| Issue |
Aquat. Living Resour.
Volume 33, 2020
|
|
|---|---|---|
| Article Number | 25 | |
| Number of page(s) | 8 | |
| DOI | https://doi.org/10.1051/alr/2020026 | |
| Published online | 23 December 2020 | |
Research Article
Marine ornamental trade in Indonesia
1
Department of Zoology and Fisheries, Faculty of Agrobiology, Food and Natural Resources, Czech University of Life Sciences Prague, Kamýcká 129, Praha-Suchdol 16500, Czech Republic
2
Centre for Disaster Studies, Research and Community Empowerment, IPB University, 166 80 Bogor, Indonesia
3
Faculty of Biology, Universitas Jenderal Soedirman, Jalan dr. Soeparno 63, Purwokerto 53122, Indonesia
4
Directorate General of Fisheries Product Competitiveness, Ministry of Marine Affairs and Fisheries, Gambir, Jakarta 10110, Indonesia
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Olivier Thebaud
Received:
14
February
2019
Accepted:
8
December
2020
Abstract
Trade with marine species as ornamentals is an important sector of the international pet trade. The vast majority of these species are collected from the wild and one of the top supplying countries is Indonesia. Detailed evidence on trade with marine resources in Indonesia is lacking or it is hardly accessible. Moreover, the exploitation of ornamental species seems to be mostly uncontrolled. This study presents detailed characteristics of such trade for Indonesia, including the offered species, their sizes, prices, and conservation status, based on data and information obtained from wholesalers in 2018. The main provinces of marine wildlife collecting are also identified. In total, 777 marine vertebrate and invertebrate species were traded, belonging to 174 families including two species classified as endangered: Banggai cardinal fish (Pterapogon kauderni) and zebra shark (Stegostoma fasciatum). Commonly traded was red lionfish (Pterois volitans), known to be a successful invader. The volume of ornamental marine fish exported from Indonesia in period 2015–2019 was 3 353 983 kgs sold for 33 123 218 USD. The province of Bali was identified as the main exporter of ornamental marine fish within Indonesia. These findings should help to establish sustainable exploitation of marine resources in relation to conservation and wildlife management.
Key words: Aquarium / coral reef / export / ornamental species / pet trade / price
© EDP Sciences 2020
1 Introduction
The keeping of ornamental species in aquaria is amongst the most popular hobbies worldwide and represents a steadily growing sector of aquaculture and a multi-billion dollar industry (Tlusty, 2002; Padilla and Williams, 2004; Livengood and Chapman, 2007; Novák et al., 2020). In the past, setting up and maintaining marine aquaria was much more expensive in comparison to freshwater ones. Recent rapid improvements in husbandry methods and technologies have resulted in a change whereby moderately experienced fish keepers are now able to build and keep marine aquaria of relatively small volumes for reasonable prices (Cato and Brown, 2003; Ribeiro et al., 2009). Moreover, the popularity of marine animals as ornamentals has greatly increased due to the release of children's movies in which charismatic and attractively coloured marine biota are the star characters (Livengood and Chapman, 2007). Contrary to freshwater species, the vast majority of ornamental marine organisms do not reproduce in captivity and they are captured in the wild, especially in tropical coral reefs (Tlusty, 2002; Cato and Brown, 2003). It is clear that along with the growing popularity of marine animals as pets, the pressure on wild populations is on the rise.
Moreover, due to the development of hobbyists preferences, the global trade in live tropical marine organisms shifted from fish-only tanks to small coral reefs with diverse biota including also many invertebrates (Rhyne et al., 2009). This shift of preferences has already increased pressure on tropical coral reefs, one of the most overexploited and critically endangered ecosystems on the planet, from which originate the vast majority of ornamental marine species (Bruckner, 2005; Leal et al., 2016a,b).
Although there is no evidence that collecting and fishing for the pet trade has led to extinction of some marine species, local depletion is well documented (Lunn and Moreau, 2004; Nañola et al., 2011; Madduppa et al., 2014).
Collectors of ornamental marine biota are usually small-scale fishermen from tropical countries who work alone or in small family groups, and who supply these animals to intermediaries and middlemen, and consequently to large wholesale/exporter companies (Wabnitz et al., 2003). Despite suggested activities aimed at improving sustainable exploitation (Tlusty et al., 2013; Calado et al., 2014; Madduppa et al., 2014; Leal et al., 2016a,b), some local collectors still use very destructive techniques including application of potassium and sodium cyanide to capture fish (see Rubec et al., 2001).
Over 3000 marine species originating from more than 100 countries are known to be subjects of the international pet market annually (Rhyne et al., 2009; Leal et al., 2016a,b). The majority of traded organisms are exported to the United States, European Union and Japan (Rhyne et al., 2012), and one of the top suppliers is the Republic of Indonesia (Lunn and Moreau, 2004; Padilla and Williams, 2004; Leal et al., 2016a,b). Many people living in Indonesian coastal communities are economically dependent on the collection of marine animals for the pet trade and these communities are among the poorest in the country (Reksodihardjo-Lilley and Lilley, 2007).
Since intensive capture can cause a considerable decline in the abundance and density of target species, data about the trade are needed to evaluate related risks. Unfortunately, exact data on trade with marine resources in Indonesia are not easily accessible, and the exploitation of ornamental species is perceived as mostly uncontrolled (Lunn and Moreau, 2004).
In the present work, the current state of the Indonesian marine pet trade was summarised and the main areas of species collection and harvesting were identified. The information presented should help to establish sustainable exploitation of marine resources with respect to local and international conservation and wildlife management.
2 Materials and methods
2.1 Desktop research
Selected pet trade wholesalers in the Czech Republic were contacted to identify their main partners in Indonesia. Subsequently, a search of the literature that reports data on numbers of collected marine organisms in Indonesia was conducted. The standard search engine Google (in English and Indonesian languages) was used. Academic experts and stakeholders in Indonesia were interviewed by email to recommend studies, governmental reports and similar references. A useful source of data was identified and consisted of export statistics of fisheries products by commodity, province and port of export (“Statistik ekspor hasil perikanan menurut komoditi, provinsi dan pelabuhan asal ekspor”) released annually in period 2015–2019 by the Ministry of Marine Affairs and Fisheries, Republic of Indonesia. These statistics sort each of the evaluated commodities regarding type, volume and value (in kg and USD, respectively), and the amount of local capture in each Indonesian province. Since the authorities keep records only for the total weight of collected animals, it is not possible to present the actual quantity of captured individuals.
2.2 On-site research
In 2018 (period between 1st January to end of June), we collected detailed information covering the offered species based on personal visits and obtained actual price lists of two of the major wholesalers/exporters in Jakarta, Indonesia, which were previously identified and contacted in advance via email communication. We also gathered information on common names used as trade names, and origin of traded species (wild-harvested or cultured).
2.3 Data analyses
A species list including common names, sex and colouration morphs (when listed separately), size categories and wholesale prices of each offered aquatic organism was compiled, based on the data obtained. Usually, ornamental marine species are not stocked in the exporter's facilities but obtained from suppliers and middlemen when wholesalers/importers order listed species in the target area (as done for a vast majority of imports to the United States, the European Union or Japan; Rhyne et al., 2017). The species names were verified using online databases FishBase (www.fishbase.org) and WoRMS (World Register of Marine Species, http://www.marinespecies.org ). Misnomers, unaccepted, fuzzy and false names were recorded and discussed with livestock managers in the Indonesian wholesalers. Corrected names were later confirmed by livestock managers in one of the main European importers based in the Czech Republic, which is considered as a gateway for ornamental aquatic species to Europe. These organisms are imported here from supporting countries, partly sold locally and partly re-exported to other European countries upon customer request (Kalous et al., 2015). Additionally, the conservation status, trends and threats of each listed species by the IUCN Red List of Threatened Species (https://www.iucnredlist.org/) were included if known. Following categories are included in the IUCN Red List: Extinct (there is no reasonable doubt that the species is no longer extant); Extinct in the wild (survives only in captivity); Critically endangered (facing an extremely high risk of extinction in the wild); Endangered (facing a very high risk of extinction in the wild); Vulnerable (facing a high risk of extinction in the wild); Near threatened (close to being at high risk of extinction); Least concern (unlikely to become extinct in the near future); and Data deficient (more information is required for a proper assessment of conservation status).
3 Results
The results are given in Table 1. The total weight of marine fish captured and exported as ornamentals between 2015 and 2019 from Indonesia was 3 353 983 kg sold for 33 123 218 USD. Adult fish represented 3 287 432 kg (32 265 994 USD) while juveniles 66 551 kg (857 224 USD). Fish were exported from 18 Indonesian provinces. Bali was the province with the highest volume of captured and exported ornamental marine fish. Export statistics about another marine biota were not available.
In total, 777 marine species advertised both in Indonesia and abroad for sale as ornamentals in 2018 were identified (Tabs. 2 and 3): 501 fish species (52 families; with 73 species, the family Labridae had the highest species diversity in the trade), 13 elasmobranchs (sharks and rays; 8 families; with 3 species, the family Dasyatidae had the highest species diversity in the trade), 56 crustaceans (22 families; with 10 species, the family Diogenidae had the highest species diversity in the trade), 113 cnidarians (38 families; with 10 species, the family Lobophylliidae had the highest species diversity in the trade), 41 echinoderms (20 families; with 5 species, the family Goniasteridae had the highest species diversity in the trade), 43 molluscs (27 families; with 6 species, the family Cypraeidae had the highest species diversity in the trade), 5 annelids (2 families; with 4 species, the family Sabellidae had the highest species diversity in the trade), two ascidians (2 families, one species from both families Clavelinidae and Styelidae was advertised), two sponges (2 families; one species from both families Chalinidae and Microcionidae was advertised), and one flatworm (family Discodorididae).
In total, 368 species were found listed in the IUCN Red List of Threatened Species (Data deficient = 21, Least concern = 311, Near threatened = 19, Vulnerable = 15, and Endangered = 2; Fig. 1). The population trend was rated as decreasing in 28 species (including endangered zebra shark Stegostoma fasciatum), stable in 112 species, unknown (update required) in 227 species, and one species, red lionfish (Pterois volitans) was evaluated as increasing.
The species with the lowest price was mitra snail (Atrimitra idea) advertised for 0.15 USD per individual. The highest price of 700.00 USD was recorded for tawny nurse shark (Nebrius ferrugineus).
Only one recorded species was cultured in captivity: Banggai cardinal fish (Pterapogon kauderni), which is endangered with a decreasing population trend, but capture in the field was also recorded. Clownfishes are known to be produced on the north shore of Bali (Rhyne et al., 2017), but these species were not listed as domestically cultured in the surveyed price lists; and no sorting by colour morphs was recorded.
Most of the species were traded as adults, but some of them also in the juvenile stage, and in the single case of coral catshark (Atelomycterus sp.), the unhatched eggs were advertised.
The ratio between export and local trade was not evaluated due to a lack of data, but various species of fish, sharks, rays, anemones, corals, echinoderms and sea snails were recorded in the street markets in Jakarta (Fig. 2). A detailed list of all species with both their scientific and trade names, sizes, prices, IUCN status and population trend is given in the supplementary material (Tab. S1).
Indonesian provinces with recorded exports of marine juvenile and adult fishes in period 2015–2019, their total weight and total price per year, and total price per whole period.
Groups of marine animals found being traded in Indonesia in 2018, and number of families, the most abundant family and number of traded species in the most abundant family in each group.
Groups of marine animals found being traded in Indonesia in 2018, and number of families and species in each group.
 |
Fig. 1 Marine aquaria with animals offered for sale on the street in Jakarta, Indonesia. |
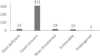 |
Fig. 2 Indonesian ornamental marine species listed in the IUCN Red List of Threatened Species and their categories. |
4 Discussion
Marine pet trade statistics are often uninterpretable because when available, they include the export of fish in water in total weight rather than as a number of individuals (Lunn and Moreau, 2004). It is also a common practice to include all marine ornamental species into a single reporting category (such as “ikan hias” − ornamental fish in Indonesian) and/or combine their trade figures with those of other commodities. Therefore, it is important to extend statistics by species, with their detailed price and ecological characteristics, providing better insights into the composition of this trade. A large number of marine animal species traded as ornamentals within the international and local pet trade in Indonesia were identified. Some species are known to be decreasing in abundance in the wild and two of them are listed as endangered in the IUCN Red List of Threatened Species. In comparison with fishery, the volume of endangered species captured for ornamental trade is mostly overlooked. For instance, Stegostoma fasciatum was recorded as bycatch in Indonesian fishery (Lindfield and Jaiteh, 2019) but detailed information on exploitation of these sharks as ornamentals should be gathered from further research. With the exception of P. kauderni, all species are harvested solely from the wild. There are 34 provinces in Indonesia and the above animals are harvested in 11 of them, with the vast majority exported from Bali, sometimes with transhipment to wholesalers in Jakarta (Java). The price of traded animals varied from 0.15 to 700.00 USD depending on the species and size, the most expensive being sharks. One can expect that the real volume of captured fish is much higher than statistics suggest, due to post-capture mortality, which is not recorded. What is alarming, up to 98% of wild-harvested marine ornamental creatures die within one year of capture (Yan, 2016).
Pterapogon kauderni is a favourite ornamental species endemic to the Banggai Islands, Indonesian archipelago. It has an oral incubation (paternal mouthbrooding) of eggs and hatchlings with a direct development (Fishelson and Gon, 2008) and dispersal of its juveniles is very limited due to the lacking stage of pelagic larvae (Vagelli, 1999). Therefore, intensive capture is very risky for the sustainability of the genetically isolated populations of this species which are due to unique life history especially vulnerable to local extinction (Lunn and Moreau, 2004; Ndobe et al., 2018).
Pterapogon kauderni can serve as a model of an ideal ornamental fish suitable for breeding in marine aquaria. It is a small and peaceful species with an unusual colouration, in which white, black and blue tones predominate. This fish reproduces well in captivity. Relatively seamless breeding, limited mainly by the number of 40–60 eggs in the father's mouth and the requirement of offspring for significant content of highly unsaturated fatty acids (HUFA) in food have impact on reality that fish farmers are unable to saturate sufficiently the demand for this species (Dodds, 2009; Schum, 2009; Vagelli, 2002, 2011). The share of tank-bred fish, especially those originating from Thailand, has been growing since 2012 significantly, probably because hobbyists prefer captive-bred fish due to the likelihood of a higher survival rate (Ndobe et al., 2018). Within its native range, isolated populations of this species occur only in surroundings of 34 from 67 islands (Vagelli, 2011, 2015; Ndobe et al., 2013). Vagelli (2015) estimated potential habitat suitable for P. kauderni fish being ca. 30 km2. Ndobe et al. (2018) pointed out that the habitat covers only 20–24 km2.
Dodds (2009) and Vaggelli (2011) estimated current population of 2.4 million individuals with about 900 000 individuals being exported yearly and with together 18 million fish collected since 1995. Harvest of this species do not represent an important part of local economy, because only about 230 collectors are involved in fishing (Lunn and Moreau, 2004). At the current payment rate of 0.01–0.025 USD per fish, it represents an average number of less than 8000 fish per collector supplemental income for each collector of between 78 and 195 USD per year (Vagelli and Erdmann, 2002). Dodds (2009) further estimates mortality between collection and export at 55%. According to Lunn and Moreau (2004), only slightly over 10 000 individuals of P. kauderni are sold in the wholesale network. This means, according to Dodds (2009), the total mortality is at 99% level during shipping from the exporter to the distributor. Iridovirus (BCIV) and bacteria of the genus Vibrio in combination with stressful conditions have the significant negative effect on the mortality of this species in the store chain (Ndobe et al., 2018).
Since P. kauderni is known for being captured in large quantities, the capture is probably one of the main reasons why the population trend is decreasing. As part of the regulation of capture and export for the pet trade, it was proposed to include P. kauderni in the CITES Appendix II, but this was consequently withdrawn by the European Union (CoP 14 Prop. 19, www.cites.org). Indonesia was encouraged to implement conservation measures to ensure the sustainability of exploitation of this species and to reduce post-capture mortality, which is about 83% (Lunn and Moreau, 2004). A possible solution is substitution of fish harvested from the wild by fish bred and reared in captivity. However, it was previously noted that fish harvested from the wild are much cheaper than captive-bred fish, and therefore still widely traded (Vagelli, 2011). On the contrary, both categories were found advertised for sale at the same price of 2.0 USD. The price is relatively low, popularity high, and thus continued capturing is expected. In the case of clownfish species, no specific colour morphs like melanistic and aberrant were offered, and hence the price was not dependent on this factor contrary to clownfishes exported from Papua New Guinea, as recently reported by Militz et al. (2018). After detailed analysis, Ndobe et al. (2018) subsequently concluded that managing of this species and its microhabitats (fishing quotes, community-based aquaculture) could provide benefit to endemic populations and at the same time enhance potential for tourism and fisheries.
Intensive trade with this species is paradoxically accompanied by the expansion of its natural range to areas of highly restricted localities along trade routes in Sulawesi, in particular in Luwuk, Palu Bay, Lembeh Strait, Tumbak, Kendari (Erdmann and Vagelli, 2001; Vagelli and Erdmann, 2002; Moore and Ndobe, 2007; Vagelli, 2011; Moore et al., 2012), where traded fish probably escaped from net cages (Lunn and Moreau, 2004, Moore et al., 2012). One population has established in Gilimanuk, North Bali (Lilley, 2008) and future records in more sites in the Banggai Archipelago is expected (Ndobe et al., 2018). Pterapogon kauderni is recently categorized as Endangered in the IUCN Red List on the basis of a very small area occupied by fragmented populations, lack of dispersal mechanisms and decrease due to exploitation for the international pet trade (Allen and Donaldson, 2007). By jurisdiction of the European Union it is only listed in CITES Appendix D Council Regulation (EC) No. 338/97 of 9 December 1996 on the protection of species of wild fauna and flora by regulating trade therein (http://data.europa.eu/eli/reg/1997/338/2020-01-01).
The vast majority of traded species are of a small size (a few centimetres) but also bigger creatures (such as tawny nurse shark, Nebrius ferrugineus) were recorded. The trade size of this species is 100 cm and it can grow up to 320 cm in body length. Nebrius ferrugineus is classified as Vulnerable, S. fasciatum as Endangered and the other advertised shark species as Near threatened even though the population trend of Carcharhinus melanopterus is decreasing. Given the relatively high price of traded large shark individuals, their popularity as ornamentals is not particularly high, and hence their harvested quantities are probably also not high. Since these species are too big for home aquaria and in view of their conservation status, capture only for support of sustainable captive populations in zoos and public aquaria can be considered desirable (and might be of a high priority; Buckley et al., 2018). Recent research appears to indicate that strict conservation of reef sharks leads to the recovery of overexploited populations (Speed et al., 2018). Unfortunately, these species are harvested without apparent regulation from the wild in Indonesia, and their continued decrease could be expected.
On the other hand, some of the traded species are known to be invasive when introduced into new localities. For example, P. volitans, which is the only species with an increasing population trend on the presented list, is considered to be one of the most invasive species in the world (Albins and Hixon, 2013). Also, invertebrates can behave as invaders. For example, the Sally lightfoot crab (Percnon gibbesi), advertised for sale for 1.14 USD in Indonesia and 20 EUR in European pet shops (Calado, 2012), is considered to be non-native but established in the Mediterranean by many authors (Félix-Hackradt et al., 2010; Katsanevakis et al., 2011; Stasolla and Innocenti, 2014), even though the natural spread of long-living planktonic larvae into the Mediterranean by the Atlantic Current cannot be excluded as a causal factor (Ulman et al., 2017).
The commercial value of marine animals in the international pet trade is high, and the popularity of aquarium keeping as a hobby is increasing year by year globally. Thus, higher pressure on animals in their native ranges is expected because of their collection in the wild. Although there are logical suggestions to capture ornamental organisms in their non-native range to mitigate their spread and to alleviate the fishing pressure on species in their native range (Calado, 2012), significant reduction of import from Indonesia is not expected due to socio-economic reasons. Although the negative consequences on marine ecosystems of excess harvesting of ornamental species are well known, effective policy measures are lacking in many cases (Patoka et al., 2018).
The spectrum of traded species from Indonesia is broad, including vertebrates and invertebrates, mostly inhabiting coral reef environments. The lack of continuous and systematic monitoring precludes the establishment of harvesting targets and management strategies ensuring the sustainable exploitation of these living marine resources. In total, 502 species of fishes, 13 of sharks and rays, and 276 of invertebrates were found in the marine pet trade in 2018. In contrast, Rhyne et al. (2017) reported 992 species of fishes and 301 of invertebrates to be exported from Indonesia to the United States in 2011 which suggests that some species can be traded as misidentified and wrongly labelled in Indonesia. Further survey is needed in this regard.
5 Conclusion
Species-specific information on trade (local and exports-based) of ornamental species provides a better understanding of incentives which may drive the development of harvesting (knowing price ranges), as well as an indirect measure of fishing pressure, which can be related to the knowledge of the biological status of the resources. The geographical information also helps assess the location of this pressure, and whether it seems to apply to areas where the species may be invasive, or not. Gathered data can be used as pre-requisites to the development of species-specific targets and management strategies for the development of sustainable industries, with ensuing ecological, social and economic benefits.
To established sustainable exploitation of living marine resources, conservationists and wildlife managers must implement appropriate and effective monitoring, including both field surveys and market analyses, followed by local regulations or restrictions focused mainly on endangered species or selected populations. On the other hand, only regulation and restriction are not particularly effective, and efforts aimed at public education are crucial. Importantly, not only protection of local species is needed, but also awareness of potentially invasive species. This is because the regions that have climatic conditions and habitats suitable for tropical marine species common in the pet trade are also considered to be places with a high probability of non-native species establishment (Semmens et al., 2004).
Finally, it is worth mentioning that a detailed future survey of the local trade in marine ornamentals in Indonesia is very important. An understanding of the full environmental and socio-economic complexity of this growing sector of the pet industry is a premise for proper implementation of measures to improve marine wildlife management.
Supplementary Material
Table S1. Detailed list of marine species traded in Indonesia as ornamentals; group of animals, family, scientific and common names, size in cm, price over the entire year in USD, IUCN conservation status (if known), and population trend based on data from IUCN (if known). Access Supplementary Material
Acknowledgements
This study was supported by the Internal Grant Agency of the Czech University of Life Sciences Prague “CIGA” (No. 20182013). We thank Julian D. Reynolds for proofreading the English.
References
- Albins MA, Hixon MA. 2013. Worst case scenario: potential long-term effects of invasive predatory lionfish (Pterois volitans) on Atlantic and Caribbean coral-reef communities. Environ Biol Fish 96: 1151–1157. [CrossRef] [Google Scholar]
- Allen GR, Donaldson TJ. 2007. Pterapogon kauderni. In: IUCN 2009. IUCN Red List of Threatened Species. Version 2009. 1. (www.iucnredlist.org) [Google Scholar]
- Bruckner A. 2005. The importance of the marine ornamental reef fish trade in the wider Caribbean. Rev Biol Trop 53: 127–137. [PubMed] [Google Scholar]
- Buckley KA, Crook DA, Pillans RD, Smith L, Kyne PM. 2018. Sustainability of threatened species displayed in public aquaria, with a case study of Australian sharks and rays. Rev Fish Biol Fisher 28: 137–151. [CrossRef] [Google Scholar]
- Calado R. 2012. Invasive alien crabs can be used for marine aquariums − turning a threat into profit. Crustaceana 85: 257–265. [Google Scholar]
- Calado R, Leal MC, Vaz M, Brown C, Rosa R, Stevenson TC, Cooper CH, Tissot BN, Li YW, Thornhill DJ. 2014. Caught in the Act: how the US Lacey Act can hamper the fight against cyanide fishing in tropical coral reefs. Conserv Lett 7: 561–564. [Google Scholar]
- Cato JC, Brown CL. Marine ornamental species: collection, culture and conservation, 1st edn, Iowa State Press, Ames, Iowa, 2003. [Google Scholar]
- Dodds K. 2009. Cardinal sin: The plight of the Banggai cardinalfish Pterapogon kauderni Koumans 1933. Trop Fish Hobbyist 57: 92–98. [Google Scholar]
- Erdmann MV, Vagelli A. 2001. Banggai cardinalfish invade Lembeh Strait. Coral Reefs 20: 252–253. [Google Scholar]
- Félix-Hackradt F, Hackradt C, Treviño-Otón J, García-Charton J. 2010. Continued expansion of Percnon gibbesi (Crustacea: Decapoda: Plagusiidae) into western Mediterranean waters. Mar Biodivers Rec 3: e22. [CrossRef] [Google Scholar]
- Fishelson L, Gon O. 2008. Comparative oogenesis in cardinal fishes (Apogonidae, Perciformes), with special focus on the adaptive structures of the egg envelopes. Environ Biol Fishes 8: 397–414. [Google Scholar]
- Kalous L, Patoka J, Kopecký O. 2015. European hub for invaders: Risk assessment of freshwater ornamental fish exported from the Czech Republic. Acta Ichthyol Piscat 45: 239–245. [CrossRef] [Google Scholar]
- Katsanevakis S, Poursanidis D, Yokes MB, Mačić V, Beqiraj S, Kashta L, Sghaier YR, Zakhama-Sraieb R, Benamer I, Bitar G. 2011. Twelve years after the first report of the crab Percnon gibbesi (H. Milne Edwards, 1853) in the Mediterranean: current distribution and invasion rates. J Biol Res Thessalon 16: 224–236. [Google Scholar]
- Leal MC, Ferrier‐Pagès C, Petersen D, Osinga R. 2016a. Coral aquaculture: applying scientific knowledge to ex situ production. Rev Aquacult 8: 136–153. [CrossRef] [Google Scholar]
- Leal MC, Vaz MCM, Puga J, Rocha RJM, Brown C, Rosa R, Calado R. 2016b. Marine ornamental fish imports in the European Union: an economic perspective. Fish Fish 17: 459–468. [CrossRef] [Google Scholar]
- Lilley R. 2008. The Banggai cardinalfish: an overview of conservation challenges. SPC Live Reef Fish Information Bulletin 18: 3–12. [Google Scholar]
- Lindfield S, Jaiteh V. 2019. Assessing shark and ray bycatch in Indonesian deepwater snapper-grouper fisheries. The Nature Conservancy Indonesia, 1–13. [Google Scholar]
- Livengood E, Chapman F. 2007. The ornamental fish trade: an introduction with perspectives for responsible aquarium fish ownership. University of Florida IFAS Extension FA124, 1–8. [Google Scholar]
- Lunn K, Moreau M-A. 2004. Unmonitored trade in marine ornamental fishes: the case of Indonesia's Banggai cardinalfish (Pterapogon kauderni). Coral Reefs 23: 344–351. [Google Scholar]
- Madduppa HH, von Juterzenka K, Syakir M, Kochzius M. 2014. Socio-economy of marine ornamental fishery and its impact on the population structure of the clown anemonefish Amphiprion ocellaris and its host anemones in Spermonde Archipelago, Indonesia. Ocean Coast Manag 100: 41–50. [Google Scholar]
- Militz TA, Foale S, Kinch J, Southgate PC. 2018. Natural rarity places clownfish colour morphs at risk of targeted and opportunistic exploitation in a marine aquarium fishery. Aquat Living Resour 31:18. [CrossRef] [EDP Sciences] [Google Scholar]
- Moore A, Ndobe S. 2007. Discovery of an introduced Banggai cardinalfish population in Palu Bay, Central Sulawesi, Indonesia. Coral Reefs 26: 569–569. [Google Scholar]
- Moore A, Ndobe S, Salanggon A, Rahman A. 2012. Banggai cardinalfish ornamental fishery: the importance of microhabitat, Proceedings of the 12th International Coral Reef Symposium (July), pp. 9–13. [Google Scholar]
- Nañola CL, Aliño PM, Carpenter KE. 2011. Exploitation-related reef fish species richness depletion in the epicenter of marine biodiversity. Environ Biol Fish 90: 405–420. [CrossRef] [Google Scholar]
- Ndobe S, Soemarno, Herawati EY, Setyohadi D, Moore A, Palomares MLD, Pauly D. 2013. History of Banggai cardinalfish, Pterapogon kauderni (Actinopterygii: Perciformes: Apogonidae), from Banggai Islands and Palu Bay, Sulawesi, Indonesia. Acta Ichthyol Piscat 43: 237–250. [CrossRef] [Google Scholar]
- Ndobe S, Yasir I, Noore AM, Biondo MV, Foster SJ. 2018. A study to assess the impact of international trade on the conservation status of Pterapogon kauderni (Banggai cardinalfish). International Union for Conservation of Nature AC30, 16. [Google Scholar]
- Novák J, Kalous L, Patoka J. 2020. Modern ornamental aquaculture in Europe: early history of freshwater fish imports. Rev Aquacult DOI:10.1111/raq.12421. [Google Scholar]
- Padilla DK, Williams SL. 2004. Beyond ballast water: aquarium and ornamental trades as sources of invasive species in aquatic ecosystems. Front Ecol Environ 2: 131–138. [Google Scholar]
- Patoka J, Magalhães ALB, Kouba A, Faulkes Z, Jerikho R, Vitule JRS. 2018. Invasive aquatic pets: failed policies increase risks of harmful invasions. Biodivers Conserv 27: 3037–3046. [Google Scholar]
- Reksodihardjo-Lilley G., Lilley R. 2007. Towards a sustainable marine aquarium trade: an Indonesian perspective. SPC Live Reef Fish Inform Bull 17: 11–19. [Google Scholar]
- Rhyne A, Rotjan R, Bruckner A, Tlusty M. 2009. Crawling to collapse: ecologically unsound ornamental invertebrate fisheries. PLoS One 4: e8413. [Google Scholar]
- Rhyne AL, Tlusty MF, Schofield PJ, Kaufman L, Morris Jr JA, Bruckner AW. 2012. Revealing the appetite of the marine aquarium fish trade: the volume and biodiversity of fish imported into the United States. PLoS One 7: e35808. [Google Scholar]
- Rhyne AL, Tlusty MF, Szczebak JT, Holmberg RJ. 2017. Expanding our understanding of the trade in marine aquarium animals. PeerJ 5: e2949. [CrossRef] [PubMed] [Google Scholar]
- Ribeiro F, Collares-Pereira M, Moyle P. 2009. Non-native fish in the fresh waters of Portugal, Azores and Madeira Islands: a growing threat to aquatic biodiversity. Fish Manag Ecol 16: 255–264. [Google Scholar]
- Rubec PJ, Cruz F, Pratt V, Oellers R, McCullough B, Lallo F. 2001. Cyanide-free net-caught fish for the marine aquarium trade. Aquar Sci Conserv 3: 37–51. [CrossRef] [Google Scholar]
- Schum T. 2009. Breeding Banggai Cardinalfish: a hobbyist́s perspective. Trop Fish Hobbyist 57: 96–98. [Google Scholar]
- Semmens BX, Buhle ER, Salomon AK, Pattengill-Semmens CV. 2004. A hotspot of non-native marine fishes: evidence for the aquarium trade as an invasion pathway. Mar Ecol Prog Ser 266: 239–244. [Google Scholar]
- Speed CW, Cappo M, Meekan MG. 2018. Evidence for rapid recovery of shark populations within a coral reef marine protected area. Biol Conserv 220: 308–319. [Google Scholar]
- Stasolla G, Innocenti G. 2014. New records of the invasive crabs Callinectes sapidus Rathbun, 1896 and Percnon gibbesi (H. Milne Edwards, 1853) along the Italian coasts. BioInvasions Rec 3: 39–43. [Google Scholar]
- Tlusty M. 2002. The benefits and risks of aquacultural production for the aquarium trade. Aquaculture 205: 203–219. [Google Scholar]
- Tlusty MF, Rhyne AL, Kaufman L, Hutchins M, Reid GMG, Andrews C, Boyle P, Hemdal J, McGilvray F, Dowd S. 2013. Opportunities for public aquariums to increase the sustainability of the aquatic animal trade. Zoo Biol 32: 1–12. [CrossRef] [PubMed] [Google Scholar]
- Ulman A, Ferrario J, Occhpinti-Ambrogi A, Arvanitidis C, Bandi A, Bertolino M, Bogi C, Chatzigeorgiou G, Çiçek BA, Deidun A. 2017. A massive update of non-indigenous species records in Mediterranean marinas. PeerJ 5: e3954. [Google Scholar]
- Vagelli A. 1999. The reproductive biology and early ontogeny of the mouthbrooding Banggai cardinalfish, Pterapogon kauderni (Perciformes, Apogonidae). Environ Biol Fish 56: 79–92. [CrossRef] [Google Scholar]
- Vagelli AA. 2002. Notes on the biology, geographic distribution, and conservation status of the Banggai cardinalfish Pterapogon kauderni Koumans 1933, with comments on captive breeding techniques. Trop Fish Hobbyist 51: 84–88. [Google Scholar]
- Vagelli AA. 2011. The Banggai cardinalfish: natural history, conservation, and culture of Pterapogon kauderni. Chichester, UK, John Wiley & Sons., pp. 1–203. [Google Scholar]
- Vagelli AA. 2015. Update on populations' condition of the Banggai cardinalfish Pterapogon kauderni. Unpublished report (available upon request at: https://federalregister.gov/a/2016-00943). [Google Scholar]
- Vagelli AA, Erdmann M. 2002. First comprehensive ecological survey of the Banggai cardinalfish, Pterapogon kauderni . Environ Biol Fish 63: 1–8. [CrossRef] [Google Scholar]
- Wabnitz C, Taylor M, Green E, Razak T. 2003. From ocean to aquarium: the global trade in marine ornamental species. Cambridge, UK, UNEP/Earthprint. [Google Scholar]
- Yan G. 2016. Saving Nemo–Reducing mortality rates of wild-caught ornamental fish. SPC Live Reef Fish Inform Bull 21: 3–7. [Google Scholar]
Cite this article as: Akmal SG, Zámečníková-Wanma BPD, Prabowo RE, Khatami AM, Novák J, Petrtýl M, Kalous L, Patoka J. 2020. Marine ornamental trade in Indonesia. Aquat. Living Resour. 33: 25
All Tables
Indonesian provinces with recorded exports of marine juvenile and adult fishes in period 2015–2019, their total weight and total price per year, and total price per whole period.
Groups of marine animals found being traded in Indonesia in 2018, and number of families, the most abundant family and number of traded species in the most abundant family in each group.
Groups of marine animals found being traded in Indonesia in 2018, and number of families and species in each group.
All Figures
 |
Fig. 1 Marine aquaria with animals offered for sale on the street in Jakarta, Indonesia. |
| In the text | |
 |
Fig. 2 Indonesian ornamental marine species listed in the IUCN Red List of Threatened Species and their categories. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


