘triple whammy’ of coasts under threat – why we should be worried! Mar Pollut Bull 163: 111832. [Google Scholar]
- Dias JM, Lopes JF, Dekeyser I. 2000. Tidal propagation in Ria de Aveiro Lagoon, Portugal. Phys Chem Earth Part B: Hydrol Oceans Atmos 25: 369–374. [Google Scholar]
- DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. 1956. Colorimetric method for determination of sugars and related substances. Anal Chem 28: 350–356. [CrossRef] [Google Scholar]
- Ellman GL, Courtney KD, Andres V, Featherstone RM. 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7: 88–95. [CrossRef] [PubMed] [Google Scholar]
- Fernández B, Campillo JA, Martínez‐Gómez C, Benedicto J. 2012. Assessment of the mechanisms of detoxification of chemical compounds and antioxidant enzymes in the digestive gland of mussels, Mytilus galloprovincialis, from Mediterranean coastal sites. Chemosphere 87: 1235–1245. [Google Scholar]
- Figueiredo C, Grilo TF, Lopes C, Brito P, Caetano M, Raimundo J. 2022. Lanthanum and Gadolinium availability in aquatic mediums: New insights to ecotoxicology and environmental studies. J Trace Elem Med Biol 71: 126957. [Google Scholar]
- Freitas R, Almeida Â, Calisto V, Velez C, Moreira A, Schneider RJ, Esteves VI, Wrona FJ, Figueira E, Soares AMVM. 2016. The impacts of pharmaceutical drugs under ocean acidification: New data on single and combined long‐term effects of carbamazepine on Scrobicularia plana. Sci Total Environ 541: 977–985. [Google Scholar]
- Freitas R, Cardoso CED, Costa S, Morais T, Moleiro P, Lima AFD, Soares M, Figueiredo S, Águeda TL, Rocha P, Amador G, Soares AMVM, Pereira E. 2020. New insights on the impacts of e‐waste towards marine bivalves: the case of the rare earth element dysprosium. Environ Pollut 260: 113859. [Google Scholar]
- Freitas R, Leite C, Pinto J, Costa M, Monteiro R, Henriques B, Di Martino F, Coppola F, Soares AMVM, Solé M, Pereira E. 2019. The influence of temperature and salinity on the impacts of lead in Mytilus galloprovincialis. Chemosphere 235: 403–412. [Google Scholar]
- Freitas RO, Fraga M. Relatório Sanitário Para Zonas de Produção de Moluscos Bivalves: Ria de Aveiro, Instituto Português do Mar e da Atmosfera, 2020. [Google Scholar]
- Gissi E, Manea E, Mazaris AD, Fraschetti S, Almpanidou V, Bevilacqua S, Coll M, Guarnieri G, Lloret‐Lloret E, Pascual M, Petza D, Rilov G, Schonwald M, Stelzenmüller V, Katsanevakis S. 2021. A review of the combined effects of climate change and other local human stressors on the marine environment. Sci Total Environ 755: 142564. [Google Scholar]
- Gulani V, Calamante F, Shellock FG, Kanal E, Reeder SB. 2017. Gadolinium deposition in the brain: summary of evidence and recommendations. Lancet Neurol 16: 564–570. [Google Scholar]
- Guo BJ, Yang ZL, Zhang LJ. 2018. Gadolinium deposition in brain: current scientific evidence and future perspectives. Front Mol Neurosci 11: 335. [Google Scholar]
- Habig WH, Pabst MJ, Jakoby WB. 1974. Glutathione S‐transferases. J Biol Chem 249: 7130–7139. [CrossRef] [PubMed] [Google Scholar]
- Hanana H, Turcotte P, André C, Gagnon C, Gagné F. 2017. Comparative study of the effects of gadolinium chloride and gadolinium – based magnetic resonance imaging contrast agent on freshwater mussel, Dreissena polymorpha. Chemosphere 181: 197–207. [Google Scholar]
- Henriques B, Coppola F, Monteiro R, Pinto J, Viana T, Pretti C, Soares A, Freitas R, Pereira E. 2019. Toxicological assessment of anthropogenic Gadolinium in seawater: biochemical effects in mussels Mytilus galloprovincialis. Sci Total Environ 664: 626–634. [Google Scholar]
- Henriques IS, Alves A, Tacão M, Almeida A, Cunha Â, Correia A. 2006. Seasonal and spatial variability of free‐living bacterial community composition along an estuarine gradient (Ria de Aveiro, Portugal). Estuar Coast Shelf Sci 68(1–2): 139–148. [Google Scholar]
- Hosokawa M, Satoh T. 2002. Measurement of carboxylesterase (CES) activities. Curr Protoc Toxicol 4: 4.7.1–4.7.14. [Google Scholar]
- IPCC. 2019. Climate change and land: an IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems, in: P. R. Shukla, J. Skea, E. Calvo Buendía, V. Masson‐Delmotte, H.‐O. Pörtner, D. C. Roberts, P. Zhai, R. Slade, S. Connors, R. van Diemen, et al. (Eds.), Intergovernmental Panel on Climate Change, www.ipcc.ch/srccl/ [Google Scholar]
- IPCC. 2023. Climate change 2023: synthesis report. Summary for policymakers, in: Core Writing Team, H. Lee, J. Romero (Eds.), Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, IPCC, 10.59327/IPCC/AR6‐9789291691647.001 [Google Scholar]
- Krasznai Z, Morisawa M, Krasznai ZT, Morisawa S, Inaba K, Bazsáné ZK, Rubovszky B, Bodnár B, Borsos A, Márián T. 2003. Gadolinium, a mechano‐sensitive channel blocker, inhibits osmosis‐initiated motility of sea‐ and freshwater fish sperm, but does not affect human or ascidian sperm motility. Cell Motility 55: 232–243. [Google Scholar]
- Krishna S, Lemmen C, Örey S, Rehren J, Pane JD, Mathis M, Wirtz KW. 2025. Interactive effects of multiple stressors in coastal ecosystems. Front Mar Sci 11: 1481734. [Google Scholar]
- Lachaux N, Otero‐Fariña A, Minguez L, Sohm B, Rétif J, Châtel A, Poirier L, Devin S, Pain‐Devin S, Gross EM, Giamberini L. 2023. Fate, subcellular distribution and biological effects of rare earth elements in a freshwater bivalve under complex exposure. Sci Total Environ 905: 167302. [Google Scholar]
- Laczovics A, Csige I, Szabó S, Tóth A, Kálmán FK, Tóth I, Braun M. 2023. Relationship between gadolinium‐based MRI contrast agent consumption and anthropogenic gadolinium in the influent of a wastewater treatment plant. Sci Total Environ 877: 162844. [Google Scholar]
- Leite C, Russo T, Polese G, Soares AMVM, Pretti C, Pereira E, Freitas R. 2024. Effects of the interaction of salinity and rare earth elements on the health of Mytilus galloprovincialis: the case of Praseodymium and Europium. J Xenobiotics 14: 2015–2038. [Google Scholar]
- Li Y, Li Y, Lai Q, Yao Z, Zhou K, Niu D. 2024. Proline metabolism and its osmotic regulatory role in enhancing hypersalinity tolerance of the razor clam (Sinonovacula constricta). Aquaculture 589. https://doi.org/10.1016/j.aquaculture.2024.741019. [Google Scholar]
- Lopes JF, Lopes CL, Dias JM. 2021. Extreme meteorological events in a coastal lagoon ecosystem: the ria de Aveiro lagoon (Portugal) case study. J Mar Sci Eng 9: 727. [Google Scholar]
- Magnani L, Gaydou EM, Hubaud JC. 2000. Spectrophotometric measurement of antioxidant properties of flavones and flavonols against superoxide anion. Anal Chim Acta 411(1–2): 209–216. [Google Scholar]
- Mennillo E, Casu V, Tardelli F, De Marchi L, Freitas R, Pretti C. 2017. Suitability of cholinesterase of polychaete Diopatra neapolitana as biomarker of exposure to pesticides: In vitro characterization. Comp Biochem Physiol Part C: Toxicol Pharmacol 191: 152–159. [Google Scholar]
- Mesquita CS, Oliveira R, Bento F, Geraldo D, Rodrigues JV, Marcos JC. 2014. Simplified 2,4‐dinitrophenylhydrazine spectrophotometric assay for quantification of carbonyls in oxidized proteins. Anal Biochem 458: 69–71. [Google Scholar]
- Moreira I, Pereira E, Soares AMVM, Freitas R. 2025. Gadolinium: a review on concentrations and impacts in marine and coastal systems. Environ Pollut 381: 126453. [Google Scholar]
- Mossotto C, Anselmi S, Trevisan S, Provenza F, Maganza A, Gabetti A, Esposito G, Elia AC, Prearo M, Renzi M, Pastorino P. 2025. Assessing the toxicity of gadolinium in freshwater and marine ecosystems: effects across trophic levels. Environ Toxicol Pharmacol 115: 104673. [Google Scholar]
- Munari M, Matozzo V, Marin MG. 2011. Combined effects of temperature and salinity on functional responses of haemocytes and survival in air of the clam Ruditapes philippinarum. Fish Shellfish Immunol 30(4–5): 1024–1030. [Google Scholar]
- Ohkawa H, Ohishi N, Yagi K. 1979. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95: 351–358. [CrossRef] [PubMed] [Google Scholar]
- Packard T, King F. 1975. Respiration and the activity of the respiratory electron transport system in marine zooplankton1. Limnol Oceanogr 20: 849–854. [Google Scholar]
- Patel N, Khan MDZA, Shahane S, Rai D, Chauhan D, Kant C, Chaudhary VK. 2020. Emerging pollutants in aquatic environment: source, effect, and challenges in biomonitoring and bioremediation‐ a review. Pollution 6. https://doi.org/10.22059/poll.2019.285116.646. [Google Scholar]
- Perrat E, Parant M, Py J‐S, Rosin C, Cossu‐Leguille C. 2017. Bioaccumulation of gadolinium in freshwater bivalves. Environ Sci Pollut Res 24: 12405–12415. [Google Scholar]
- Queirós V, Leite C, Azeiteiro UM, Belloso MC, Soares AMVM, Santos JL, Alonso E, Barata C, Freitas R. 2024. Salinity influence on Mytilus galloprovincialis exposed to antineoplastic agents: a transcriptomic, biochemical, and histopathological approach. Environ Pollut 363: 125135. [Google Scholar]
- Rainbow PS. 2002. Trace metal concentrations in aquatic invertebrates: why and so what? Environ Pollut 120: 497–507. [Google Scholar]
- Regoli F, Giuliani ME. 2014. Oxidative pathways of chemical toxicity and oxidative stress biomarkers in marine organisms. Mar Environ Res 93: 106–117. [Google Scholar]
- Rétif J, Zalouk‐Vergnoux A, Briant N, François Y, Poirier L. 2024a. Trophic dilution of rare earth elements along the food chain of the Seine estuary (France). Mar Pollut Bull 206: 116671. [Google Scholar]
- Rétif J, Zalouk‐Vergnoux A, Kamari A, Briant N, Poirier L. 2024b. Trophic transfer of rare earth elements in the food web of the Loire estuary (France). Sci Total Environ 914: 169652. [Google Scholar]
- Revel M, Van Drimmelen CK, Weltje L, Hursthouse A, Heise S. 2025. Effects of rare earth elements in the aquatic environment: implications for ecotoxicological testing. Crit Rev Environ Sci Technol 55: 334–375. [Google Scholar]
- Rivera‐Ingraham GA, Lignot J‐H. 2017. Osmoregulation, bioenergetics and oxidative stress in coastal marine invertebrates: raising the questions for future research. J Exp Biol 220: 1749–1760. [Google Scholar]
- Robinson HW, Hogden CG. 1940. The biuret reaction in the determination of serum proteins. J Biol Chem 135: 727–731. [Google Scholar]
- Rogowska J, Olkowska E, Ratajczyk W, Wolska L. 2018. Gadolinium as a new emerging contaminant of aquatic environments. Environ Toxicol Chem 37: 1523–1534. [Google Scholar]
- Schmidt K, Bau M, Merschel G, Tepe N. 2019. Anthropogenic gadolinium in tap water and in tap water-based beverages from fast-food franchises in six major cities in Germany. Sci Total Environ 687: 1401–1408. [Google Scholar]
- Sanchez W, Burgeot T, Porcher J‐M. 2013. A novel “Integrated Biomarker Response” calculation based on reference deviation concept. Environ Sci Pollut Res 20: 2721–2725. [Google Scholar]
- Sauvé S, Desrosiers M. 2014. A review of what is an emerging contaminant. Chem Central J 8: 15. [Google Scholar]
- Santos D, Leite C, Pinto J, Soares AM, Pereira E, Freitas R. 2023. How will different scenarios of rising seawater temperature alter the response of marine species to lithium?. Sci Total Environ 856: 158728. [Google Scholar]
- Santos L, Cunha Â, Silva H, Caçador I, Dias JM, Almeida A. 2007. Influence of salt marsh on bacterial activity in two estuaries with different hydrodynamic characteristics (Ria de Aveiro and Tagus Estuary): influence of salt marsh on bacterial activity in estuaries. FEMS Microbiol Ecol 60: 429–441. [Google Scholar]
- Secco S, Cunha M, Libralato G, Trifuoggi M, Giarra A, Soares AMVM, Freitas R, Scalici M. 2024. Evaluating the impact of gadolinium contamination on the marine bivalve Donax trunculus: implications for environmental health. Environ Toxicol Pharmacol 112: 104580. [Google Scholar]
- Solé M, Rivera‐Ingraham G, Freitas R. 2018. The use of carboxylesterases as biomarkers of pesticide exposure in bivalves: a methodological approach. Comp Biochem Physiol Part C: Toxicol Pharmacol 212: 18–24. [Google Scholar]
- Spampinato M, Siciliano A, Travaglione A, Chianese T, Mileo A, Libralato G, Guida M, Trifuoggi M, De Gregorio V, Rosati L. 2024. Unravelling the ecotoxicological impacts of gadolinium (Gd) on Mytilus galloprovincialis embryos and sperm in seawater: a preliminary study. Heliyon 10: e31087. [Google Scholar]
- Stączek S, Kunat‐Budzyńska M, Cytryńska M, Zdybicka‐Barabas A. 2024. Proline‐rich antimicrobial peptides from invertebrates. Molecules 29: 5864. [Google Scholar]
- Sturla Lompré J, Cunha M, Pinto J, Soares AMVM, Pereira E, Freitas R. 2025. Cellular responses of Ruditapes philippinarum clams to global changes: assessing the risks of rising temperatures and E‐waste. Estuar Coast Shelf Sci 319: 109262. [Google Scholar]
- Trapasso G, Chiesa S, Freitas R, Pereira E. 2021. What do we know about the ecotoxicological implications of the rare earth element gadolinium in aquatic ecosystems? Sci Total Environ 781: 146273. [Google Scholar]
- Telgmann L, Wehe CA, Birka M, Kuünnemeyer J, Nowak S, Sperling M, Karst U. 2012. Speciation and isotope dilution analysis of gadolinium-based contrast agents in wastewater. Environ Sci Technol 46: 11929–11936. [Google Scholar]
- van der Molen AJ, Quattrocchi CC, Mallio CA, Dekkers IA, European Society of Magnetic Resonance in Medicine, Biology Gadolinium Research, Educational Committee (ESMRMB‐GREC). 2024. Ten years of gadolinium retention and deposition: ESMRMB‐GREC looks backward and forward. Eur Radiol 34: 600–611. [Google Scholar]
- Vaz L, Frankenbach S, Serôdio J, Dias JM. 2019. New insights about the primary production dependence on abiotic factors: Ria de Aveiro case study. Ecol Indic 106: 105555. [Google Scholar]
- Velez C, Figueira E, Soares AMVM, Freitas R. 2017. Effects of seawater temperature increase on economically relevant native and introduced clam species. Mar Environ Res 123: 62–70. [Google Scholar]
- Willett CS, Burton RS. 2002. Proline biosynthesis genes and their regulation under salinity stress in the euryhaline copepod Tigriopus californicus. Comp Biochem Physiol Part B: Biochem Mol Biol 132: 739–750. [Google Scholar]
- Yang F, Zhao L, Yan X, Wang Y. 2013. Bioaccumulation of trace elements in Ruditapes philippinarum from China: public health risk assessment implications. Int J Environ Res Public Health 10: 1392–1405. [Google Scholar]
- Zhou C, Xu L, Song H, Feng J, Hu Z, Yang M‐J, Shi P, Li Y‐R, Guo Y‐J, Li H‐Z, Zhang T. 2023. Examination of the regulation of energy metabolism, antioxidant response, and ammonia detoxification in hard clam, Mercenaria mercenaria, under hypersalinity stress. Aquaculture 563: 738916. [Google Scholar]
- Zitoun R, Marcinek S, Hatje V, Sander SG, Völker C, Sarin M, Omanović D. 2024. Climate change driven effects on transport, fate and biogeochemistry of trace element contaminants in coastal marine ecosystems. Commun Earth Environ 5: 560. [Google Scholar]
- Zuykov M, Pelletier E, Harper DAT. 2013. Bivalve mollusks in metal pollution studies: From bioaccumulation to biomonitoring. Chemosphere 93: 201–208. [Google Scholar]
Cite this article as: Moreira I, Cunha M, Leite C, Soares AMVM, Pereira E, Freitas R. 2026. Contrasting responses of Ruditapes philippinarum and Mytilus galloprovincialis to gadolinium and salinity stress. Aquat. Living Resour. 39: 5. https://doi.org/10.1051/alr/2025023
All Tables
Gadolinium (Gd) concentration (µg/g dry weight (DW)) in the dried soft tissues of clams after 14 days of non‐exposure and exposure to 10 µg/L of Gd at salinities of 30 or 40.
Gadolinium (Gd) concentration (µg/g dry weight (DW)) in the dried soft tissues of mussels after 14 days of non‐exposure and exposure to 10 µg/L of Gd at salinities of 30 or 40.
Integrated Biomarker Response index version 2 (IBRv2) in Ruditapes philippinarum and Mytilus galloprovincialis after 14 days of exposure to 0 μg/L (CTL) and 10 μg/L of gadolinium (Gd) at different salinity levels (30 and 40).
All Figures
 |
Fig. 1 A: Electron transport system (ETS) activity; B: Carbohydrates (CARB) content; C: Protein (PROT) content, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are presented as means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
| In the text | |
 |
Fig. 2 A: Superoxide dismutase (SOD) activity; B: Catalase (CAT) activity; C: Carboxylesterases (CbEs) activity with pNPB substrate; D: Glutathione S‐transferases (GSTs) activity, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are presented as means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
| In the text | |
 |
Fig. 3 A: Proline (PROL) content; B: Lipid peroxidation (LPO) levels; C: Protein carbonylation (PC) levels, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
| In the text | |
 |
Fig. 4 Acetylcholinesterase (AChE) activity, in clams and mussels exposed to 0 µg/L (CTL, white bars) and 10 µg/L of gadolinium (Gd, colored bars) at different salinity levels (30 and 40). Results are means with standard deviations. For each species, different lowercase letters represent significant differences (p < 0.05) between non‐exposed individuals; for each species, different uppercase letters represent significant differences (p < 0.05) between Gd‐exposed individuals; asterisks represent significant differences (p < 0.05) between Gd‐exposed and non‐exposed treatments. |
| In the text | |
 |
Fig. 5 Centroid ordination diagram of Principal Coordinates Analysis (PCO) based on tested treatments (control, CTL; Gadolinium, Gd; salinities 30 or 40) and biochemical parameters measured in Ruditapes philippinarum (C) and Mytilus galloprovincialis (M). Biochemical parameters are represented with name abbreviations. Correlation vectors (r > 0.70) are superimposed as supplementary variables. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


