| Issue |
Aquat. Living Resour.
Volume 36, 2023
|
|
|---|---|---|
| Article Number | 30 | |
| Number of page(s) | 10 | |
| DOI | https://doi.org/10.1051/alr/2023026 | |
| Published online | 30 October 2023 | |
Research Article
Genetic parameters and response to selection for thermal tolerance, summer survival and growth in hybrid oyster (Crassostrea gigas ♀ × C. angulata ♂)
1
Key Laboratory of Mariculture, Ministry of Education, Ocean University of China, Qingdao 266003, China
2
Laboratory for Marine Fisheries Science and Food Production Processes, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266237, China
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Pierre Boudry
Received:
26
June
2023
Accepted:
23
September
2023
Abstract
The Pacific oyster Crassostrea gigas is the most commonly farmed mollusks worldwide, while its production has been hindered by massive mortalities linked to high temperatures. Selective breeding focusing on thermal tolerance is an attractive option for reducing the impact of massive mortalities, but the genetic basis underlying this trait is currently unknown. Hybridization between C. gigas and C. angulata was conducted and followed by one generation of family selection for thermal tolerance. The genetic parameters for thermal resistance, summer survival and growth and selection response were investigated by using 50 full-sibling families. The mean survival of all families ranged from 30.2 to 69.5%, reflecting the large variation of thermal tolerance in the hybrid population. The estimates of heritability for thermal tolerance were low to moderate, ranging from 0.19 ± 0.03 to 0.27 ± 0.05, confirmed that there is genetic basis for thermal tolerance. The phenotypic (r = 0.537, P < 0.01) and genetic correlations (r = 0.546, P < 0.01) between thermal tolerance and summer survival were positive and significant, while the phenotypic and genetic correlations between growth and survival (including thermal tolerance and summer survival) were positive but low (P > 0.05). High response to selection (ΔG = 36.33%) was observed after one generation of selection for thermal tolerance, and there was also corresponding response (ΔG = 14.46%) for summer survival. These results demonstrated that genetic selection to improve summer survival of oyster may be facilitated by selecting highly heat-tolerant lines.
Key words: Summer mortality / thermal tolerance / hybrid strain / genetic parameter / selection response
© G. Jiang et al., Published by EDP Sciences 2023
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
The Pacific oyster Crassostrea gigas is one of the most commonly farmed mollusks around the world. The global aquaculture production of C. gigas was 626 thousand tons in 2021 (FAO, 2023). C. gigas is also the representative indigenous aquaculture species of China, and it is mainly cultured in Shandong, Liaoning, Jiangsu, Hebei offshore, with an annual production of 1.93 million tons in 2022 (BOF, 2023). However, incidences of “Summer Mortality Syndrome (SMS)”, also known as “Pacific Oyster Mortality Syndrome (POMS)”, have become a global challenge for C. gigas aquaculture (Wendling and Wegner, 2013). SMS arises from a complex interaction between high-temperature stress, physiological stress related to gonad maturation, and pathogenic (such as ostreid herpesvirus 1 (OsHV-1) and bacteria) infections (Samain and McCombie, 2008). Such episodes have dramatically risen in northern China, mostly in Shandong and Liaoning Province since 1994, resulting in 50–80% mortality of adult C. gigas (Mao et al., 2005; Yang et al., 2021). Nevertheless, the epidemiological investigations of the case were unable to uncover evidence of OsHV-1 infection in C. gigas in China (Bai et al., 2015). Instead of OsHV-1, the outbreaks of SMS were related to severe physiological stress resulting from high temperatures, which are the major limiting factor for C. gigas farming in northern China (Lian et al., 2010; Yang et al., 2021; Li et al., 2023). Thus, genetic improvement program to breed a heat-tolerant strain is an attractive option for reducing the impact of SMS on oyster production.
Selective breeding is one of the most effective approaches for enhancing productive traits in aquaculture species (Gjedrem and Rye, 2018). Such a strategy has been commonly and successfully used in oysters to improve growth rate, survival rate, disease resistance, environmental tolerance, shell shape and color (Sheridan, 1997; Li et al., 2011; Vu et al., 2020; Chi et al., 2022; Jiang et al., 2023). Family selection has been reported somewhat successful in controlling several diseases of oysters, including OsHV-1 infection in C. gigas (Camara et al., 2017), JOD (juvenile oyster disease) and MSX (Haplosporidium nelsoni) disease in C. virginica (Farley et al., 1998; Calvo et al., 2003), and QX (Marteilia sydneyi) disease in Sacostrea glomerata (Nell and Perkins, 2006; Dove et al., 2013; Dove et al., 2020). Genetic selection is mainly effective due to the accumulation of relatively abundant additive genetic variances (Perry et al., 2005). New additive genetic variances can be introduced from intra- or interspecific hybridization in aquaculture species (Grant and Grant, 1994; Abbott et al., 2013). As a result, crosses between different populations or species are often used to develop foundation populations containing numerous variations (Bartley et al., 2001; Lu et al., 2017), and a new strain with superior traits can thus be bred through selection. For instance, a tilapia variety with improved growth rate and salinity tolerance was bred by selection from an interspecific hybrid of Oreochromis mossambicus and O. niloticus (de Verdal et al., 2014).
In order to achieve genetic improvement, traits must have genetic basis and allow to be measured accurately and reliably (Noble et al., 2020). Thus, thermal tolerance is generally measured as mortality during controlled thermal challenges in selective breeding programs (Perry et al., 2005; Doyle et al., 2011; Zhang et al., 2014). Moreover, the estimates of genetic parameters could provide a basic understanding of the influences of genetic factors on specific traits (Fu et al., 2015). To date, several selective breeding programs has been successively initiated in the United States (Beattie et al., 1980; Hershberger et al., 1984), China (Ding et al., 2020) and Mexico (Juárez et al., 2021) to tackle the problems of SMS related to high temperatures. Such programs reveal the potential for genetic improvement of summer survival by thermally selection in cultured C. gigas. However, little data is available concerning genetic parameters for thermal tolerance in oysters as well as estimates of the correlation between thermal tolerance and summer survival. Therefore, more examinations on the genetic basis for the heritability of thermal tolerance and the correlation between heat tolerance and summer survival are required.
In the present study, a selective breeding program has been implemented to address the problem of mortalities associated with high temperatures. It began with the F2 hybrid of C. gigas and C. angulata to introgress thermal tolerance traits of C. angulata and increase genetic variances. These two genetically closely related species can be clearly distinguished using mtDNA COI sequence differences at specific nucleotide positions (In et al., 2017). Fifty full-sibling families were produced using a nested half-sib mating design to estimate the genetic parameters for thermal resistance, summer survival and growth of the hybrid oysters. Furthermore, the responses to selection for these traits were also analyzed after one generation of selection. The aim of the present study was to determine whether summer survival of oysters could be improved indirectly through the selection for thermal tolerance.
2 Materials and methods
2.1 Broodstock selection
The base population originated from a F2 hybrid population of C. gigas and C. angulata. Briefly, C. gigas and C. angulata were collected from Rongcheng, Shandong province (37°11′N, 122°35′E) and Zhangzhou, Fujian province (24°28′N, 118°16′E), respectively, and used to produce the F1 hybrid (GA–C. gigas ♀ × C. angulata ♂) in 2019 (Jiang et al., 2021). In 2020, GA was used as the parental population to establish the F2 hybrid. In 2021, 1500 individuals of the F2 hybrids were collected from Rongcheng, and transferred to a hatchery in Laizhou, Shandong province (37°31′N, 119°90′ E). The top 138 (9.2%) out of 1500 individuals were selected as broodstock of the selection families based on thermal tolerance. Specifically, oysters were randomly distributed in four 500-L polyethylene tanks equipped with suspended mesh baskets (n = 375 individuals/basket). The seawater temperature was kept at a daily oscillating temperature of 25 to 34 °C for 25 days to prevent excessive thermal stress (Juárez et al., 2021). The daily temperature cycle consisted of two parts: a temperature-raising and a temperature-decreasing period. During the first period, the temperatures rose from 25 °C to 34 °C at a rate of 1 °C/h and remained at 34 °C for approximately three hours. The seawater cooled naturally to 25 °C and maintained at 25 °C for the latter period. Survivors of the thermal exposure were used as parents to construct the thermally selection families. In addition, a sub-sample (without heat shock challenge) of the F2 hybrids containing 50 oysters was collected from Rongcheng to produce the control families.
2.2 Family production and rearing
In June 2021, all survivors were strip spawned and crossed using a nested half-sib mating design (every male was mated with two different females) to produce 72 full-sibling selection families (including 36 half-sib families). Meanwhile, 16 full-sibling control families (including eight half-sib families) were produced with the same mating design. Artificial fertilization and rearing management of families were conducted as described by Li et al. (2011). Fertilized eggs from each family were incubated in a 100-L polyethylene bucket until the D-larvae stage when larvae were collected and relocated to additional polyethylene buckets. The rearing densities of larvae in each family were four to five individuals/mL and decreased with larval growth. The seawater temperature was kept at 21–23 °C and the salinity was 30–31 psu. Larvae were fed a daily ration of microalgae in accordance with their stage of development using Isochrysis galbana and Platymonas sp. When eye spots occurred, a bunch of scallop shells was put into rearing buckets as substrate. Finally, a total of 40 full-sibling selection families (including 11 half-sib families) and 10 full-sibling control families (including two half-sib families) were successfully obtained. The family structures of the selection and control families were given in the Supplementary Table 1. All families were transferred to an outdoor pond for temporary rearing.
2.3 Grow-out testing in field
In August 2021, the 50 families were relocated to the Sanggou Bay, a commercial mariculture area with a history of summer mortality problems in Rongcheng, Shandong province (Fig. 1A). Families were initially cultivated using the long-line method. On-site investigation showed that the deaths of C. gigas were mainly adult oysters (over one year old) (Yang et al., 2021). Thus, adult oysters were artificially detached from scallop shells and transferred to 10-layer lantern nets for field test on July 1st 2022. At the same time, the shell heights of 30 randomly selected oysters per family were recorded. To alleviate common full-sib effects, families were deployed in three replicates, with 20 oysters per replicate and a single family per layer. All families were submerged in lantern nets on floating rafts. Survival rates in summer field test were assessed on September 30th 2022. The seawater temperature in Shandong coastal area was downloaded from National Marine Data Center (http://mds.nmdis.org.cn/).
 |
Fig. 1 Family breeding sites (A) and annual seawater temperature (B) in coastal areas of Shandong Province, China in 2022. |
2.4 Thermal challenge testing
On July 1st 2022, 3000 adult oysters from 50 families (60 individuals/family) were randomly collected from Sanggou Bay and used to evaluate their thermal tolerance. Oysters allowed to acclimatize in 20-m3 concrete pond with filtered seawater (salinity 30 psu, temperature 18 °C) for 10 days prior to the thermal challenge. No dead oysters were observed during the acclimatization period. Sixty oysters per family were randomly divided into triplicate plastic baskets (20 individuals/basket) and secured to the bottom of three different polyethylene tank (∼ 500 L). Each polyethylene tank contained 10 families. The highest natural seawater temperature in the coastal areas of Shandong Province is about 26 °C (Fig. 1B), thus this temperature was used for thermal challenge. Desired temperature at which oysters were subjected to chronic thermal challenge was subsequently reached using a water bath unit with heaters at a rate of 0.1 °C/h. The control group was reared at 18 °C to confirm that mortality was due to thermal challenge. Oysters were fed with live Phaeodactylum tricornutum Bohlin three times a day. Thirty percent of the seawater was exchanged daily. Baskets were checked every six hours per day for mortality, and dead or moribund individuals were picked out. The thermal challenges were terminated when the daily mortality rate was below 1%. The experiment lasted for approximately 15 days in total.
2.5 Genetic analyses
2.5.1 Genetic parameters
The genetic parameters for summer survival and thermal tolerance were estimated by linear models and threshold models. Survival rates, both challenge test and field test, were assessed as a binary variable, based on whether the oyster were alive (score = 1) or dead (score = 0) at the end of the experiment. Survived oysters were assumed to be more resistant than those died. The growth trait (shell height) was also analyzed using linear animal model. The terms in the model were:
(1) Linear animal model (LAM)
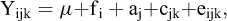
where Yijk is the observed value of the ith oyster, µ is the overall mean, fi is the fixed effect (“basket” effect in challenge test), aj is the random additive genetic effect of the jth oyster, a ∼ (0, A ), A is the relationship matrix; cjk is the random common environment effect, c ∼ (0, I
), A is the relationship matrix; cjk is the random common environment effect, c ∼ (0, I ), I is the identity matrix; eijk is the random residual effect, e ∼ (0, I
), I is the identity matrix; eijk is the random residual effect, e ∼ (0, I ).
).
(2) Threshold (logit) animal model (TAMl)

where all parameters are same as described in LAM above.
(3) Threshold (probit) animal model (TAMp)

where Φ represents the standard cumulative normal distribution function; all parameters are same as described in LAM above.
The variance components were estimated based on restricted maximum likelihood (REML) by using ASReml-R3.0 software package (Butler et al., 2009). The significance of fixed effect was tested using a Wald-F statistics. The significance of common environment effect was tested using likelihood ratio test (LRT test) (Lynch and Walsh, 1998). The narrow-sense heritability for traits was estimated from univariate analyses as follows:

where  is the additive genetic variance,
is the additive genetic variance,  is the common full-sib environmental variance (which were not included in the formula when the common environment effect was not significant),
is the common full-sib environmental variance (which were not included in the formula when the common environment effect was not significant),  is the residual variance (
is the residual variance ( = π2/3 and 1 in the TAMl and TAMp model, respectively).
= π2/3 and 1 in the TAMl and TAMp model, respectively).
The Z-score was used to assess whether the heritability was significantly different from zero (Nguyen et al., 2007):

where Xi and Xj are the estimates of heritability for the three traits; σi and σj are the respective standard error. Both of Xj and σj are defined as zero when genetic parameter is tested for its difference from zero.
The phenotype and genetic correlations between traits were estimated as the Pearson correlation between phenotype values and between estimated breeding values (EBVs), respectively, using the SPSS 26.0 software (Barros et al., 2018; Han et al., 2022).
2.5.2 Model comparison
To assess the consistency among genetic predictions of the three models, the Pearson and Spearman correlation coefficients among the full-sib family survival (thermal tolerance) EBVs from each model were calculated using the SPSS 26.0 software. The family EBVs were calculated as follows (Zhai et al., 2021):

where μs and μd are the sire and dam EBVs of each full-sib family, respectively.
2.5.3 Response to selection
Direct genetic gains for thermal tolerance, summer survival and shell height were calculated as the differences in EBVs between the selection families and control families (Nguyen et al., 2014; Vu et al., 2020). The model LAM was used to estimate EBVs with a minimum bias for the three traits. The mean EBVs were presented in actual units (millimeter for shell height and % for summer survival and thermal tolerance).
3 Results
3.1 Descriptive statistics
The mean monthly seawater temperature along the coast of Shandong Province in 2022 was over 22 °C during the summer period, especially above 25 °C in July and August (Fig. 1B). After summer, the survival rates of oysters varied greatly among the selection and control families, ranging from 35.0 to 88.3% (Tab. 1). The mean survival rates for the selection and control population were 66.9 ± 12.1% and 49.5 ± 11.1%, respectively. The coefficients of variation for the summer survival were slightly reduced in the selection population (18.1%) compared to the control population (22.4%).
In general, selection families showed the higher survival rate than control families in the thermal challenge testing (Fig. 2). The mean survival rate of selection population was 69.5 ± 10.5% (Tab. 1), with the mortality starting on day 2 and continuing to day 13 (Fig. 3). The mean survival rate of control population was 30.2 ± 15.6%, with the mortality starting on day 1 and continuing to day 14. The majority of mortality occurred from day 7 to 9 in selection population, while occurred on day 5 and 10 in control population. The coefficient of variation for thermal tolerance in control population (51.9%) was significantly higher than that in selection population (15.1%). There was no mortality in the control baskets for the challenge test, indicating that high temperature was a primary factor of mortality found during thermal challenge.
The family mean shell heights for the selection and control population were 86.42 ± 13.22 mm and 84.00 ± 11.48 mm, respectively (Tab. 1). The coefficients of variation for shell height ranged from 13.66 to 15.29%.
 |
Fig. 2 The mean survival rate of 50 full-sib families at day 15 after challenge with high temperature. |
Descriptive statistics of thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
 |
Fig. 3 Total number of dead oysters from all families (including the selection and control families) during the thermal challenge testing. |
3.2 Estimation of genetic parameters
The estimates of heritabilities for thermal tolerance were low to moderate, ranging from 0.19 ± 0.03 (TAMl model) to 0.27 ± 0.05 (LAM model) (Tab. 2). The common full-sib environmental effects (c2) estimated from the LAM model were low, but statistically higher than zero (P < 0.05, LRT test). The estimates of heritabilities for summer survival were 0.13 ± 0.03, 0.12 ± 0.02, and 0.15 ± 0.03, from the LAM, TAMl, and TAMp models, respectively (Tab. 2). Similarly, the c2 obtained from the LAM model was significantly different from zero (P < 0.05, LRT test). The estimate of heritability was moderate at 0.32 ± 0.07 for shell height (Tab. 2).
Interestingly, phenotypic (0.537) and genetic correlation (0.546) estimates between thermal tolerance and summer survival were positive and moderate (P < 0.01, Tab. 3). The shell height was positively correlated with thermal tolerance (0.227, P > 0.05) and summer survival (0.249, P > 0.05) at genetic level but weakly correlated with thermal tolerance (0.016, P > 0.05) and summer survival (0.053, P > 0.05) at phenotypic level.
Estimation of additive genetic variance ( ), common full-sib environmental variance (
), common full-sib environmental variance ( ), residual variance (
), residual variance ( ), heritabilities (h2) ± standard error (SE), and common full-sib environmental effect (c2) ± standard error (SE) for thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
), heritabilities (h2) ± standard error (SE), and common full-sib environmental effect (c2) ± standard error (SE) for thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
The genetic (above diagonal) and phenotypic (below diagonal) correlations between thermal tolerance (TT), summer survival (SS) and shell height (SH).
3.3 Model comparison
The Pearson and Spearman correlation coefficients among the three models were highly positive correlation (above 0.998, Tab. 4). The highest correlation was observed between TAMl and TAMp, with both correlation coefficients of 1. The above results indicated that a nearly identical family rankings were produced regardless of which model was used.
The Pearson rank correlation coefficients (above diagonal) and Spearman rank correlation coefficients (below diagonal) of the thermal tolerance EBVs between the linear animal model (LAM), threshold (logit) animal model (TAMl), and threshold (probit) animal model (TAMp).
3.4 Response to selection
Significantly, direct genetic gain in thermal tolerance (the only selection criterion) obtained from the first generation of oyster breeding program was 36.33% (Tab. 5). Selection for increased thermal tolerance also resulted in correlated increases in summer survival with a genetic gain of 14.46%. However, there was little genetic gain in shell height (2.15 mm).
Mean family EBVs and selection response (ΔG)* in thermal tolerance (TT), summer survival (SS) and shell height (SH).
4 Discussion
The present work is the first to develop a founder F2 hybrid between C. gigas (with favorable growth traits) and C. angulata (with favorable thermal tolerance traits) and to implement a family-based genetic selection for improving thermal resistance and passively summer survival of oysters. Results showed that the breeding program resulted in an increased survival in thermal challenge and in summer field.
4.1 Establishment of the base population
The first, and perhaps most important, step in initiating a selective breeding scheme is to establish a founder population with an abundant genetic variation (Thodesen et al., 2013). In practice, crossing with different populations/species is often initially utilized to construct base stocks with high genetic diversity. For example, a cross between Oreochromis niloticus and O. mossambicus was conducted and followed by backcrossing with O. mossambicus to obtain a base population used in a genetic selection aimed at improving growth and salt tolerance (de Verdal et al., 2014). The Pacific oyster C. gigas is a global aquaculture species because of its rapid growth rate, while its closely related species C. angulata is known to be more tolerant to warm seawater (Jiang et al., 2022). Because of segregation and recombination of gametes from crossbred parents, the F2 hybrids had abundant additive genes or gene combinations that should allow for effective selection of lines with desirable traits (Steffens et al., 1990; Argue et al., 2014). Therefore, the base population in this selection program was established using genetic materials from a F2 hybrid population derived from female C. gigas and male C. angulata. Thermal-tolerant individuals were screened from the base population using high-temperature treatments and used as parents for further genetic improvement.
4.2 Estimates of heritability of thermal tolerance, summer survival and growth
The economic importance of thermal tolerance in aquaculture species has given rise to many programs with the aim of genetic improvement. Thermal tolerance is a quantitative trait that displays continuous variation and has a pattern of inheritance involving many genes (Falconer and Mackay, 1996). Most researches estimating the heritability of thermal tolerance in aquatic animals have shown relatively low heritability. For instance, Zhang et al. (2014) reported the heritability of upper thermal tolerance was 0.087 in turbot Scophthalmus maximus. Baer and Travis (2000) estimated the heritability of thermal tolerance in poeciliid fish Heterandria formosa and reported a low heritability (less than 0.15). However, Perry et al. (2005) conducted an acute thermal challenge on rainbow trout Oncorhynchus mykiss and found a heritability of 0.407–0.477 for thermal tolerance. There are few reports concerning the genetic parameters for thermal tolerance in oyster. In this study, heritabilies for thermal tolerance for hybrid oysters were 0.19–0.27 estimated from three models, which was similar to the estimates (0.25) for C. angulata (Han et al., 2022). The moderate magnitude of estimated heritabilities suggested that thermal tolerance in oyster was partly under additive genetic control. In this scenario, family-based selection is a feasible approach for genetic improvement of thermal tolerance (Wang et al., 2019). However, these medium estimates of heritability were higher than the heritability estimates of 0.08 to 0.15 for C. gigas (Camara et al., 2017). A common factor that can influence heritability estimates is the effect of common environment (Falconer and Mackay, 1996). In this study, weak genetic links between families (13 half-sib families out of 50 full-sib families) may result in an inaccurate assessment of common environmental variance. Because common environmental effects may be absorbed in the additive genetic variance component, estimates of heritability are often overestimated (Setyawan et al., 2022).
The summer mortality has been reported for the Pacific oyster C. gigas in recent years and is a main concern for oyster production around the world (Huvet et al., 2004). Earlier studies of field-based data with outbreak of diseases, have suggested a genetic basis in summer survival (Hershberger et al., 1984; Dégremont et al., 2007). The heritability estimates for summer survival were low in all models (0.12–0.15) but significantly different from zero (P < 0.05) in this study. It is generally believed that survival is a trait highly correlated with individual fitness, and such traits usually show low heritabilities (Falconer and Mackay, 1996; Barros et al., 2018). Low heritability estimates for survival have also been reported in other aquatic animals, such as the abalone (0.04) (Jonasson et al., 1999), rainbow trout (0.08) (Vehviläinen et al., 2008), synthetic breed of Nile tilapia (0.12) (Rezk et al., 2009), intra-specific hybrid of shrimp (0.09) (Lu et al., 2017). Contrarily to the results of this study, a significant additive genetic basis was observed for summer survival as heritability estimates ranged from 0.47 to 1.08, with higher values in locations where mortality was higher (Dégremont et al., 2007). The observed differences could be related to the different magnitude of summer mortality between the two sites. In this study, oyster survival varied between 11.7 and 65.0% during summer, lower than the levels (2–82%) reported by Dégremont et al. (2007).
The estimated heritabilities of the summer survival were lower than that of the thermal tolerance. The low heritabilities of field survival have been also reported in other aquatic animals, including Atlantic salmon (Wetten et al., 2007), blue tilapia (Thodesen et al., 2013), Pacific oyster (Camara et al., 2017). Summer survival in oyster is a complex trait that is influenced by various internal and environmental factors (Huvet et al., 2004). Thus, the low heritabilities estimated for summer survival may be due to increased environmental “noise”, which resulted in a lower proportion of additive genetic variation in the total variation (Camara et al., 2017). Studies on a number of aquatic species have suggested that resistance to specific pathogens usually exhibits moderate to high levels of heritability (Liang et al., 2017; Zhai et al., 2021; Dégremont et al., 2015).
In aquaculture animals, linear animal model (LAM) and threshold animal model (TAM) are frequently adopted to fit binary traits, such as survival rate (Ødegård et al., 2010, 2011). Thus, thermal tolerance and summer survival were assessed in this study as binary traits at specific time using classic LAM, TAMl and TAMp models. Our results suggested slight differences in the estimated heritability obtained from the three models, which may be largely attributed to differences in trait definition and analysis (Wang et al., 2019). In terms of thermal tolerance, the TAM model seemed to slightly underestimate the heritability compared to the LAM model. Several studies have also reported biased estimates for the genetic component of binary data in TAM model (Ødegård et al., 2010; Liang et al., 2017). Nevertheless, the strong correlations among the family EBVs obtained from the three models may indicated similar predictive power of these models on family EBVs.
Genetic parameters for growth-related traits have been extensively assessed in oysters (Dégremont, 2007). However, very few researches have yielded heritability estimates for growth of hybrid oysters. The current study obtained a moderate estimate of heritability (h2 = 0.32) for growth of hybrid oysters, indicating a high potential for improving this trait through family-based selection. This finding parallels the finding in hybrid tilapia strain that heritability was moderate for growth-related traits (Barros et al., 2018).
4.3 Correlations between thermal tolerance, summer survival and growth traits
Basically, outbreaks of mass summer mortality in C. gigas are most prevalent throughout the world when seawater temperatures are high (Cheney et al., 2000; Dégremont et al., 2010; Chaney and Gracey, 2011). Therefore, a genetic selection project for more robust C. gigas resilience to high temperature was conducted on the west coast of USA, which resulted in significant increases in summer survival among the resulting offspring (Beattie et al., 1980). However, there is no conclusive evidence for a correlation between thermal tolerance and summer survival. In New Zealand, a study demonstrated that the genetic correlations (r = 0.17, P = 0.21) between heat shock survival and on-farm survival were low and not significant (Camara et al., 2017). In contrast, we found that the phenotypic (r = 0.537, P < 0.01) and genetic correlations (r = 0.546, P < 0.01) between thermal tolerance and summer survival were positive and moderate. This discrepancy may be attributed to differences in the main causes of summer mortality at the two sites. It has been reported that OsHV-1 is the main cause of massive summer mortality in farmed C. gigas in New Zealang (Keeling et al., 2014; de Kantzow et al., 2019). However, the epidemiological investigation did not show evidence of OsHV-1 infection in C. gigas from China (Bai et al., 2015). As this study showed, the natural water temperature in the breeding areas of C. gigas in Shandong Province is usually above 25 °C throughout the summer. High temperatures significantly affect the immune system of oysters and lead to oxidative stress (Li et al., 2023), thus being a key environmental factor contributing to mortality outbreaks in China (Lian et al., 2010; Yang et al., 2021). Encouragingly, the positive phenotypic and genetic correlations between thermal tolerance and summer survival implied that thermally selection could be used as a mean to reduce summer mortality.
The main objective of genetic improvement program for aquaculture species is usually to obtain greater economic benefits. Thus, there is little commercial value in survival selection without considering growth rate. Correlations between different traits are caused by pleiotropy and linkage disequilibrium of genes (Falconer and Mackay, 1996). However, the associations between morphological and fitness-related (such as environmental tolerance and disease resistance) traits in aquatic animals are often variable (Perry et al., 2005). In this study, the phenotypic and genetic correlations between growth and survival (including thermal tolerance and summer survival) were positive but low (P > 0.05). Similar to the results presented here, low and non-significant positive phenotype (0.05–0.11) and genetic correlations (0.04–0.13) between thermal tolerance and shell height were also observed in C. angulata (Han et al., 2022), indicating selection for thermal tolerance will not result in any undesirable changes in shell height. In addition, Dégremont et al. (2007) also reported a low but positive genetic correlation between growth and summer survival in C. gigas. In this case, future attempts at genetic improvement of oysters should collectively consider improving both survival and growth traits.
4.4 Response to selection
A main finding of our study is the positive response to selection (ΔG = 36.33%) after one generation of family-based selection to increase the tolerance to high temperature of hybrid oysters. Response to selection for thermal tolerance of shellfish has not been recorded in the literature; however, Hershberger et al. (1984) observed that survival of thermally selected C. gigas was 43% higher after three generations of family-based selection, compared to that of the controls. Estimated selection response of thermal tolerance was consistent with phenotypic values in this genetic selection program, reflecting the large genetic variation of thermal tolerance in the hybrid oyster population. Also, the positive genetic gain suggested that selection for thermal tolerance was following the intended direction. Other programs selecting the tolerance to high-temperature have also showed improved survival of the selected lines over the controls, as reported in rainbow trout O. mykiss strain (Ineno et al., 2005, 2018), and in bluegill Lepomis macrochirus strain (Holland et al., 1974). Notably, this thermally selection automatically improved the summer survival, which was 17.4% in our study. The genetic gain (ΔG = 14.46%) of summer survival for hybrid oysters fell within the range of estimates reported for C. gigas (9.9–45.5%) (Dégremont et al., 2010; Chi et al., 2022), demonstrating that thermally selection could be used as an indirect means of improving summer survival in oysters.
5 Conclusions
This is the first report of genetic parameters for thermal tolerance, summer survival, and shell height in the hybrid oyster C. gigas ♀ × C. angulata ♂. The results of the present study suggest that thermal tolerance is a moderately heritable trait and suitable for family-based selective breeding. The phenotypic and genetic correlations between thermal tolerance and summer survival were positive and moderate, and the phenotypic and genetic correlations between survival and growth were positive and low. Significant response to selection (ΔG = 36.33%) was achieved for thermal tolerance, and there was also corresponding response (ΔG = 14.46%) for summer survival. These results suggested that genetic improvement to improve summer survival of oyster could be achieved by selecting highly heat-tolerant lines without resulting in any adverse changes in growth.
Author contribution statement
Gaowei Jiang: Conceptualisation, Data curation, Formal analysis, Methodology, Writing − Original draft. Chengxun Xu: Supervision, Resources. Qi Li: Supervision, Conceptualization, Resources, Writing − Review and editing, Funding acquisition.
Supplementary Material
Table S1. Family structures of the selection and control families used in this experiment. Access Supplementary Material
Acknowledgements
This work was funded by grants from the National Key R&D Program of China (2022YFD2400305), Earmarked Fund for Agriculture Seed Improvement Project of Shandong Province (2021ZLGX03, 2022LZGCQY010 and 2020LZGC016), and China Agriculture Research System Project (CARS-49).
References
- Abbott R, Albach D, Ansell S, et al. 2013. Hybridization and speciation. J Evol Biol 26: 229–246. [CrossRef] [PubMed] [Google Scholar]
- Argue BJ, Kuhlers D, Liu ZJ, Dunham RA. 2014. Growth of channel catfish (Ictalurus punctatus), blue catfish (I. furcatus), and their F1, F2, F3, and F1 reciprocal backcross hybrids in earthen ponds. J Anim Sci 92: 4297–4305. [CrossRef] [PubMed] [Google Scholar]
- Baer CF, Travis J. 2000. Direct and correlated responses to artificial selection on acute thermal stress tolerance in a livebearing fish. Evolution 54: 238–244. [Google Scholar]
- Bai C, Wang C, Xia J, Sun H, Zhang S, Huang J. 2015. Emerging and endemic types of Ostreid herpesvirus 1 were detected in bivalves in China. J Invertebr Pathol 124: 98–106. [CrossRef] [PubMed] [Google Scholar]
- Barros J, Winkler FM, Velasco LA. 2018. Heritability, genetic correlations and genotype-environment interactions for growth and survival of larvae and post-larvae of the Caribbean scallop, Argopecten nucleus (Mollusca: Bivalvia). Aquaculture 495: 948–954. [Google Scholar]
- Bartley DM, Rana K, Immink AJ. 2001. The use of inter-specific hybrids in aquaculture and fisheries. Rev Fish Biol Fish 10: 325–337. [Google Scholar]
- Beattie J, Chew K, Hershberger W. 1980. Differential survival of selected strains of Pacific Oysters (Crassostrea gigas) during summer mortality. Proc Natl Shellfish Ass 70: 184–189. [Google Scholar]
- BOF, Bureau of Fisheries, 2023. China Fisheries Statistic Yearbook 2023, Agriculture Press, Beijing, China ( in Chinese). [Google Scholar]
- Butler DG, Cullis BR, Gilmour AR, Gogel BJ. 2009. ASReml-R Reference Manual. Queensland Department of Primary Industries and Fisheries, NSW Department of Primary Industries, Brisbane. [Google Scholar]
- Calvo LM, Calvo GW, Burreson E. 2003. Dual resistance in a selectively bred eastern oyster, Crassostrea virginica, strain tested in Chesapeake Bay. Aquaculture 200: 69–87. [CrossRef] [Google Scholar]
- Camara MD, Yen S, Kaspar HF, Kesarcodi-Watson A, King N, Jeffs AG, Tremblay LA. 2017. Assessment of heat shock and laboratory virus challenges to selectively breed for ostreid herpesvirus 1 (OsHV-1) resistance in the Pacific oyster, Crassostrea gigas. Aquaculture 469: 50–58. [CrossRef] [Google Scholar]
- Chaney ML, Gracey AY. 2011. Mass mortality in Pacific oysters is associated with specific gene expression signature. Mol Ecol 20: 2942–2954. [CrossRef] [PubMed] [Google Scholar]
- Cheney DP, MacDonald BF, Elston RA. 2000. Summer mortality of Pacific oysters, Crassostrea gigas (Thunberg): Initial findings on multiple environmental stressors in Puget Sound, Washington, 1998. J Shellfish Res 19: 353–359. [Google Scholar]
- Chi Y, Jiang G, Liang Y, Xu C, Li Q. 2022. Selective breeding for summer survival in Pacific oyster (Crassostrea gigas): Genetic parameters and response to selection. Aquaculture 556: 738271. [CrossRef] [Google Scholar]
- de Verdal H, Rosario W, Vandeputte M, Muyalde N, Morissens P, Baroiller JF, Chevassus B. 2014. Response to selection for growth in an interspecific hybrid between Oreochromis mossambicus and O. niloticus in two distinct environments. Aquaculture 430: 159–165. [CrossRef] [Google Scholar]
- de Kantzow MC, Whittington RJ, Hick P. 2019. Prior exposure to Ostreid herpesvirus 1 (OsHV-1) at 18 °C is associated with improved survival of juvenile Pacific oysters (Crassostrea gigas) following challenge at 22 °C. Aquaculture 507: 443–450. [CrossRef] [Google Scholar]
- Dégremont L, Ernaude B, Bédier E, Boudry P. 2007. Summer mortality of hatchery-reared Pacific oyster (Crassostrea gigas). I. estimation of genetic parameters for survival and growth. Aquaculture 262: 41–53. [CrossRef] [Google Scholar]
- Dégremont L, Bédier E, Boudry P. 2010. Summer mortality of hatchery-produced Pacific oyster spat (Crassostrea gigas). II. response to selection for survival and its infiuence on growth and yield. Aquaculture 299: 21–29. [CrossRef] [Google Scholar]
- Dégremont L, Norrry M, Maurouard E. 2015. Mass selection for survival and resistance to OsHV-1 infection in Crassostrea gigas spat in field conditions: response to selection after four generations. Aquaculture 446: 111–121. [CrossRef] [Google Scholar]
- Ding F, Li A, Cong R, Wang X, Wang W, Que H, Zhang G, Li L. 2020. The phenotypic and the genetic response to the extreme high temperature provides new insight into thermal tolerance for the pacific oyster Crassostrea gigas. Front Mar Sci 7: 399. [CrossRef] [Google Scholar]
- Dove M, Nell J, O'Connor W. 2013. Evaluation of the progeny of the fourth-generation Sydney rock oyster Saccostrea glomerata (Gould, 1850) breeding lines for resistance to QX disease (Marteilia sydneyi) and winter mortality (Bonamia roughleyi). Aquac Res 44: 1791–1800. [Google Scholar]
- Dove M, Kube P, Wilkie E, Cumbo V, Raftos D, O'Connor W. 2020. New Soth Wales Department of Primery Industries (Fisheries). Accelerated Sydney rock oyster (SRO) breeding research. in: Future Oysters CRC -P Project 2016–802. [Google Scholar]
- Doyle CM, Leberg PL, Klerks PL. 2011. Heritability of heat tolerance in a small livebearing fish, Heterandria Formosa. Ecotoxicology 20: 535–542. [CrossRef] [PubMed] [Google Scholar]
- Falconer DS, Mackay TFC. (1996). Introduction to Quantitative Genetics, 4th edn, Pearson Education Ltd., Essex, England. [Google Scholar]
- FAO 2023. Global Aquaculture Production. Fisheries and Aquaculture Division (online). Rome. https://www.fao.org/fishery/en/collection/aquaculture?lang=en. [Google Scholar]
- Farley CA, Lewis EJ, Relyea D, Zahtila J, Rivara G. 1998. Resistance studies for juvenile oyster disease (JOD) 1997: some early insights into genetic aspects. J Shellfish Res 17: 352–353. [Google Scholar]
- Fu J, Shen Y, Xu X, Li J. 2015. Genetic parameter estimates for growth of grass carp, Ctenopharyngodon idella, at 10 and 18 months of age. Aquaculture 450: 342–348. [Google Scholar]
- Gjedrem T, Rye M. 2018. Selection response in fish and shellfish: a review. Rev Aquac 10: 168–179. [CrossRef] [Google Scholar]
- Grant PR, Grant BR. 1994. Phenotypic and genetic effects of hybridization in Darwin's finches. Evolution 48: 297–316. [CrossRef] [Google Scholar]
- Han Z, Guo X, Lu Z, Song Y, Chen R, Han X, Yu S, Tu K, Liu L, Que H. 2022. Heritability estimates for gonadal development traits and their genetic correlations with growth and heat tolerance traits in the Fujian Oyster Crassostrea angulata. Front Mar Sci 9: 986441. [CrossRef] [Google Scholar]
- Hershberger WK, Perdue JA, Beattie JH. 1984. Genetic selection and systematic breeding in Pacific oyster culture. Aquaculture 39: 237–245. [CrossRef] [Google Scholar]
- Holland WE, Smith MH, Gibbons J, Brown DH. 1974. Thermal tolerances of fish from a reservoir receiving effluent from a nuclear reactor. Physiol Zool 47: 110–118. [CrossRef] [Google Scholar]
- Hopkins SR, Dean JM. 1975. The response of developmental stage of Fundulus to acute thermal shock. in: Vernberg FJ (Ed.), Physiological Ecology of Estuarine Organisms, University South Carolina Press, Columbia. [Google Scholar]
- Huvet A, Herpin A, Dégremont L, Labreuche Y, Samain JF, Cunningham C. 2004. The identification of genes from the oyster Crassostrea gigas that are differentially expressed in progeny exhibiting opposed susceptibility to summer mortality. Gene 343: 211–220. [CrossRef] [PubMed] [Google Scholar]
- In VV, O'Connor W, Sang VV, Van PT, Knibb W. 2017. Resolution of the controversial relationship between Pacific and Portuguese oysters internationally and in Vietnam. Aquaculture 473: 389–399. [CrossRef] [Google Scholar]
- Ineno T, Tsuchida S, Kanda M, Watabe S. 2005. Thermal tolerance of a rainbow trout Oncorhynchus mykiss strain selected by high-temperature breeding. Fish Sci 71: 767–775. [CrossRef] [Google Scholar]
- Ineno T, Tamaki K, Yamada K, Kodama R, Tsuchida S, Tan E, Watabe S. 2018. Thermal tolerance of a thermally selected strain of rainbow trout Oncorhynchus mykiss and the pedigrees of its F1 and F2 generations indicated by their critical thermal maxima. Fish Sci 84: 671–679. [CrossRef] [Google Scholar]
- Jiang G, Li Q, Xu C, Liu S, Kong L, Yu H. 2021. Reciprocal hybrids derived from Crassostrea gigas and C. angulata exhibit high heterosis in growth, survival and thermotolerance in northern China. Aquaculture 545: 737173. [CrossRef] [Google Scholar]
- Jiang G, Zhou J, Cheng G, Meng L, Chi Y, Xu C, Li Q. 2022. Examination of survival, physiological parameters and immune response in relation to the thermo-resistant heterosis of hybrid oysters derived from Crassostrea gigas and C. angulata. Aquaculture 559: 738454. [CrossRef] [Google Scholar]
- Jiang G, Chi Y, Zhou J, Cheng G, Du L, Xu C, Li Q. 2023. Phenotypic evaluation of two genetically improved strains selected from the reciprocal hybrids of Crassostrea gigas and C. angulata. Aquaculture 576: 739808. [CrossRef] [Google Scholar]
- Jonasson J, Stefansson SE, Gudnasonm A, Steinarsson A. 1999. Genetic variation for survival and shell length of cultured red abalone (Haliotis rufescens) in Iceland. J Shellfish Res 18: 621–625. [Google Scholar]
- Juárez OE, Escodedo-Fregoso C, Arredondo-Espinoza R, Ibarra AM. 2021. Development of SNP markers for identification of thermo-resistant families of the Pacific oyster Crassostrea gigas based on RNA- s eq. Aquaculture 539: 736618. [CrossRef] [Google Scholar]
- Keeling S, Brosnahan C, Williams R, Gias E, Hannah M, Bueno R, McDonald W, Johnston C. 2014. New Zealand juvenile oyster mortality associated with ostreid herpesvirus 1 − an opportunistic longitudinal study. Dis Aquat Org 109: 231–239. [CrossRef] [PubMed] [Google Scholar]
- Li Q, Wang Q, Liu S, Kong L. 2011. Selection response and realized heritability for growth in three stocks of the Pacific oyster Crassostrea gigas. Fish Sci 77: 643–648. [CrossRef] [Google Scholar]
- Li X, Shi C, Yang B, Li Q, Liu S. 2023. High temperature aggravates mortalities of the Pacific oyster (Crassostrea gigas) infected with Vibrio: a perspective from homeostasis of digestive microbiota and immune response. Aquaculture 568: 739309. [CrossRef] [Google Scholar]
- Lian W, Weng H, Mao Y, Fang J. 2010. Study on the relationship between Pacific oyster Crassostrea gigas summer mortality with culture environment and organism condition. Prog Fish Sci 31: 92–100 (in Chinese). [Google Scholar]
- Liang B, Jiang F, Zhang S, Yue X, Wang H, Liu B. 2017. Genetic variation in vibrio resistance in the clam Meretrix petechialis under the challenge of Vibrio parahaemolyticus. Aquaculture 468: 458–463. [CrossRef] [Google Scholar]
- Lu X, Luan S, Cao B, Sui J, Dai P, Meng X, Luo K, Kong J. 2017. Heterosis and heritability estimates for the survival of the Pacific white shrimp (Litopenaeus vannamei) under the commercial scale ponds. Acta Oceanol Sin 36: 62–68. [CrossRef] [Google Scholar]
- Lynch M, Walsh JB. 1998. Genetics and Analysis of Quantitative Traits, Sinauer Assocs., Inc., Sunderland, MA. [Google Scholar]
- Mao Y, Zhou Y, Yang H, Yuan X, Wen H, Wang R. 2005. Seasonal variation in metabolic rate of Pacific oyster, Crassostrea gigas and its implication to summer mortality. Oceanol Limnol Sin 36: 445–451 (in Chinese). [Google Scholar]
- Nell JA, Perkins B. 2006. Evaluation of the progeny of third-generation Sydney rock oyster Saccostrea glomerata (Gould, 1850) breeding lines for resistance to QX disease marteilia sydneyi and winter mortality Bonamia roughleyi. Aquac Res 37: 693–700. [CrossRef] [Google Scholar]
- Nguyen NH, Khaw HL, Ponzoni RW, Hamzah A, Kamaruzzaman N. 2007. Can sexual dimorphism and body shape be altered in Nile tilapia (Oreochromis niloticus) by genetic means? Aquaculture 272: S38– S46. [Google Scholar]
- Nguyen TTT, Hayes B, Ingram BA. 2014. Genetic parameters and response to selection in blue mussel (Mytilus galloprovincialis) using a SNP-based pedigree. Aquaculture 420-421: 295–301. [Google Scholar]
- Noble TH, Coman GJ, Wade NM, Thomson PC, Raadsma HW, Khatkar MS, Guppy JL, Jerry DR. 2020. Genetic parameters for tolerance to gill-associated virus under challenge-test conditions in the black tiger shrimp (Penaeus monodon). Aquaculture 516: 734428. [CrossRef] [Google Scholar]
- Ødegård J, Meuwissen TH, Heringstad B, Madsen P. 2010. A simple algorithm to estimate genetic variance in an animal threshold model using Bayesian inference. Genet Sel Evol 42: 29. [CrossRef] [PubMed] [Google Scholar]
- Ødegård J, Gitterle T, Madsen P, Meuwissen TH, Yazdi MH, Gjerde B, Pulgarin C, Rye M. 2011. Quantitative genetics of taura syndrome resistance in pacific white shrimp (Penaeus vannamei): a cure model approach. Genet Sel Evol 43: 14. [CrossRef] [PubMed] [Google Scholar]
- Perry GML, Martyniuk CM, Ferguson MM, Danzmann RG. 2005. Genetic parameters for upper thermal tolerance and growth-related traits in rainbow trout (Oncorhynchus mykiss). Aquaculture 250: 120–128. [CrossRef] [Google Scholar]
- Rezk MA, Ponzoni RW, Khaw HL, Kamel E, Dawood T, John G. 2009. Selective breeding for increased body weight in a synthetic breed of Egyptian Nile tilapia, Oreochromis niloticus: response to selection and genetic parameters. Aquaculture 293: 187–194. [Google Scholar]
- Samain JF, McCombie H. 2008. Summer mortality of Pacific oyster Crassostrea gigas, Ed. Quae. pp 379. [Google Scholar]
- Setyawan P, Aththar MHF, Imron I, Gunadi B, Haryadi J, Bastiaansen JWM, Camara MD, Komen H. 2022. Genetic parameters and genotype by environment interaction in a unique Indonesian hybrid tilapia strain selected for production in brackish water pond culture. Aquaculture 561: 738626. [CrossRef] [Google Scholar]
- Sheridan AK. 1997. Genetic improvement of oyster production-a critique. Aquaculture 153: 165–179. [CrossRef] [Google Scholar]
- Steffens W, Jachinen H, Fredrich F. 1990. Possibilities of sturgeon culture in central Europe. Aquaculture 89: 101–122. [CrossRef] [Google Scholar]
- Thodesen J, Rye M, Wang Y, Li S, Bentsen HB, Gjedrem T. 2013. Genetic improvement of tilapias in China: Genetic parameters and selection responses in growth, pond survival and cold-water tolerance of blue tilapia (Oreochromis aureus) after four generations of multi-trait selection. Aquaculture 396–399: 32–42. [Google Scholar]
- Vehviläinen H, Kause A, Quinton C, Koskinen H, Paananen T. 2008. Survival of the currently fittest: genetics of Rainbow trout survival across time and space. Genetics 180: 507–516. [CrossRef] [PubMed] [Google Scholar]
- Vu SV, Knibb W, Nguyen NTH, Vu IV, O'Connor W, Dove M, Nguyen NH. 2020. First breeding program of the Portuguese oyster Crassostrea angulata demonstrated significant selection response in traits of economic importance. Aquaculture 518: 734664. [CrossRef] [Google Scholar]
- Wang X, Ma A, Huang Z, Sun Z, Cui W, Qu J, Yu H. 2019. Estimation of genetic parameters for upper thermal tolerances and growth-related traits in turbot Scophthalmus maximus. J Oceanol Limnol 37: 1736–1745. [CrossRef] [Google Scholar]
- Wendling CC, Wegner KM. 2013. Relative contribution of reproductive investment, thermal stress and Vibrio infection to summer mortality phenomena in Pacific oysters. Aquaculture 412–413: 88–96. [Google Scholar]
- Wetten M, Aasmundstad T, Kjøglum S, Storset A. 2007. Genetic analysis of resistance to infectious pancreatic necrosis in Atlantic salmon (Salmo salar L.). Aquaculture 272: 111–117. [CrossRef] [Google Scholar]
- Yang B, Zhai S, Li X, Tian J, Li Q, Shan H, Liu S. 2021. Identification of vibrio alginolyticus as a causative pathogen associated with mass summer mortality of the Pacific oyster (Crassostrea gigas) in China. Aquaculture 535: 736363. [CrossRef] [Google Scholar]
- Zhai S, Yang B, Zhang F, Li Q, Liu S. 2021. Estimation of genetic parameters for resistance to Vibrio alginolyticus infection in the Pacific oyster (Crassostrea gigas). Aquaculture 538: 736545. [CrossRef] [Google Scholar]
- Zhang T, Kong J, Liu B, Wang Q, Cao B, Luan S, Wang W. 2014. Genetic parameter estimation for juvenile growth and upper thermal tolerance in turbot (Scophthalmus maximus Linnaeus). Acta Oceanol Sin 33: 106–110. [CrossRef] [Google Scholar]
Cite this article as: Jiang G, Xu C, Li Q. 2023. Genetic parameters and response to selection for thermal tolerance, summer survival and growth in hybrid oyster (Crassostrea gigas ♀ × C. angulata ♂). Aquat. Living Resour. 36: 30
All Tables
Descriptive statistics of thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
Estimation of additive genetic variance ( ), common full-sib environmental variance (
), common full-sib environmental variance ( ), residual variance (
), residual variance ( ), heritabilities (h2) ± standard error (SE), and common full-sib environmental effect (c2) ± standard error (SE) for thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
), heritabilities (h2) ± standard error (SE), and common full-sib environmental effect (c2) ± standard error (SE) for thermal tolerance (TT), summer survival (SS) and shell height (SH) in C. gigas ♀ × C. angulata ♂.
The genetic (above diagonal) and phenotypic (below diagonal) correlations between thermal tolerance (TT), summer survival (SS) and shell height (SH).
The Pearson rank correlation coefficients (above diagonal) and Spearman rank correlation coefficients (below diagonal) of the thermal tolerance EBVs between the linear animal model (LAM), threshold (logit) animal model (TAMl), and threshold (probit) animal model (TAMp).
Mean family EBVs and selection response (ΔG)* in thermal tolerance (TT), summer survival (SS) and shell height (SH).
All Figures
 |
Fig. 1 Family breeding sites (A) and annual seawater temperature (B) in coastal areas of Shandong Province, China in 2022. |
| In the text | |
 |
Fig. 2 The mean survival rate of 50 full-sib families at day 15 after challenge with high temperature. |
| In the text | |
 |
Fig. 3 Total number of dead oysters from all families (including the selection and control families) during the thermal challenge testing. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


