| Issue |
Aquat. Living Resour.
Volume 39, 2026
|
|
|---|---|---|
| Article Number | 9 | |
| Number of page(s) | 9 | |
| DOI | https://doi.org/10.1051/alr/2026001 | |
| Published online | 13 March 2026 | |
Research Article
Juvenile growth performance and associated genetic parameters in common carp, Cyprinus carpio cultured in 8 ppt inland saline groundwater
1
Division of Fish Genetics and Biotechnology, Indian Council of Agricultural Research (ICAR)-Central Institute of Fisheries Education, Mumbai, India
2
Department of Breeding and Genetics, Nofima, Ås, Norway
3
Fish Genetics and Breeding, Indian Council of Agricultural Research (ICAR)-Central Institute of Fisheries Education, Rohtak centre, Rohtak, Haryana, India
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
2
May
2025
Accepted:
4
February
2026
Abstract
The degraded soils unfit for primary agricultural crops can be utilised for inland saline aquaculture of saline-tolerant species. In India, a genetic improvement program for developing a faster-growing, saline-tolerant strain of common carp has been initiated. The present study reports juvenile growth performance and genetic parameters for common carp reared at 8-9 ppt salinity. The population comprised 5075 individuals belonging to 86 full-sib families generated by single-pair mating. Until 30 days post hatch (dph) (mean body weight 1.8 g) the families were reared in separate hapas in freshwater, and thereafter until 75 dph (5.3 g) in separate hapas at 8 ppt salinity until attaining a suitable body size for PIT-tagging at 150 dph (28.1 g). Thereafter, they were reared communally in two ponds until 195 dph (55.0 g). The body weight and body length at 30, 75, 150, and 195 dph, and weight gain (WG150-195) and specific growth coefficient (SGR150-195) were analysed. The effect of stock and pond on the traits was non-significant at 30 and 75 dph, whereas pond had a significant effect on the traits at 150 and 195dph. The heritability estimates for body weight at 30 dph and body weight and length at 75 dph were medium to high (0.52-0.68), and those at 150 and 195 dph and WG150-195, and SGR150-195 were moderate (0.29-0.43). The genetic correlation of BW150 and BW195 with WG150-195 was high (0.67 ± 0.09 to 0.90 ± 0.04), whereas that of BW150 and BW195 with SGR was negative (–0.41 ± 0.14 to –0.60 ± 0.10). The genetic parameters for juvenile traits are likely to be inflated due to the full-sib family structure and their separate early rearing in hapas. A follow-up study is required to estimate genetic parameters for body weight at the desired market size in carp reared in saline earthen ponds.
Key words: Inland saline / common carp / early growth traits / genetic parameter
Handling Editor: Pierre Boudry
© R. Raghul et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Soil salinisation is an ongoing threat to agricultural lands worldwide, affecting 424 million hectares of topsoil and 833 million hectares of subsoil (FAO, 2021). In India, 6.74 million hectares of land are salt-affected (Kumar and Sharma, 2020) of which 75% are in the states of Gujarat (2.23 million ha), Uttar Pradesh (1.37 million ha), Maharashtra (0.61 million ha), West Bengal (0.44 million ha), and Rajasthan (0.38 million ha) (Mandal et al., 2018). Soil salinisation significantly reduces cultivable land area and adversely affects agricultural productivity, crop choices, biodiversity, water quality, infrastructure durability, and livelihood security. Restoring salt-affected degraded lands presents a crucial opportunity to sustain food security by improving these compromised agro-ecosystems. Technological interventions like alternative land-use systems, saline aquaculture, salt-tolerant crop cultivation, agroforestry, phytoremediation, and bioremediation have proven effective in enhancing food and nutritional security, empowering women, engaging landless laborers, reducing rural migration, and restoring ecological balance through positive environmental impacts (Sharma and Chaudhari, 2012; Kumar and Sharma, 2020).
The utilisation of inland saline groundwater (ISGW) affected lands for aquaculture farming is an alternate approach for making these degraded lands into profitable ventures (Allan et al., 2009, Singh et al., 2018). Inland saline aquaculture integrates social, ecological, and economic dimensions to promote sustainable development in salt-affected regions. Socially, it enhances rural livelihoods by creating employment and supporting marginalized groups, while promoting food security. Ecologically, it utilizes inland saline waters and marginal lands, relieving pressure on freshwater resources and contributing to land reclamation through sustainable resource management. Economically, it supports species diversification and resilience by enabling profitable production of adaptable species like shrimp, tilapia, and carp (CSSRI, 2013; Belton et al., 2017; Kumar and Sharma, 2020, Rossignoli et al., 2023). However, managing ecological risks such as salinity fluctuations and water quality is essential to ensure the long-term sustainability of these socio-ecological systems.
Common carp is the oldest domesticated species, native to Europe and Asia, and one of the most popular species for aquaculture (Balon, 1995). With a production of 4.2363 million tonnes, it is the fourth highest-produced fish in global aquaculture, with a percentage share of 8.6 (FAO, 2022). Common carp is a candidate species for inland saline aquaculture and can tolerate salinity up to 12 ppt (Wang et al., 1997; Iffat et al., 2021, Rajanand, 2016; Phibi, 2017), but its growth is negatively affected by salinity beyond 6 ppt (Anand et al., 2022). Genetically improved carp strains are expected to yield more than local strains and boost aquaculture production (Dey et al., 2013). The genetic improvement in common carp has been practiced through crossbreeding or hybridisation, which exploits heterosis effects (Hume et al., 1983; Wohlfarth, 1993; Chen et al., 2022). Various recent studies have shown a significant additive genetic variation in harvest body weight, growth rate expressed as specific growth rate, SGR, and other performance traits in common carp and other cultured species (Saillant et al., 2006; Kocour et al., 2007; Vandeputte et al., 2008; Ma et al., 2008; Nielsen et al., 2010; Mas-Muñoz., 2013; Dong et al., 2015; Prchal et al., 2018), suggesting the possibility of improving them through selective breeding. In common carp, growth is a moderately heritable trait (Kocour et al., 2007; Nielsen et al., 2010).
In fish, early growth traits viz., body weight and body length, exhibit moderate to high heritability and high genetic correlation between traits recorded at the same age and only limited studies shown the possibility of improving early growth/juvenile traits in common carp (Vandeputte et al., 2004; Yousefian et al., 2011, Palaiokostas et al., 2018), grass carp (Fu et al., 2015), black bream (Doupe et al., 2003; Doupe and Lymbery, 2005a), sea bass (Chandra et al., 2000), hybrid striped bass (Wang et al., 2006), tilapia (Tave and Smitherman, 1980), yellow croaker (Yu et al., 2020), oliveflounder (Li et al., 2019), brown trout (Vandeputte et al., 2002) and red drum (Saillant et al., 2007) through selective breeding and none of them reported the extent to which the same trait at different ages is affected by the same or by different genes; However, estimates of genetic correlations between growth traits at different ages are few (Crandell and Gall, 1993; Gjerde et al., 1994; Nilsson, 1994; Doupe and Lymbery, 2005b; Saillant et al., 2006; Wang et al., 2006; Vandeputte, 2008; Nielson et al., 2010; Hu et al., 2017) but are required to predict the correlated responses in harvest weight when preselection for growth at an early age or the correlated gain in early growth when selection for harvest weight. Early selection for growth rate may reduce the generation interval and thus both the genetic gain per year and the cost of the breeding program. However, if the proportion of sexual maturing fish at an early age is lower, and early sexual maturity maybe also a not desired trait, the increased expected benefit of early selection for growth may be lost through a reduced selection intensity and undesirable correlated gains in sexual maturity and other traits.
Recent studies suggest that combining non-random visual selection of larger individuals with random sampling at tagging can enhance genetic improvement for harvest weight, particularly in carp (Hamilton et al., 2022). A study in juvenile olive flounder demonstrated that early growth rates may serve as predictor of later growth, up to market size, provided there is a positive genetic correlation between growth at later stages (Li et al., 2019). This is contradicted by weak/no genetic correlation between ages in fish species (Chandra et al., 2000; Vandeputte et al., 2008; Nielsen et al., 2010). Studies have reported strong positive phenotypic correlations between growth-related traits measured at the juvenile and harvest stages (Ninh et al., 2013), whereas other research has found correlations close to zero (Hu et al., 2017), indicating variability in the correlation depending on the age of the population or species studied. However, careful evaluation of this relationship is essential before relying on juvenile performance as a reliable predictor of harvest weight in breeding programs.
In India, a genetic improvement program is initiated to develop a faster-growing, saline-tolerant strain of common carp for inland saline aquaculture. Earlier, a base population of common carp (F0) was established from five geographic stocks, and their genetic diversity was assessed using morphometry and mt-D loop analysis (Lalramnunsanga et al., 2024). The present study was performed on the offspring of selected fish from this base population. The goal was to evaluate performance and estimate genetic parameters for early growth-related traits during an initial rearing period in which families were reared in separate hapa and, thereafter, individually PIT-tagged and reared communally in a pond at a salinity of 8-9 ppt.
2 Materials and methods
2.1 Ethical statement
The present experiment was part of a research project funded by the World Bank-ICAR NAHEP to the ICAR- Central Institute of Fisheries Education (Deemed University), Mumbai, India. The experimental procedures followed complied with the institute's guidelines.
2.2 Induced spawning and family production
The broodfish (offspring of wild animal) from five stocks with high body weight at 200 days of age were selected based on their estimated breeding values (EBVs). Prior to the induced spawning the broodfish were fed at rate of 3% of body weight with ABIS grower floating fish feed (size 4mm, crude protein: 28%, fat: 3% and fiber: 7%). For induced spawning, the brooders were injected a single dose of commercially available Gonopro FH® synthetic hormone at 0.4 ml/kg for females and 0.2-0.3 ml/kg for males during evening hours. After the injection, the pair of male and female brood fish were released into separate spawning hapas (2m x 1m x 1m) placed in a pond with freshwater (salinity 0 ppt) and plastic strips were provided as substratum for the adhesion of the fertilized eggs. The spawning occurred within 16 h of injection, after which the male and female brooders were removed from the hapa. A total of 86 full-sib families were produced by single pair mating within each of the five stocks between 17 and 22 March 2022 (Tab. 1).
Stocks of common carp used for the production of families and the number of families stocked into the separate rearing hapas in the two earthen ponds A and B.
2.3 Fry nursery rearing
The fertilized eggs hatched in the spawning hapas (Fig. 1), and three days post-hatching, after yolk sac absorption, larval feeding was initiated. The commercial ABIS hatchery feed (size 250 micron, crude protein: 38%, fat: 6% and fiber: 3%) was fed to the larvae twice a day at 6% body weight.
 |
Fig. 1 Spawning hapa for common carp. |
2.4 Juvenile rearing
After one month of rearing in the spawning hapas, each family was shifted to rearing hapa (6.5m × 2m × 1.5 m) in two earthen saline ponds (Pond A with 59 hapas and Pond B with 27 hapas) (Tab. 1), each of size 1400 m2 and water depth of 1.5 m (Fig. 2). The same water source and aquaculture conditions were maintained across both ponds. The stocking density in the rearing hapas was about 4.5 fish/m3. The salinity was gradually increased by 2 ppt each day until it reached 8–9 ppt, and this level was maintained throughout the 45-day culture period from 30 to 75 dph and 75 days (from 75 dph to 150 dph until tagging) in rearing hapa and further rearing period upto 195 dph. Water quality parameters such as temperature (°C) and salinity (ppt) were monitored regularly using a multiple-parameter water quality meter (DKK-TOA, WQC-24, Japan). The observed salinity in the water during the culture period was between 8.05–9.02 ppt, while the water temperature, DO and pH ranged between 30-33 °C, 4.5–5.5 mg/l and 8.02–8.52, respectively.
 |
Fig. 2 Rearing hapa for common carp. |
2.5 Tagging and communal rearing
At 150 dph, 1740 fish belonging to 86 families were randomly sampled and individually tagged with PIT tags. After tagging, the fish were observed in FRP tanks for 72 hrs to check for mortality due to tagging. No significant mortality was observed within this period. After three days of conditioning, the fish were pooled and put back into the same earthen ponds (Pond A with 949 fish from 47 families and Pond B with 791 fish from 39 families, resulting in a total of 1,740 fish) for communal rearing in 8-9 ppt salinity after the rearing hapas had been removed from these ponds.
2.6 Recording of growth traits
The individual fish body weight at 30 dph (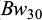 ); i.e. body weight after one month in the spawning hapas with freshwater) was recorded prior to stocking the families into the rearing hapas in the two saline ponds. After 45 days culture in the rearing hapas (8-9 ppt saline water), complete harvesting of a total of 2952 individuals were performed using a dip net, after which their individual body weights (
); i.e. body weight after one month in the spawning hapas with freshwater) was recorded prior to stocking the families into the rearing hapas in the two saline ponds. After 45 days culture in the rearing hapas (8-9 ppt saline water), complete harvesting of a total of 2952 individuals were performed using a dip net, after which their individual body weights (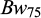 ) and body lengths (
) and body lengths (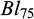 ), were recorded and they were released back to their same two rearing hapas. Further, 1740 fish were recorded for their body weights (
), were recorded and they were released back to their same two rearing hapas. Further, 1740 fish were recorded for their body weights (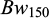 ) and body lengths (
) and body lengths (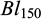 ) at tagging. Further, at 45 days of communal rearing in the two ponds, their individual body weights and body lengths
) at tagging. Further, at 45 days of communal rearing in the two ponds, their individual body weights and body lengths 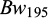 &
& 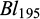 ) were recorded on a random partial sampling of 982 fish. The body weights were recorded to the nearest 0.01 g using a portable weighing balance, and the body lengths (to the nearest 0.1mm) with a ruler.
) were recorded on a random partial sampling of 982 fish. The body weights were recorded to the nearest 0.01 g using a portable weighing balance, and the body lengths (to the nearest 0.1mm) with a ruler.
2.7 Data analysis
For individual fish the body weight gain from day 150 (Bw150) to day 195 (Bw195) was calculated as:
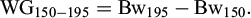
To account for the possible hapa effect on the growth rate of the fish until tagging at day 150, the specific growth rate (SGR, %/day; Hopkins, 1992) from day 150 until day 195 was calculated as:
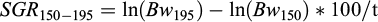
where t is the duration of the experiment (45 days).
Estimates of (co)variances of the random animal, residual effects in the below mixed linear model for the studied traits were obtained from a single trait analyses of Bw30, bivariate analyses of the Bw75 and Bl75, and six bivariate analyses of all combinations of the traits Bw150, Bw195, WG150-95 and SGR150-95, using the restricted maximum likelihood (REML) algorithm, implemented by ASReml 4.1 software (Gilmour et al., 2015). In a model with combinations of three of the four above-mentioned traits, the parameters and/or the log-likelihood did converge. In matrix notation, the model can be written as:
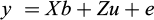
where the dependent variable is a column vector of the single trait (Bw30), or the seven above mentioned bivariate traits, β is the solution vector for fixed effects of pond and stock, u is a vector of random additive genetic values for animals within pond and stock with 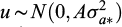 and
and 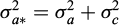 , where
, where 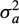 is the additive genetic variance,
is the additive genetic variance, 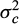 is the effects common to fullsibs other than additive genetics (i.e. non-additive genetic effect common to full-sibs and the environmental effect of hapa), and A is the additive genetic numerator relationship matrix among the recorded animals; X is an incidence matrix that assign each trait record to its appropriate level of the two fixed effects pond and stock; Z is an incidence matrix that assign each trait record to its animal; and e ∼ (0, V
is the effects common to fullsibs other than additive genetics (i.e. non-additive genetic effect common to full-sibs and the environmental effect of hapa), and A is the additive genetic numerator relationship matrix among the recorded animals; X is an incidence matrix that assign each trait record to its appropriate level of the two fixed effects pond and stock; Z is an incidence matrix that assign each trait record to its animal; and e ∼ (0, V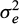 ) where
) where 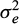 is the random residual (environmental) variance. The sires and dams of the full-sib families were considered as base population animals and were assumed to be unrelated. The effect of stock was not significant (P > 0.05).
is the random residual (environmental) variance. The sires and dams of the full-sib families were considered as base population animals and were assumed to be unrelated. The effect of stock was not significant (P > 0.05).
In the preliminary analysis, the significance of the fixed effect was tested based on Wald F statistics in the ASReml-R 4.0 software package (Gilmour et al., 2015).
From the estimated (co)variance component, the heritability for the studied traits was calculated as:
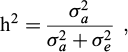
while the genetic (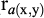 ) and residual (
) and residual (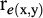 ) correlations between pairs (x, y) of the traits were calculated as:
) correlations between pairs (x, y) of the traits were calculated as:
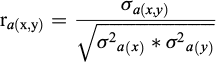
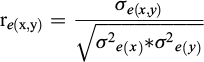
3 Results
3.1 Growth performance
The descriptive statistics for the traits viz., 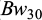 ,
, 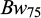 , and
, and 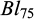 for each pond are provided in Table 2. At stocking at 30 days of age the mean body weight of the fish in Pond A (1.8 g) was higher compared to Pond B (1.2g)., while at 75 days of age the mean body weight of the fish in the two ponds was the same (5.3g). The coefficient of variation for body weights was very high in both ponds, but lower at 75 dph than at 30 dph. The survival from 30 to 75 dph was 61.2% in Pond A and 51.6% in Pond B. The survival from 75 to 150 dph was 52.6% in Pond A and 50.2% in Pond B. The families exhibited heterogeneity in survival at Bw150, ranging from 7.5% to 95% within an overall mean survival of 51.6% (SD: 21.2%).
for each pond are provided in Table 2. At stocking at 30 days of age the mean body weight of the fish in Pond A (1.8 g) was higher compared to Pond B (1.2g)., while at 75 days of age the mean body weight of the fish in the two ponds was the same (5.3g). The coefficient of variation for body weights was very high in both ponds, but lower at 75 dph than at 30 dph. The survival from 30 to 75 dph was 61.2% in Pond A and 51.6% in Pond B. The survival from 75 to 150 dph was 52.6% in Pond A and 50.2% in Pond B. The families exhibited heterogeneity in survival at Bw150, ranging from 7.5% to 95% within an overall mean survival of 51.6% (SD: 21.2%).
The descriptive statistics for traits recorded at 150 and 195 dph and growth rate indices for each pond provided in Table 3. The means for the different traits in the two ponds were very similar (28.1 g for Bw150 and 55 g for Bw195), and with a high coefficient of variation for the weight traits (49.6–58.3%), and as expected, a lower coefficient of variation for body length (16.2–18.5%). The mean weight gain between ponds was similar (25.6 g in pond A and 27.7 g in pond B). Further, the 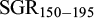 in the two ponds were very similar and with a much lower coefficient of variation than for WG150-195. The effect of pond was significant for all traits (P < 0.01), whereas the effect of stock was not significant (P > 0.05).
in the two ponds were very similar and with a much lower coefficient of variation than for WG150-195. The effect of pond was significant for all traits (P < 0.01), whereas the effect of stock was not significant (P > 0.05).
Descriptive statistics of body weight (Bw) and body length (Bl) of common carp in Pond A and B at 30 and 75 days post hatch.
Descriptive statistics of body weight (Bw) and body length (Bl) of common carp in Pond A and B at 150 and 195 days post hatch.
3.2 Estimates of heritability
The heritability estimates for the growth traits were relatively high both at 30 dph (0.68 ± 0.07) and 75 dph (0.52 ± 0.07) for body weight and 0.57 ± 0.07 for body length at 75 dph (Tab. 4). As only single pair matings were performed and with only one spawning and rearing hapa per family, the additive genetic effects, non-additive genetic effects, and hapa effect are confounded and likely to inflate the heritability estimate, particularly those at 30 and 75 dph. The heritability estimates for the growth traits at 150 dph and 195 dph, 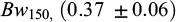 ,
, 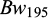 (0.39 ± 0.08),
(0.39 ± 0.08), 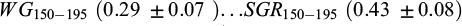 were medium to high (Tab. 5). With Bw150 as a covariate in the model, the single trait heritability estimates for WG and SGR were 0.29 ± 0.06 and 0.35 ± 0.07, respectively.
were medium to high (Tab. 5). With Bw150 as a covariate in the model, the single trait heritability estimates for WG and SGR were 0.29 ± 0.06 and 0.35 ± 0.07, respectively.
Estimates of heritability (on diagonal), genetic correlation (above diagonal) and phenotypic correlation (below diagonal) of body weight of common carp at 75 days post hatch and (Bw) and body length (Bl) at 75 days post hatch.
3.3 Estimates of genetic and residual correlations
The genetic correlation between body weight and body length at 75 (0.91) dph was high (Tab. 4). The genetic correlations (rg) and residual correlations (re) between the body weight traits and the two growth traits recorded at and between 150, 195 dph are presented in Table 5. The genetic correlation (rg) was very high between 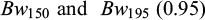 and high between Bw195 and WG150-195 (0.90), while the genetic correlation of Bw150 and Bw195 with SGR150-195 were negative and that between WG150-195 and SGR150-195 was zero. The residual correlations (re) between Bw150, Bw195, and WG150-195 were medium to high (≥0.71), while that between SGR150-195 and WG150-195 was medium, while those of SGR150-195 with Bw150 and Bw195 were close to zero (Tab. 5). At 150 and 195 days of age, the genetic correlation between body weight and body length was very high, 0.90 at 150 days and 0.92 at 195 days.
and high between Bw195 and WG150-195 (0.90), while the genetic correlation of Bw150 and Bw195 with SGR150-195 were negative and that between WG150-195 and SGR150-195 was zero. The residual correlations (re) between Bw150, Bw195, and WG150-195 were medium to high (≥0.71), while that between SGR150-195 and WG150-195 was medium, while those of SGR150-195 with Bw150 and Bw195 were close to zero (Tab. 5). At 150 and 195 days of age, the genetic correlation between body weight and body length was very high, 0.90 at 150 days and 0.92 at 195 days.
Estimates of heritability1 (on diagonal), genetic correlation (above diagonal) and residual correlation (below diagonal) of body weight of common carp at 150 and 195 days post hatch, and body weight gain (WG150-195) and specific growth rate (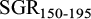 ) from 150 to 195 days.
) from 150 to 195 days.
4 Discussion
Until 75 dph the average daily gain (ADG) of the common carp reared in the inland saline water of 8-9 ppt in this study was 0.071 g. This is lower than the ADG of 0.098 g of common carp juveniles reared for 8 weeks in freshwater (Vandeputte et al., 2004). Until 195 dph the ADG was 0.289 g and thus similar to the ADG of 0.297 g of common carp reared in freshwater ponds for 8 months (Wang et al., 2006). Although the fish in these studies were reared under different environmental conditions, the results of this study indicate that common carp display good growth performance in earthen ponds with low-saline water. This is further supported by several reports, which suggest that freshwater fish can tolerate and grow well in low salinities (Chandra and Joshi, 2015; Ansal et al., 2016). In northern states like Haryana and Punjab, the breeding and cultivation of salinity-tolerant common carp offer an excellent aquaculture avenue and a means to address seed availability.
The high coefficient of variation in body weight at both 30 and 75 dph can be attributed to the young age and separate rearing of families in hapas compared to communal rearing. This finding aligns with a study by Ninh et al. (2011), which reported higher CV (42.6 to 61.0%) in separate early rearing compared to early communal rearing (15.8 to 57.5%). Similar coefficients of variation for body weight have been reported for other aquatic animal species, e.g., 48% to 60% in tilapia (Ponzoni et al., 2005), 39% to 47% in common carp (Wang et al., 2006), and 64% to 89% in seabass (Chandra et al., 2000). The decreasing CV with advancement of age in our study is similar to the reports in common carp by (Wang et al., 2006) and in Nile tilapia (Thodesen et al., 2013). As expected, the CV for body length traits was much lower, as length is a one-dimensional measure of body size, as compared to the three-dimensional measure of body weight.
The relative high heritability estimates for body weight at 30 (0.68) and 75 (0.52) dph are probably biased upwardly by non-additive genetic and hapa effects due to the single paired mating design and the rearing of each full-sib family in one hapa only until these ages, as also reported for separate and early rearing of families of common carp by Ninh et al. (2011) and for juvenile black bream (Doupe et al., 2003). At 150 and 195 dph, the magnitude of the heritability estimates for body weight was comparatively lower and in line with heritability estimates from other early growth studies in common carp (0.32 - 0.50) (Vandeputte et al., 2004; Nielsen et al., 2010; Spasić et al., 2010; Yousefian et al., 2011; Ninh et al., 2011). The medium magnitude of heritability for body weight at 195 dph, and its large phenotypic variation (CV ∼ 0.50), indicate a substantial genetic variation for early growth and thus a large potential for improving early growth of common carp through selective breeding. However, as BW195 was recorded after a very short (45 days) communal rearing period, the heritability estimate at 195 days of age is most likely biased upwards by a significant carryover effect of the separate rearing of the families until being tagged at 150 days of age, as also reported for other aquaculture species (Rezk et al., 2009; Ninh et al., 2011; Sae-Lim et al., 2013; Vandeputte and Haffray, 2014; Freitas et al., 2021). During this relatively short growth study period, body weight at 195 dph is strongly influenced by initial body weight, especially when the growth rate is just catching up. This was demonstrated by the high phenotypic correlations between BW150 and BW195 (0.94). This might also explain the similar magnitude of heritability estimates for BW195 (0.39 ± 0.08) and initial body weight BW150 (0.37 ± 0.06).
Similar to our study, high genetic and phenotypic correlations between body weight and length (0.90-0.92) have been reported in juvenile common carp (Vandeputte et al., 2004; Yousefian et al., 2011) and in other juveniles of cultured fishes, including, Grass carp (Fu et al., 2015), rainbow trout (Fishback et al., 2002), Atlantic salmon (Gunnes and Gjedrem, 1978), black bream (Doupe et al., 2003), and chinook salmon (Winkelman and Peterson, 1994). This suggests that length could serve as an indirect selection criterion for juvenile fish.
An attempt was made to account for the hapa effect by using two derived traits of growth, weight gain (WG150-195) and specific growth rate (SGR150-195). These traits showed negligible genetic correlation (approximately zero). In our study the use of SGR may fit well for young fish since their gain in weight remains in the exponential growth phase, this holds true for most juvenile fish over short culture intervals, but breaks down for larger fish or when culture durations are extended, even though its disadvantage of being only suited for comparing same age group fish, unsuitability for different life stages and dependency on initial weight (Hopkins, 1992; Aunsmo et al., 2014; Lugert et al., 2016). SGR expresses body weight gain on a logarithmic scale, minimizing variance heterogeneity between measurements taken at different ages (De Verdal et al., 2018).
In the present study, the heritability of SGR150-195 was found to be medium-high (0.43 ± 0.08). Furthermore, the single-trait heritability of SGR (0.35 ± 0.07), after including BW150 as a covariate in the model suggests a substantial genetic variation for juvenile growth rate. This is in line with previous studies showing medium high heritability for SGR during second overwintering (0.47 ± 0.11) and third growing season (0.49 ± 0.10) in common carp, and SGR of 0.48 ± 0.16 in juvenile red drum (Ma et al., 2008) and SGR of 0.27-0.39 in tilapia (Luan et al., 2010) and SGR (179 to 689 days age) of 0.20 ± 0.07 in gilthead seabream (Lee-Montero et al., 2015) and in contrast higher, than heritability of SGR (0.15 ± 0.07) reported by Aslam et al. (2020) in gilthead seabream.
The negative genetic correlations observed for SGR150–195 and body weight at 150 (BW150) and 195 (BW195) days suggest that using log-transformed body weights in the calculation of SGR150–195 may disproportionately accentuate common environmental (hapa) effects. Specifically, this transformation may penalize both fast- and slow-growing families too much, thereby reducing the apparent genetic association between SGR150–195 and its two body weight traits. However, the extent of this effect cannot be quantified, as the genetic family effect and the hapa effect during the period until tagging are completely confounded. However, Prchal et al. (2018), who reported a negative genetic correlation between body weight and SGR in the third growing season of common carp, suggested that smaller fish were performing better in terms of catching growth rate than their large counterparts. Furthermore, the low residual correlation between SGR and BW150 and BW195 indicates that these traits were largely independent after accounting for fixed and random effects. The plausible biological explanation for the negative genetic correlations observed between SGR150–195 and body weight at 150 (BW150) and 195 (BW195) is due to the hapa effect. There are family-level differences in survival at BW150 (7.5% to 95%). The different early rearing densities among families (individual hapas) due to mortalities may have contributed to differential growth constraints and subsequent compensatory growth upon release into the pond environment.
The limitation of the present study is that the data were generated from full-sib families in a single-pair mating design and reared in single non-replicated hapas, leading to upwardly biased heritability and genetic correlation estimates, which therefore should be interpreted with caution. In addition, these estimates are based on body weight recorded at a very young age and are therefore at a body weight far from the desired marketing size of about 1 kg of common carp in the saline production pond environment. Therefore, a new set of genetic parameters for the growth of common carp in this production environment needs to be obtained and should be based on both early and harvest body weights recorded on both full- and half-sibs tagged and pooled at an early age to ensure that the estimated parameters are less biased by the hapa effect. This information will be helpful to understand the extent to which growth traits at a younger age may be used as an indirect selection criterion for the growth of the breeding candidates until the desired marketing size e.g. by including also body weight at tagging as a selection criterion for the breeding objective trait harvest body weight. The relative weighting of these traits in the selection index depends on the production cost until tagging relative to the cost from tagging to harvest.
Acknowledgments
The study is funded by ICAR- NAHEP CAAST World Bank. The study was part of the first author's M.F.Sc research program and authors would like to acknowledge the Director/Vice-Chancellor, ICAR‒Central Institute of Fisheries Education, Mumbai, India for providing all infrastructure to carry out the study.
Funding
The work was financially supported by the National Agricultural Higher Education Project, the Centre for Advanced Agricultural Science and Technology, and the World Bank Funded Project.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Authors declare no competing interests.
Data availability statement
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
- Allan GL, Fielder DS, Fitzsimmons KM, et al. 36 - Inland saline aquaculture, in: G. Burnell, G. Allan (Eds.), New Technologies in Aquaculture, Woodhead Publishing, 2009, pp 1119–1147. [Google Scholar]
- Anand G, dar S, Srivas PP, Varghese T, Gupta S. 2022. Rearing in hypersaline inland ground saline water affect growth and osmoregulatory responses of Common carp (Linnaeus 1758). https://doi.org/10.21203/rs.3.rs-1164718/v2. [Google Scholar]
- Ansal MD, Dhawan A, Singh G, Kaur K. 2016. Species selection for enhancing productivity of freshwater carps in inland saline water of Punjab—a field study. Indian J Ecol 43: 45–49. [Google Scholar]
- Aslam ML, Carraro R, Sonesson AK, et al. 2020. Genetic variation, GWAS and accuracy of prediction for host resistance to sparicotyle chrysophrii in farmed gilthead sea bream (Sparus aurata). Front Genet 11. https://doi.org/10.3389/fgene.2020.594770. [Google Scholar]
- Aunsmo A, Krontveit R, Valle PS, Bohlin J. 2014. Field validation of growth models used in Atlantic salmon farming. Aquaculture 428: 249–257. [Google Scholar]
- Balon EK. 1995. Origin and domestication of the wild carp, Cyprinus carpio: from Roman gourmets to the swimming flowers. Aquaculture 129: 3–48. [Google Scholar]
- Belton B, Padiyar A, Ravibabu G, Gopal Rao K. 2017. Boom and bust in Andhra Pradesh: development and transformation in India's domestic aquaculture value chain. Aquaculture 470: 196–206. [Google Scholar]
- Chandra KP, Kailasam M, Thirunavukkarasu AR, Abraham M. 2000. Genetic parameters for early growth traits in Lates calcarifer (Bloch). J Mar Biol Assoc India 42: 194–199. [Google Scholar]
- Chandra S, Joshi Kd. 2015. Growth and survival of carps in salt-affected ponds under different treatments. J Inland Fish Soc India 47: 36–42. [Google Scholar]
- Chen L, Xu J, Sun X, Xu P. 2022. Research advances and future perspectives of genomics and genetic improvement in allotetraploid common carp. Rev Aquac 14: 957–978. [Google Scholar]
- Crandell PA, Gall GAE. 1993. The genetics of body weight and its effect on early maturity based on individually tagged rainbow trout (Oncorhynchus mykiss). Aquaculture 117: 77–93. [Google Scholar]
- CSSRI. 2013. Annual Report, 2013–14, Karnal: Central Soil Salinity Research Institute. [Google Scholar]
- De Verdal H, Komen H, Quillet E, et al. 2018. Improving feed efficiency in fish using selective breeding: a review. Rev Aquac 10: 833–851. [Google Scholar]
- Dey MM, Kumar P, Chen OL, Khan Md A, Barik NK, Li L, Nissapa A, Pham NS. 2013. Potential impact of genetically improved carp strains in Asia. Food Policy 43: 306–320. [Google Scholar]
- Dong Z, Nguyen NH, Zhu W. 2015. Genetic evaluation of a selective breeding program for common carp Cyprinus carpio conducted from 2004 to 2014. BMC Genetics 16:94. [Google Scholar]
- Doupe RG, Lymbery AJ. 2005a. Additive genetic and other sources of variation in growth traits of juvenile black bream Acanthopagrus butcheri. Aquac Res 36: 621–626. [Google Scholar]
- Doupe RG, Lymbery AJ. 2005b. Genetic covariation in production traits of sub-adult black bream Acanthopagrus butcheri after grow-out. Aquac Res 36: 1128–1132. [Google Scholar]
- Doupé RG, Lymbery AJ, Greeff J. 2003. Genetic variation in the growth traits of straight-bred and crossbred black bream (Acanthopagrus butcheri Munro) at 90 days of age: Genetic variation in black bream growth traits. Aquac Res 34: 1297–1301. [Google Scholar]
- El-Ibiary HM, Joyce JA. 1978. Heritability of body size traits, dressing weight and lipid content in channel catfish. J Anim Sci 47: 82–88. [Google Scholar]
- Fishback AG, Danzmann RG, Ferguson MM, Gibson JP. 2002. Estimates of genetic parameters and genotype by environment interactions for growth traits of rainbow trout (Oncorhynchus mykiss) as inferred using molecular pedigrees. Aquaculture 206: 137–150. [Google Scholar]
- FAO. 2021. Global map of salt-affected soils. https://scholar.google.com/scholar_lookup?title = Global%20Map%20of%20Salt% 20Affected%20Soils%20Version%201.0&publication_year = 2021&author = FAO (Accessed 29 Apr 2024) [Google Scholar]
- FAO. The state of world fisheries and aquaculture 2022, in: Towards Blue Transformation, FAO, Rome, 2022. https://doi.org/10.4060/cc0461en. [Google Scholar]
- Freitas MV, Lira LVG, Ariede RB, et al. 2021. Genotype by environment interaction and genetic parameters for growth traits in the Neotropical fish pacu (Piaractus mesopotamicus). Aquaculture 530: 735933. [Google Scholar]
- Fu J, Shen Y, Xu X, Li J. 2016. Genetic parameter estimates for growth of grass carp, Ctenopharyngodon idella, at 10 and 18 months of age. Aquaculture 450: 342–348. [CrossRef] [Google Scholar]
- Fu J, Shen Y, Xu X, Liu C, Li J. 2015. Genetic parameter estimates and genotype by environment interaction analyses for early growth traits in grass carp (Ctenopharyngodon idella). Aquacult Int 23: 1427–1441. [Google Scholar]
- Gilmour AR, Gogel BJ, Cullis BR, Welham SJ, Thompson R. Asreml user guide release 4.1 functional specification, VSN International Ltd, Hemel Hempstead, 2015. www.vsni.co.uk. Accessed 29 Apr 2024. [Google Scholar]
- Gjerde B, Simianer H, Refstie T. 1994. Estimates of genetic and phenotypic parameters for body weight, growth rate and sexual maturity in Atlantic salmon. Livestock Prod Sci 38: 133–143. [Google Scholar]
- Gunnes K, Gjedrem T. 1978. Selection experiments with salmon: IV. Growth of Atlantic salmon during two years in the sea. Aquaculture 15: 19–33. [Google Scholar]
- Hamilton MG, Mekkawy W, Alam MdB, Benzie JAH. 2022. Early selection to enhance genetic gain in a rohu (Labeo rohita) genetic improvement program. Aquaculture 553: 738058. [Google Scholar]
- Hopkins KD. 1992. Reporting fish growth: a review of the basics. J World Aquac Soc 23: 173–179. [Google Scholar]
- Hu X, Li C, Shang M, et al. 2017. Inheritance of growth traits in Songpu mirror carp (Cyprinus carpio L.) cultured in Northeast China. Aquaculture 477: 1–5. [Google Scholar]
- Hume DJ, Fletcher AR, Morison AK. 1983. Interspecific hybridization between carp (Cyprinus carpio L.) and golfish (Carassius auratus L.) from Victorian Waters. Mar Freshw Res 34: 915–919. [Google Scholar]
- Iffat J, Tiwari VK, Pavan-Kumar A, Verma AK, Harikrishna V, Babitha Rani AM, Chadha NK, Anand G. 2021. The effect of inland saline groundwater on growth, maturation, and osmoregulation of common carp. North Am J Aquac 83: 15–25. [Google Scholar]
- Kocour M, Mauger S, Rodina M, Gela D, Linhart O, Vandeputte M. 2007. Heritability estimates for processing and quality traits in common carp (Cyprinus carpio L.) using a molecular pedigree. Aquaculture 270: 43–50. [Google Scholar]
- Kumar P, Sharma PK. 2020. Soil salinity and food security in India. Front Sustain Food Syst 4. https://doi.org/10.3389/fsufs.2020.533781. [Google Scholar]
- Lalramnunsanga, Mishra A, Singh AL, Prakash S, Salvi A, Kumar AP, Pathan MA. 2024. Genetic diversity of common carp Cyprinus carpio in the base population of a selective breeding programme in India. Discov Anim 1: 3. [Google Scholar]
- Lee-Montero I, Navarro A, Negrín-Báez D, et al. 2015. Genetic parameters and genotype–environment interactions for skeleton deformities and growth traits at different ages on gilthead seabream (Sparus aurata L.) in four Spanish regions. Anim Genet 46: 164–174. [Google Scholar]
- Li Y, Zhang B, Yang Y, Chen S. 2019. Estimation of genetic parameters for juvenile growth performance traits in oliveflounder (Paralichthys olivaceus). Aquac Fish 4: 48–52. [Google Scholar]
- Liu Y-X, Wang G-X, Wang Y-F, Si, F Sun, Z-H, Zhang X-Y, Wang J-D, Liu H-J. 2011. Estimation of genetic parameters for growth traits of Japanese flounder Paralichthys olivaceus using an animal model. Fish Sci 77: 87–93. [Google Scholar]
- Luan TD, Olesen I, Kolstad K. Genetic parameters and genotype by environment interaction for growth of Nile tilapia in low and optimal temperature, in: Proceedings of the 9th World Congress on Genetics Applied to Livestock Production. Leipzig, Germany, 2010 [Google Scholar]
- Lugert V, Thaller G, Tetens J, et al. 2016. A review on fish growth calculation: multiple functions in fish production and their specific application. Rev Aquac 8: 30–42. [Google Scholar]
- Ma L, Saillant E, Gatlin DM, Gold JR. 2008. Estimates of heritability of larval and early juvenile growth traits in red drum (Sciaenops ocellatus). J Appl Aquac 20: 134–147. [Google Scholar]
- Mandal S, Raju R, Kumar A, Kumar P, Sharma PC. Current status of research, technology response and policy needs of salt-affected soils in India – a review, 2018 [Google Scholar]
- Mas-Muñoz J, Blonk R, Schrama JW, et al. 2013 Genotype by environment interaction for growth of sole (Solea solea) reared in an intensive aquaculture system and in a semi-natural environment. Aquaculture 410–411: 230–235. [Google Scholar]
- Nielsen HM, Ødegård J, Olesen I, Gjerde B, Ardo L, Jeney G, Jeney Z. 2010. Genetic analysis of common carp (Cyprinus carpio) strains: I: Genetic parameters and heterosis for growth traits and survival. Aquaculture 304: 14–21. [Google Scholar]
- Nilsson J. 1994. Genetics of growth of juvenile arctic char. Trans Am Fish Soc 123: 430–434. [Google Scholar]
- Ninh NH, Ponzoni RW, Nguyen NH, et al. 2011. A comparison of communal and separate rearing of families in selective breeding of common carp (Cyprinus carpio): estimation of genetic parameters. Aquaculture 322–323: 39–46. [Google Scholar]
- Ninh NH, Ponzoni RW, Nguyen NH, Woolliams JA, Taggart JB, McAndrew BJ, et al. 2013. A comparison of communal and separate rearing of families in selective breeding of common carp (Cyprinus carpio): responses to selection. Aquaculture 40: 152–159. [Google Scholar]
- Palaiokostas C, Kocour M, Prchal M, Houston RD. 2018. Accuracy of genomic evaluations of juvenile growth rate in common carp (Cyprinus carpio) using genotyping by sequencing. Front Genet 9. https://doi.org/10.3389/fgene.2018.00082. [Google Scholar]
- Phibi Philip Naduvathu. 2017. Performance Comparison of Cyprinus carpio (Linnaeus, 1758) Populations Reared in Inland Saline Waters at Varying Salinities. M.F.Sc. Dissertation, ICAR-Central Institute of Fisheries Education (Deemed to be University Established Under Section 3 of UGC Act 1956) Panch Marg, Off Yari Road, Versova, Andheri (W), Mumbai – 400 061. [Google Scholar]
- Ponzoni RW, Hamzah A, Tan S, Kamaruzzaman N. 2005. Genetic parameters and response to selection for live weight in the GIFT strain of Nile Tilapia (Oreochromis niloticus). Aquaculture 247: 203–210. [Google Scholar]
- Prchal M, Kause A, Vandeputte M, Gela D, Allamellou J-M, Kumar G, Bestin A, Bugeon J, Zhao J, Kocour M. 2018. The genetics of overwintering performance in two-year old common carp and its relation to performance until market size. PLOS ONE 13: e0191624. [Google Scholar]
- Rajanand S. 2016. A Study on Acclimatization and Survival Analysis of Cyprinus Carpio (Linnaeus,1758) Reared at Different Salinities, M.F.Sc. Dissertation, ICAR-Central Institute of Fisheries Education (University Established Under Section 3 of UGC Act 1956) Panch Marg, Off Yari Road, Versova, Andheri (W), Mumbai – 400 061 [Google Scholar]
- Rezk MA, Ponzoni RW, Khaw HL, et al. 2009 Selective breeding for increased body weight in a synthetic breed of Egyptian Nile tilapia, Oreochromis niloticus: response to selection and genetic parameters. Aquaculture 293: 187–194. [Google Scholar]
- Rossignoli CM, Obi C, Ali SA, et al. 2023. Production system and challenges of saline aquaculture in Punjab and Sindh provinces of Pakistan. Front Aquac 2: 1302571. [Google Scholar]
- Sae-Lim P, Komen H, Kause A, et al. 2013. Enhancing selective breeding for growth, slaughter traits and overall survival in rainbow trout (Oncorhynchus mykiss). Aquaculture 372–375: 89–96. [Google Scholar]
- Saillant E, Dupont-Nivet M, Haffray P, Chatain B. 2006. Estimates of heritability and genotype–environment interactions for body weight in sea bass (Dicentrarchus labrax L.) raised under communal rearing conditions. Aquaculture 254: 139–147. [Google Scholar]
- Saillant E, Ma L, Wang X, et al. 2007. Heritability of juvenile growth traits in red drum (Sciaenops ocellatus L.). Aquac Res 38: 781–788. [Google Scholar]
- Sharma DK, Chaudhari SK. 2012. Agronomic research in salt affected soils of India: an overview. Indian J Agron 57: 175–185. [Google Scholar]
- Singh G, Ansal MD, Kaur VI. 2018. Salinity tolerance and survival of freshwater carp, Cyprinus carpio Linn. in inland saline water. Indian J Ecol 45: 598–601. [Google Scholar]
- Spasić MM, Poleksić V, Stanković M, et al. 2010. Selective breeding programme of common carp (Cyprinus carpio L.) in Serbia: preliminary results. J Agric Sci (Belgrade) 55: 243–251. [Google Scholar]
- Tave D, Smitherman RO. 1980. Predicted response to selection for early growth in Tilapia nilotica. Trans Am Fish Soc 109: 439–445. [Google Scholar]
- Thodesen (Da-Yong Ma) J, Rye M, Wang Y-X, Li S-J, Bentsen HB, Gjedrem T. 2013. Genetic improvement of tilapias in China: Genetic parameters and selection responses in growth, pond survival and cold-water tolerance of blue tilapia (Oreochromis aureus) after four generations of multi-trait selection. Aquaculture 396–399: 32–42. [Google Scholar]
- Vandeputte M, Quillet E, Chevassus B. 2002. Early development and survival in brown trout (Salmo trutta fario L.): indirect effects of selection for growth rate and estimation of genetic parameters. Aquaculture 204: 435–445. [Google Scholar]
- Vandeputte M, Haffray P. 2014. Parentage assignment with genomic markers: a major advance for understanding and exploiting genetic variation of quantitative traits in farmed aquatic animals. Front Genet 5. https://doi.org/10.3389/fgene.2014.00432. [Google Scholar]
- Vandeputte M, Kocour M, Mauger S, Dupont-Nivet M, De Guerry D, Rodina M, Gela D, Vallod D, Chevassus B, Linhart O. 2004. Heritability estimates for growth-related traits using microsatellite parentage assignment in juvenile common carp (Cyprinus carpio L.). Aquaculture 235: 223–236. [Google Scholar]
- Vandeputte M, Kocour M, Mauger S, Rodina M, Launay A, Gela D, Dupont-Nivet M, Hulak M, Linhart O. 2008. Genetic variation for growth at one and two summers of age in the common carp (Cyprinus carpio L.): heritability estimates and response to selection. Aquaculture 277: 7–13. [Google Scholar]
- Wang C, Li S, Xiang S, Wang J, Liu Z, Pang Z, Duan J, Xu Z. 2006. Genetic parameter estimates for growth-related traits in Oujiang color common carp (Cyprinus carpio var. color). Aquaculture 259: 103–107. [Google Scholar]
- Wang J-Q, Lui H, Po H, Fan L. 1997. Influence of salinity on food consumption, growth and energy conversion efficiency of common carp (Cyprinus carpio) fingerlings. Aquaculture 148: 115–124. [Google Scholar]
- Wang X, Ross KE, Saillant E, et al. 2006. Quantitative genetics and heritability of growth-related traits in hybrid striped bass (Morone chrysops♀\times Morone saxatilis♂). Aquaculture 261: 535–545. [Google Scholar]
- Winkelman AM, Peterson RG. 1994. Heritabilities, dominance variation, common environmental effects and genotype by environment interactions for weight and length in chinook salmon. Aquaculture 125: 17–30. [Google Scholar]
- Wohlfarth GW. 1993. Heterosis for growth rate in common carp. Aquaculture 113: 31–46. [Google Scholar]
- Wohlfarth GW, Moav R. 1972. The regression of weight gain on initial weight in carp: I. Methods and results. Aquaculture 1: 7–28. [Google Scholar]
- Yousefian M, Sharifrohani M, Hosseinzadeh- Sahafi H, et al. 2011. Heritability estimation for growth-related traits in juvenile wild common carp (Cyprinus carpio L.) in the south of Caspian Sea. Iranian J Fish Sci 10: 740–748. [Google Scholar]
- Yu X, Adnoy T, Lv Z, et al. 2020. Phenotypic and genetic parameter estimation for growth traits in juvenile large yellow croaker (Larimichthys crocea). Fish Aquac J 11: 1–6. [Google Scholar]
Cite this article as: Raghul R, Pathan MA, Gjerde B, Singh AL, Misra A, PS N, Prakash S. 2026. Juvenile growth performance and associated genetic parameters in common carp, Cyprinus carpio cultured in 8 ppt inland saline groundwater, Aquat. Living Resour. 39: 9. https://doi.org/10.1051/alr/2026001
All Tables
Stocks of common carp used for the production of families and the number of families stocked into the separate rearing hapas in the two earthen ponds A and B.
Descriptive statistics of body weight (Bw) and body length (Bl) of common carp in Pond A and B at 30 and 75 days post hatch.
Descriptive statistics of body weight (Bw) and body length (Bl) of common carp in Pond A and B at 150 and 195 days post hatch.
Estimates of heritability (on diagonal), genetic correlation (above diagonal) and phenotypic correlation (below diagonal) of body weight of common carp at 75 days post hatch and (Bw) and body length (Bl) at 75 days post hatch.
Estimates of heritability1 (on diagonal), genetic correlation (above diagonal) and residual correlation (below diagonal) of body weight of common carp at 150 and 195 days post hatch, and body weight gain (WG150-195) and specific growth rate (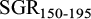 ) from 150 to 195 days.
) from 150 to 195 days.
All Figures
 |
Fig. 1 Spawning hapa for common carp. |
| In the text | |
 |
Fig. 2 Rearing hapa for common carp. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


