| Issue |
Aquat. Living Resour.
Volume 39, 2026
Dynamics of small-scale fishery social-ecological systems: interdisciplinary insights from Madagascar
|
|
|---|---|---|
| Article Number | 6 | |
| Number of page(s) | 17 | |
| DOI | https://doi.org/10.1051/alr/2025020 | |
| Published online | 10 February 2026 | |
Research Article
Fine-scale fishery patterns reveal challenges and opportunities for coastal management and conservation in Madagascar
1
Institut Halieutique et des Sciences Marines (IH.SM), University of Toliara, Toliara, Madagascar
2
ENTROPIE (IRD, University of Reunion, University of New Caledonia, Ifremer), IUEM, Plouzané, France
3
DECOD (L’Institut Agro, Ifremer, INRAE), Rennes, France
4
CREST (ENSAI, University of Rennes), Bruz, France
5
IRMAR (L’Institut Agro, University of Rennes, CNRS), Rennes, France
6
AMURE (IRD, University of Western Brittany, Ifremer, CNRS), Plouzané, France
* Corresponding authors: This email address is being protected from spambots. You need JavaScript enabled to view it.
; This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
11
July
2025
Accepted:
13
November
2025
Abstract
The blue economy agenda has generated tensions over marine space use, often marginalising small-scale fisheries in development policies. Boat tracking technology has only recently begun to be applied in these fisheries, but it offers a promising approach to accurately map fishing distribution. In this study, we explored how environmental, technological, and social factors influenced fishers’ spatial behaviour and catches at sea in one of the Madagascar's most heavily exploited coral reef fisheries. We recorded boat movements at 30 s intervals and reef fish catches simultaneously during a 12-month participatory survey. The spatial distribution of annual fishing effort (h·ha−1) and fish catch rates (kg·ha−1) was characterised by fishing community and gear type (beach seine, mosquito net trawl, gillnet, handline, and speargun) at 250−m resolution. A total of 75 reef fish families were recorded in catches (1,466 t·yr−1) across approximately 218 km2. Annual catch rates of the seven dominant families (comprising 62% of total catches) were highly variable and heterogeneous (mostly 1–391 kg·ha−1) across marine habitats. A total of 7,359 tracks by 521 boats were recorded. Fishing pressure was highly variable spatially (mostly 1−150 h·ha−1) among communities and gear types. The results revealed preferential target areas, informal marine tenure, limited travel distance to fishing grounds, and overexploitation patterns within the fishery, offering critical insights for fishery management and inclusive marine spatial planning. This study showed the usefulness and opportunities of deploying combined boat tracking and catch surveys in small-scale fisheries through participatory research.
Key words: Participatory research / coral reef fishes / GPS tracking data / marine spatial planning / territory
Publisher note: The article has been added in the following special issue “Dynamics of small-scale fishery social-ecological systems: interdisciplinary insights from Madagascar” on 13 March 2026.
© F. Behivoke et al., Published by EDP Sciences 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Small-scale fisheries have experienced declining catch rates globally over the past 50 yr, largely due to overfishing and environmental change (e.g., Muallil et al., 2014; Zeller et al., 2015). Their heterogeneity, diversity, and informality, coupled with limited institutional capacity, contribute to chronic data scarcity and policy invisibility compared with industrial fisheries (Basurto et al., 2025). In addition, dominant, economic and conservation interests have threatened local livelihoods, seafood value chains, and access to marine resources through ocean grabbing (Bennett et al., 2015; Blythe et al., 2023). The blue economy agenda, which seeks to balance marine exploitation and conservation while improving coastal livelihoods, has generated additional tensions and conflicts over marine resource and space uses, often marginalising small-scale fisheries (Cohen et al., 2019; Gill et al., 2023). This challenge is further exacerbated by climate change, since shifting target species distribution drives change in fishing practices, necessitating adaptive management approaches (Beckensteiner et al., 2023). Addressing the monitoring gap is therefore critical for small-scale fishery sustainability. Evaluating the sustainability of small-scale fisheries necessitates accurate quantitative and spatial data on fishing effort, catches (Wilen, 2004; McCluskey and Lewison, 2008; Navarrete Forero et al., 2017), and its drivers (Turner et al., 2013). For instance, in the absence of high resolution spatial information, it remains unclear which fishing activities are most impacted by decline or how they affect ecosystems (Prince and Hordyk, 2019). Spatial fishery knowledge is also crucial in marine spatial planning (Grantham et al., 2009; Janßen et al., 2018), e.g., for assessing the economic and/or conservation value of fishing areas or for revealing fishers’ behaviour based on local rules (Pellowe and Leslie, 2020).
Diverse methodologies have been developed to spatially characterize fishing activities in small-scale fisheries based on coarse indicators and proxy variables such as distance to ports, markets, or vessel density (Thiault et al., 2017; Chollett et al., 2022) and through fisher surveys (Guillemot et al., 2009; Turner et al., 2013). Map-based fisher interviews remain a cost-effective and widely used approach to assess the spatial distribution of fishing pressure across countries, fisheries, and target species using local knowledge (Grati et al., 2022). However, the reliability and precision of such data may be questioned due to multiple factors, e.g., fishers’ mistrust (Couclelis, 2003), intentional or unintentional reporting errors (Close and Brent Hall, 2006), limited sampling representativeness of fisher interview surveys (Scholz et al., 2011; Gill et al., 2019), and/or the fact that such surveys hardly capture the temporal variability of fisher behaviours (Léopold et al., 2014). The development of affordable global positioning system (GPS) devices has recently provided viable alternatives to participatory fisher surveys and new opportunities for mapping small-scale fisheries precisely and reliably, as in industrial fisheries using vessel monitoring system (VMS) and automatic identification system (AIS) technologies. Industrial fisheries have been extensively characterised over the last 15 yr using VMS and AIS data to analyze the temporal and spatial dynamics of fishing activities across different fleets and gear types (e.g., Lee et al., 2010; Gerritsen and Lordan, 2011; Watson et al., 2018; Tassetti et al., 2019). Real-time tracking data allows for mapping fishery patterns accurately to better assess the spatial distribution of fishing pressure and improve quantitative fishery-dependent indices, such as spatial catch-per-unit-effort (CPUE). In small-scale fisheries, boat tracking tools have increasingly been employed to map resource exploitation distribution and support fishery assessment and management (e.g., Burgos et al., 2013; Fernández-Boán et al., 2013; Tassetti et al., 2019; Méndez-Espinoza et al., 2020; Tilley et al., 2020; Thoya et al., 2020; Palmer et al., 2024). However, tracking surveys remain uncommon, often limited in duration and/or geographic scope, and rarely coupled with detailed catch data due to methodological and operational constraints. In this perspective, extensive tracking-based studies combining spatial fishing effort and catch data look critical in small-scale fisheries to further investigate the usefulness of such integrated, high-technology monitoring programs for guiding management.
The objectives of the study were to describe the fine-scale spatial distribution of fishing effort and catches of fish taxa and explore fishing patterns among fishing villages in one of the most heavily exploited coral reef fisheries in Madagascar. We recorded small-scale fishers’ movements and fish catches through a 12-month tracking survey. We then examined the annual distribution of fishing effort (in h·ha−1) and fish catches at 250-m resolution according to villages, gear types, and reef geomorphology through spatial and statistical analysis of tracking and catch data. We discussed the value of map outputs in revealing key spatial fishery characteristics, including preferential target areas, informal access rules, overexploitation patterns, and the implications for management and marine spatial planning in the context of increasing coverage of marine protected areas in the Western Indian Ocean (WIO).
2 Materials and methods
2.1 Study area
The study was carried out in Southwest Madagascar, near the regional capital city of Toliara (approx. 320,000 inhabitants). The marine area in the bay of Toliara (170 km2) includes three primary reef geomorphological units (Andrefouët et al., 2013; Fig. 1). A large shallow terrace (64.2 km2) features internal reef patches and intertidal seagrass beds. The lagoon area (59.4 km2), reaching depths of up to 15−m, is bounded by the southern and northern passes. Barrier reefs include the Nosy Tafara reef and the Grand Récif de Toliara reef (42.1 km2 and 18-km long), which is the largest continuous barrier reef in the WIO. Mangrove areas (4.3 km2) are distributed in isolated patches. The external reef slope extends in open water, reaching depths of approximately 200-m.
Coastal small-scale fishing is the primary livelihood and income source of households in six urban (Mahavatsy 2, Besakoa, and Ambohitsabo, Ankiembe-haut, Mahavatsy 1, and Ankiembe-bas) and two rural (Sarodrano and Ankilibe) villages. The population of these villages (approx. 36,000 inhabitants) is composed of diverse ethnic groups (mainly Vezo, Sara, Tanalana, Masikoro, Mahafaly, and Tandroy). Fishers operated traditional sailing monoxyl outrigger pirogues (2.5-m to 7.5-m long) with no outboard power or refrigeration. Five main fishing gear types were used: gillnet (n=358), handline (n=142), speargun (n=133), mosquito net trawl (n=131), and beach seine (n=128) (Tab. 1). Although the bay of Toliara falls within the “bay of Ranobe” marine protected area, no formal fishing regulations were enforced in the coral reef fishery.
In addition to fishing activities, seaweed (red algae Kappaphycus alvarezii) and sea cucumber (sandfish Holothuria scabra) farming has developed since 2014 in suitable coastal marine habitats with the support of scientific institutions and private investments (Fig. 1). Maritime infrastructures in the study area remained limited, including the commercial port wharf for international shipments and national cabotage while tourism activities were anecdotal (Fig. 1).
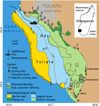 |
Fig. 1 Survey area in the bay of Toliara, southwestern Madagascar. Reef geomorphology units are shown as distinct colours. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8). Recent fishing settlements are shown in italics. |
Fishery and survey characteristics per village and gear type in the study area. The total number of fishing boats (% sampled in brackets), GPS tracks, catch landing records, and participatory catch records are shown. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8).
2.2 Data collection
2.2.1 Boat GPS tracking
In 2017, a total of 892 fishing boats (employing around 2,000 fishers) operated in the area (Behivoke et al., 2021). We recorded fishing boat trajectories using GPS trackers (i-GotU GT600 and CatLog Gen2) for 12 consecutive months from May 2018 to April 2019 (hereafter “GPS dataset”) (Fig. 2). A total of 100 boats were sampled each month and proportionally distributed across villages and fishing gear types (Tab. 1). Because strict random selection of boats was not achieved due to the voluntary and participatory nature of the monitoring protocol, boat samples partly overlapped across months, resulting in 560 boats (62.7% of total boats) being surveyed during 1–3 months over the study period. Participating fishers received a fixed monthly incentive of USD 9 boat−1. A local assistant was trained and hired in each village to help coordinate the work of volunteer fishers.
GPS trackers recorded boat position at 30 s intervals for about 25 consecutive days. High temporal resolution was required due to the expected short duration of fishing operations as generally observed in small-scale fisheries (Rufino et al., 2023). GPS data were downloaded monthly in CSV format and uploaded into a PostgreSQL database for statistical processing and spatial analysis.
 |
Fig. 2 Sampling and analytical procedure of fishery data used for characterizing the spatial distribution of fishing effort and catches in the coral reef fishery. |
2.2.2 Reef fish catch monitoring
Tracking data on fishing trips was combined with reef fish catch monitoring. The study focused on reef fishes due to their economic and nutritional importance. Catch per trip was recorded through a participatory catch survey (quantity) and a landing survey (composition) (Ranaivomanana et al., 2023) (Fig. 2). As part of the participatory catch survey, all sample fishers recorded fishing and non-fishing days in a logsheet as well as the total reef fish catch weight per trip using scales (precision 0.25 kg). As part of the landing survey, one trip of each sample boat was randomly sampled at landing sites once every two months (i.e., in May–June 2018, July–August 2018, September–October 2018, November–December 2018, January–February 2019, and March–April 2019) for fish identification. For each sample trip, reef fishes were identified at the family level from a 1−3 kg subsample of the catch using regional fish identification guides (Fisher and Bianchi, 1984; Taquet and Diringer, 2012). The number of individuals by family and the length of each specimen in the subsamples were then measured using standardised photographs following Andrialovanirina et al. ’s (2020) method.
2.3 Data analysis
2.3.1 Estimating and mapping annual fishing effort
Tracking data was processed as follows. First, fishing hauls were identified for each trip (Fig. 2; step E1). As mentioned in the introduction, reliable automated analytical frameworks using machine learning techniques have been developed for modelling boat movements and differentiating fishing and non-fishing activities during fishing trips across diverse gear types and contexts, which allows for high-resolution spatial and hourly temporal quantification and localization of harvesting events. We used an automatic classification method developed by Behivoke et al. (2021) for this study area. This model was deemed to detect fishing events at 74% to 89% accuracy across the five gear types based on 306 ground-truthed fishing tracks used to train a random forest algorithm following a cross-validation procedure (see Behivoke et al., 2021 for methodological details). The raw GPS fishing positions were aggregated for each trip (with 1 reconstructed position ∼ 1 min) to estimate fishing effort per trip (in min). Trips shorter than 30 min were excluded to remove artefacts and non-representative data based on empirical evidence of the minimum fishing trip duration in the area.
Second the spatial distribution of the total fishing effort of the GPS dataset was characterised by counting the number of fishing positions within a 250 × 250 m square grid (Fig. 2; step E2). Grid resolution (250 × 250 m, or 6.25 ha) was chosen to match the spatial precision suitable for fishery mapping in the area. The cumulative duration of fishing (in hours) in the sample data was straightforwardly calculated in each cell of the grid. Third, the fishing effort derived from the GPS dataset was extrapolated by sample stratum (village x gear type) for the total number of boats at the annual level through statistical inference following Ranaivomanana et al. ’s (2023) method (Fig. 2; step E3). Finally, annual fishing effort was mapped (in h·ha−1) i) by village and by gear type and ii) for the whole fishery by summing up these spatial estimates across villages and gear types (Fig. 2; step E4).
2.3.2 Travel time and distance to fishing grounds
The travel distance, time, and speed to and from the fishing sites were derived from the 306 fishing tracks of the observational dataset used for the random forest training procedure at step E1 (Fig. 2). The number of GPS non-fishing positions categorised as “travel” to the fishing ground and back to the landing site was first counted in each fishing track. The travel time (in min) per trip was then estimated as the average of the outgoing and returning travel times. The travel distance per trip (in km) was calculated as the cumulated distance between two consecutive GPS non-fishing positions during travel time. The travel speed per trip (in km·h−1) was derived by dividing the travel distance by the travel time for that trip. Few trips (<5% of total trips) with speeds above 7 knots were flagged as potential outliers (e.g., GPS error) and excluded from final analysis. To investigate potential effects of geographical factors, the average travel distance, time, and speed were finally estimated for each village using a generalised linear mixed model (GLMM). The village and gear type were incorporated as fixed and random effects, respectively, which accounted for the nested data structure (gear types within villages).
2.3.3 Estimating and mapping catch level and composition
A total of 15,903 and 525 fishing trips were monitored by the participatory catch survey and the landing survey, respectively (Tab. 1). During the landing survey, 75 families were observed from a total of 23,164 fish. Catch estimates were calculated for 74 out of the 75 families following the procedure described below. The 590 fishes (2.4% of the total) belonging to the Caracanthidae family were excluded in the analysis due to unavailable information on length-weight relationships.
To analyse catch composition at the family level in the fishery, we first retrieved the length-weight relationship coefficients from Fishbase for each reef fish species of the families recorded both during the landing survey and in Madagascar’s checklist (Fricke et al., 2018), computed the average coefficients for each family, and estimated the weight of each family in the catch samples for each sampled trip of the landing survey (Fig. 2; step C1). To account for the variability in sampling effort across villages and gear types, we then computed the average catch weight per fishing trip for each unique combination of fish family, gear type, and village (Fig. 2; step C2). We finally aggregated the extrapolated annual catch weight of each fish family across all gear types and villages and calculated their respective percentage contribution to the estimated total catch (Fig. 2; step C3). The most abundant families, contributing over 50% of the total annual catch, were identified as “dominant families”.
The spatial distribution of annual catches – both total and by dominant families – was then mapped within the fishery using the participatory catch survey dataset and the landing survey dataset, respectively. The mean catch-per-unit-of-effort (CPUE, in kg·h−1) of all species and the dominant families was first estimated for each village and gear type (Fig. 2; step C4) by combining the participatory catch data and landing survey data, respectively (cf. step C1), with the processed tracking dataset (cf. step E1). We filtered to retain only trips with complete fishing trips with no missing data on fishing time and catch, including zero-catch records (Fig. 2; step C4a). Then, we estimated total CPUE per village and gear type using two-part hurdle generalised linear mixed models (Fig. 2; step C4b). Random-effect hurdle models are commonly used to account for zero-inflated distribution and right skewness in fisheries data (catch per trip) to generate reliable estimates of fish catch within a haul (e.g., Cantoni et al., 2017). Such models include two random processes. The first component of the GLMM used a binomial distribution to predict the probability of non-zero catch while the second component used a Gamma distribution with a log link function to model the positive, non-zero CPUE values. Villages and gear types were included as fixed effects of the models. Individual fishers were included as a random intercept effect to account for repeated measurements over time, since total catch measurements were repeated among multiple sampled fishers over time as described in the participatory survey. For model selection and validation, we assessed the significance of the hurdle gamma model for total CPUE estimates using analysis of variance (ANOVA). The binomial and the gamma model outputs were multiplied to estimate CPUE for each village-gear type combination. Model fit was evaluated using the R-squared and root mean squared error (RMSE) by comparing observed mean catch rates of all reef fishes in each sampling stratum (village x gear type).
The mean CPUE (in kg·h−1) in each stratum (cf. step C4) was multiplied by the corresponding extrapolated annual fishing effort (in h) i) for that village and gear type (cf. step E3) and ii) within each 250 m × 250 m grid cell (cf. step E4) to compute annual catch estimates (in kg) for that stratum in total (Fig. 2; step C5a) and per cell (Fig. 2; step C5b), respectively. Finally the annual catch estimates per cell were multiplied by the mean percentage contribution of each dominant family to the annual catches for that village and gear type (cf. step C3), i.e., assuming a homogeneous catch composition at the family level in each stratum (Fig. 2; step C5c). Spatial annual catches in total and at the family level were then derived by summing up the respective estimates across villages and gear types in each cell. These spatial catch estimates per cell were divided by 6.25 ha to convert to standardised catch rates (in kg·ha−1·year−1).
Statistical analyses and data manipulations were performed using R statistical software (R Core Team, 2024). The primary packages used were adehabitatLT R (Calenge, 2006) to interpolate boat positions at a regular 1 min intervals, geosphere and ggdist for estimating travel distance, glmmTMB (Brooks et al., 2017) for mixed-effects modelling, dplyr (Wickham et al., 2023) for data manipulation, and ggplot2 (Wickham, 2016) for visualisation. Model diagnostics were performed using the car (Fox and Weisberg, 2018) and DHARMa (Hartig, 2024) packages.
3 Results
3.1 Spatial distribution of fishing effort
A total of 7,359 trips by 521 boats (58% of all boats) were spatially monitored during the survey period (Tab. 1), representing an average of 14 tracks per boat (SD = 9.4). The total annual fishing effort was estimated at 578,376 h (mean = 648 h boat−1·year−1 and 3.3 h· trip−1) and distributed over 218 km2 (mean = 2,652 h· km−2) both within and outside the bay of Toliara (72% and 28% of the fishing area, respectively; Tab. 3). Line, speargun, and gillnet fishing were widespread across the villages while the use of mosquito net trawl and beach seine was more developed in one and two villages, respectively (Fig. 3; Tab. 2). Gillnet accounted for the highest share of fishing effort (38.6%), used mainly in the three most active villages (Ankilibe, Ankiembe-bas, and Mahavatsy 1), while the other four gear types contributed between 12.9−18.1% of total fishing effort (Tab. 2). Overall each village contributed from 4.0% to 29.7% of annual fishing effort, which was aligned with the respective number of fishing boats recorded in each of them (Tab. 1).
Fishing occurred over 88−98% of most marine habitats (excluding mangrove) although intensity varied greatly (Tab. 3, Fig. 3F). The sea cucumber farming area was the only place where no fishing was observed within the bay of Toliara. Overall, low-intensity fishing (<1 h·ha−1·year−1 lowest quantile) was observed over 29.1% (63.4 km2) of the area fished, and ranged between 1 and 1,300 h·ha−1 elsewhere (154.7 km2, median = 6.5 h·ha−1· year−1). Fishing pressure peaked on the barrier reef (mean = 53.0 h·ha−1 · year−1, SD = 84.6) and the shallow terrace (mean = 36.3 h·ha−1· year−1, SD = 69.5) while it ranged between 8.4 h·ha−1· year−1 (SD = 17.1) and 15 h·ha−1 · year−1 (SD = 38.0) outside the bay and in the lagoon area, respectively (Fig. 3F).
Gear-specific use patterns also varied significantly across marine habitats. Gillnet, handline, and speargun were deployed over much larger areas (144 km2, 155 km2, and 115 km2, respectively) than those of mosquito net trawl (71 km²) and beach seines (61 km²) (Figs. 3A–3E). Gillnet use was the most ubiquitous, covering 48% to 60% of marine habitats (except mangroves) throughout the bay, although it was more heavily used on the barrier and intermediate reefs than in the outer reef, lagoon, and shallow terrace (Fig. 3A). Handline effort showed similar widespread, although scattered, spatial patterns as those of gillnet use (Fig. 3B). Spearfishing was concentrated on the barrier reef and the outer slope, with only occasional use in the lagoon and shallow terrace zones (Fig. 3C). Mosquito net trawl and beach seine were mostly used on soft bottoms habitats of reef flats and seagrass beds (Figs. 3D–3E).
Mean fishing frequency was 16.5 fishing trip·fisher−1· month−1. Overall, the average travel distance (3.5 km) and time (1.25 h) to fishing grounds were highly variable between villages and gear types (ANOVA, p<0.05; Tab. 4). Villages’ fishing zones varied in size (39–164 km2, representing 17−72% of the total area) and up to a 14−km distance from their respective landing sites (Figs. 4A–4H; Tab. 4). Tracking showed that only a few fishers occasionally undertook multi-day, long-distance fishing trips southwards and northwards over the survey period.
The villages deployed varying fishing pressure over their respective fishing grounds, highlighting preferential target areas over the shallow terrace and the barrier reefs (Figs. 4A–4H). Ankilibe had the largest high-intensity fishing zone (>20 h·ha−1) compared to other villages (Fig. 4G).
Fishing zones were unevenly distributed among villages. Most villages had overlapping fishing areas within the bay of Toliara while about 36% of the total fishing area was exclusively targeted by fishers from a single village (Fig. 4I). Indeed, both rural villages (villages #7 and #8, Fig. 6I) and two urban villages (villages #2 and #6, Fig. 6I) showed large exclusive fishing areas (about 30 km2, 11 km2, 23 km2, and 12 km2, respectively; Tab. 2). These zones were located either on the shallow terrace up to (4−6 km from rural villages) or on the outer barrier reef (8−14 km distance from urban villages).
Total and relative distribution of estimated fishing effort (in h) by village and gear type in the study area. The extent of total fishing area and exclusive fishing area for each village (in km2) are indicated. The proportion of fishing area by village is estimated compared to the total fishing area (218 km2). The proportion of exclusive fishing area by village (%) is estimated compared to the total fishing area for each village. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8).
Distribution of fishing pressure and fish catches across habitat types in the study area. Data include total fishing area (% of total per habitat between brackets), low-intensity fishing area (% of total per habitat between brackets), and average fishing pressure (standard deviation between brackets) in each habitat. Data about fish catches include average and median catch rates (standard deviation between brackets) and total and relative (% of total between brackets) catches in each habitat.
 |
Fig. 3 Spatial distribution of fishing effort by fishing gear type and in total (F) in the coral reef fishery in the study area. The fishing villages are indicated: Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8). |
Accessibility to fishing grounds by village in the bay of Toliara as derived from the fishing tracks of the observational dataset. Fishing trip characteristics include mean and median travel time (in h), distance (in km), and speed (in km·h−1) to fishing grounds along with standard deviations (SD), and the minimum and maximum distance travelled (in km). Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Ankilibe (7), and Sarodrano (8).
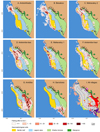 |
Fig. 4 Spatial distribution of annual fishing effort by village (A-H; in kg·ha−1) and spatial extent of villages’ fishing grounds (I) in the study area. Shared fishing areas (I, in grey) and villages’ exclusive fishing areas (I, in distinct colours) are shown. Numbers refer to Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8) villages’ fishing areas. |
3.2 Catch composition and distribution
Total annual reef fish catches were estimated at 1,466 tonnes, corresponding to an average of 1.6 t·boat−1·year−1 and 2.5 kg·h−1 based on total fishing effort estimates. Overall CPUE significantly varied across gear types (Type II ANOVA, χ2(4)=306.734, p<0.001) and villages (Type II ANOVA, χ2(7)=88.215, p<0.001). Mosquito net trawl and beach seine provided highest average CPUE estimates (5.9−7.3 kg·h−1 and 2.0−3.5 kg·h−1; Tab. 2) while handline CPUE yielded the lowest (0.3–1.5 kg·h−1; Tab. 2). Fish catch was very unevenly distributed by village, ranging between 1.7% and 44.5% of the annual total (Tab. 2). Annual catch rates varied considerably across habitats, ranging from <1 kg·ha−1 over one third of the fishing area to 1,560 kg·ha−1 (mean = 48.4 kg·ha−1) (Fig. 5 H; Tab. 3). The average catch rate was 10 times higher over the shallow terrace (mean = 102 kg · ha−1· year−1, SD = 189) and the barrier reefs (mean = 86.1 kg·ha−1. year−1, SD = 139) than in open waters (8.7 kg·ha−1· year−1, SD = 32.7) and the lagoon area (23.0 kg·ha−1· year−1, SD = 58.9). Altogether, 82% of the catches occurred over the terrace (653 t·year−1, 53%) and barrier reefs (362 t·year−1, 29%), while the open water and lagoon areas accounted for 82 t·year−1(6.6%) and 136 t·year−1(11%), respectively.
The seven dominant families accounted for 62% of total annual catches, as follows: Labridae (17.7%), Scaridae (11.5%), Acanthuridae (9.5%), Lethrinidae (7.5%), Siganidae (5.7%), Gobiidae (5.4%), and Mullidae (4.4%) (Fig. 5). Those families were targeted by all gear types except the Gobiidae, which were harvested by fishers using mosquito net trawls and beach seines. The distribution of catch rates of the dominant families followed two main spatial patterns associated with reef geomorphology and gear use (Fig. 6). The Gobiidae were harvested within the shallow terrace and, to a lower extent, the seagrass beds of the great barrier reef (Fig. 6E), while the catches of the other families were widespread throughout the fishing area (Figs. 6A–6D, 6F and 6G). Annual catch rates of dominant families generally ranged from 10 kg·ha−1 to 50 kg·ha−1 with Labridae and Gobiidae reaching higher levels (50–200 kg·ha−1) over a large part of the areas fished (Figs. 6A and 6E).
 |
Fig. 5 Relative contribution (%) of the 75 reef fish families in estimated annual catches in the study area. The seven dominant families (62% of total catches) are shown (in pink): Labridae, Scaridae, Acanthuridae, Lethrinidae, Gobiidae, and Mullidae. |
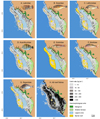 |
Fig. 6 Distribution of annual catch rates (kg·ha−1) of the dominant target reef fish families (AG) and in total (H) in the study area. Numbers refer to Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8) villages. |
4 Discussion
We combined GPS tracking data with catch monitoring data through a 1 yr extensive survey of the multi-gear, multispecies fishery of the Bay of Toliara in southwestern Madagascar, that supports the livelihoods of hundreds of fishing households. Spatial analysis revealed the allocation of fishing effort and reef fish catches at fine spatial scale and allowed for characterizing fishing pressure and catch rates among villages, gear types, and reef geomorphology units accurately. Map outputs provided critical new insights into preferential fishing areas, overexploitation patterns, and marine space uses.
4.1 Drivers of fishers’ behaviour at sea
We observed that environmental, technological, and social interactions structured fishers’ behaviour at sea in the study area. Our study revealed fine-scale spatial use of marine areas, highlighting villages’ shared and exclusive fishing grounds, which challenged the common perception of the bay of Toliara as communal and open-access (Bruggemann et al., 2012; Baker-Médard and Kroger, 2023). More specifically, we found that four out of eight villages have established large exclusive fishing areas up to 25% of their community fishing grounds. These exclusive areas were strategically located nearby southernmost rural villages while the fishers of both urban villages have established their respective exclusive fishing areas outside the bay of Toliara, at a larger distance from their place of residence (Fig. 4). Those patterns suggest tacit spatial appropriation of specific fishing grounds, which is a critical component of informal territoriality (Turner et al., 2013). This may result from a range of territorial behaviours influenced by gear type, competition, and enforcement cost and capacity (Begossi, 2001). For instance, Ankilibe’s fishers, i.e. the largest fishing community group in the area, can hardly access open waters through the southern and northern passes due to the village location. Since the 2000s, they have intensively used mosquito net trawl in the nearby seagrass beds and excluded fishers from other villages, which allows for high annual catch levels of juvenile fishes (Ranaivomanana et al., 2023) and benthic crustaceans (Herinirina et al., 2023). This suggests that they have actively claimed this easily-accessed productive area as a “nucleated” territory to secure harvest of seagrass-associated resources and income (Acheson, 1988). The fact that no other gear is used by this community in this restricted area further suggests that the latter is actually beneficial to a small fisher group of the village (mosquito net trawl users).
Overall, the spatial organization of fishing activities provide evidence that communities have self-regulated access to shared and territory fishing grounds as driven by technological constraints, social relationships at varying scale (i.e., among and within villages), and fishers’ reliance on coral reef resources. For instance, defending village-based fishing territories in the middle of the bay and close to Toliara urban area may not have been possible or worthwhile due to the high number of villages close one to another and/or the distance of the fishing areas (Acheson and Gardner, 2004). In this context, alternative forms of perimeter-defended territories associated with specific groups of fishers from a single or multiple village(s) have emerged, based on technological and social spatial factors (King, 2019). Handline fishers of two other urban villages (Besakoa and Ankiambe-haut) have established fishing territories in the deeper waters alongside the barrier reef slope that are not accessible to other gear types, while beach seiners from three urban villages (Mahavatsy 2, Ankiembe-bas, and to a lower extent, Mahavatsy 1; Tab. 1) were the exclusive user group of most of the shallow terrace nearby the city of Toliara (Figs. 3 and 4). The fishery maps revealed gear- and village-based communities of practice, which shed light on the undocumented local marine tenure that convey fishers’ sense of place (Randriatsara et al., 2025). This self-organization may foster the resilience of the fishery system so as to accommodate ongoing resource and catch decline as described below (Berkes and Turner, 2006). Further anthropological research is needed to understand the origins and functions of these informal rules within and among communities and that shape everyday fishing practices, including its potential implications for collaborative fishery management and marine spatial planning.
4.2 Spatial resource overexploitation and management issues
Fine-scale spatial catch distribution highlights significant overexploitation of the fishery, as shown by different key indicators. Firstly, the average annual catch rate declined from 12 t. km−1· year−1 in the 1990s (Laroche and Ramananarivo, 1995) to 6.7 t· km−1· year−1(−44%), which is close to the recovery threshold (5–15 t· km−1. year−1) commonly suggested in coral reef ecosystem (Spalding et al., 2001; Newton et al., 2007). Secondly, annual catches declined by about 30% despite a 25% increase in fisher numbers and the growing use of efficient gear types (Laroche and Ramananarivo, 1995). Indeed gillnet, mosquito net trawl, and beach seine are now the dominant gears in terms of total fishing effort and catches and provided fishing yields one order of magnitude larger than those of handline use, that showed significant decline over time. Thirdly, gear diversification led to a >100% increase in the number of reef fish families harvested, since Laroche and Ramananarivo’s 1995 survey, which identified 35 families in catches. Catch diversification is commonly observed in coastal fisheries facing serial overexploitation as a harvest strategy to maintain catch level and income (Robinson et al., 2020). For instance, low economic value and small fish species, such as Apogonidae, Blenniidae, and Gobiidae are now harvested by small-mesh gears in shallow seagrass areas as described above and used for self-consumption or sale (Herinirina et al., 2023; Ranaivomanana et al., 2023). Increased fishing effort has failed to offset the decline in resource abundance, resulting in fishers getting a decreasing annual catch level and share.
Fishers’ behaviour was constrained by limited boat fishing power and flexibility, which has likely shaped territoriality and exacerbated the overexploitation process as described above. Fishers mostly performed short travel distances and daily trips limited to the terrace-lagoon-reef complex within or nearby the bay of Toliara, although rare distant trips northwards and southwards were tracked during the survey period. Globally, improvements in vessel power have been central to fishing success to increase the amount of gear onboard, fishing frequency, crew number, trip duration, choice of fishing location, and/or access to distant fishing grounds including offshore waters (e.g., Engelhard, 2016). In contrast, traditional sailing pirogues in Southern Madagascar have smaller capacity and flexibility than motorised fishing boats, e.g., the speed, distance, and direction of fishing trips depended on the combination of wind direction and strength and one’s place of residence. As a result, fishing frequency remained low (about every other day) while the geographical extent of the fishery outside the bay of Toliara (Tab. 3) has been marginal since the 1990s (Laroche and Ramananarivo, 1995). Lack of technical innovation in fishing boats may stem from limited financial capital and poor access to public loan, which are strong determinants of fishers’ investment and adaptive capacities (Hallén et al., 2023). Alternatively, fishers have intensified and diversified gear technology to target new reef fish species as well as valuable invertebrates and pelagic fish in surrounding areas as described above (Maka et al., 2022; Herinirina et al., 2023). Evidence also suggests that some fishers temporarily migrate to other regions in search of more abundant commercial resources, such as sharks or sea cucumbers (Muttenzer, 2015; Cripps and Gardner, 2016). Further research is needed to determine whether local fishers are caught in an overexploitation trap, to help better characterise social ecological interactions, and to understand fishers’ coping and exit strategies and opportunities.
This study confirmed that fine-scale spatial analysis of fishery data advances the knowledge base for designing spatially-explicit tools for the use of the marine environment. Catch and effort maps quantify losses and identify which fishers are most affected in the development of maritime activities alternative to fishing. Specifically, we found that the privatized enclosures for sea cucumber farming were surrounded by the Ankilibe village’s territory fishing area, suggesting disproportionately affected local fishers within that community and particularly mosquito net trawl fishers. Catch rates for key species near the privatized site were low suggesting that the resulting fishers’ catch and associated income loss was likely low, but still significant in the local context of poverty. From this perspective, sea cucumber farming may have led to marine space dispossession, which was interpreted as the cause of thefts and violence in recent years (Baker-Médard and Kroger, 2023). Our spatial analysis can help quantify the fishery-related costs of aquaculture and identify who bears them, as compared to the level and distribution of socioeconomic benefits between and within the neighbouring fishing communities (Lavitra et al., 2024). Such detailed spatial knowledge may therefore facilitate reconciliating fishers’ livelihoods and informal local tenure with aquaculture development and fishery sustainability targets (e.g., conserving fish nursery seagrass areas currently harvested by beach seines and mosquito net trawls). More broadly, such analysis can support more equitable and inclusive marine spatial planning so as to engage marginalized actors such as small-scale fisher communities, as demonstrated in other coastal zones worldwide (Trouillet 2019; Chollett et al., 2022; Papageorgiou et al., 2024). The challenges and complexity of the social ecological system within the Bay of Toliara cannot however be underestimated.
4.3 Up-scaling tracking surveys in small-scale fisheries
This is, to our knowledge, the first study to map annual fishing effort (in h·ha−1) and catch rates (in kg·ha−1) using GPS tracking data in a small-scale fishery in the Global South. Reef fisheries in particular exhibit high diversity in gear use, species, and target marine habitats in these countries, such as in the WIO marine biodiversity hotspot (McClanahan, 2015), which raises multiple monitoring challenges (e.g., routine visual species identification). Replicating this approach elsewhere and at larger scales will require tailored technical and logistical strategies. Given the spatial temporal heterogeneity of small-scale fishery activities, intensive data collection was required for capturing the distribution of fishers’ movements and catch rates representatively (e.g., 64% of fishing boats were sampled over 1 yr). By contrast previous GPS-based small-scale fishery monitoring surveys were not generalized temporally or acquired vessel positioning data at lower temporal resolution. Small boats typically cannot carry expensive equipment like VMS or AIS, but recent alternatives have made tracking feasible in small-scale fisheries (e.g., Galotto-Tébar et al., 2022; Tassetti, et al. 2022). Furthermore, the statistical processing of large tracking dataset may be automated (Guitton and Soumah, 2023; Tilley and al., 2024), which should facilitate the broader adoption of this technology without expertise in complex statistical analysis. Further research should however explore spatial inference methods (e.g., geostatistics) to produce map representations of fishing intensity and catch gradients based on tracking data (e.g., Thorson et al., 2015; Le Guyader et al., 2017).
To simultaneously collect tracking and catch data from a large sample of fishers as described above, fishers’ participation was required. Participatory monitoring offers multiple benefits from community engagement to cost-effective data collection, such as engaging communities in marine resource management, producing low-cost data for data-poor fisheries, and empowering fishing communities through learning (e.g., Hind, 2014; Stephenson et al., 2016; House et al., 2023). In order to collect GPS tracking data on a large scale, the tracking device operating mode, the data actually recorded, and the results produced should be clarified to local fishers and communities as part of a transparent, collaborative approach. When fishers are trained to collect data, they can lower costs and enhance data quality for fisheries assessments and management, particularly for collecting sensitive spatial temporal data via on-board GPS (Méndez-Espinoza et al., 2020). This participatory protocol may offer a scalable, cost-effective model for small-scale fisheries, particularly in developing countries, where operational costs are low.
5 Conclusion
We investigated the fine-scale spatial distribution of fishing activities in a small-scale coral reef peri-urban fishery in southwestern Madagascar. The fine-scale resolution maps of fishing effort and catches revealed informal, gear- and village-based tenure as well as strong overexploitation patterns. These findings offer critical insights for fishery management, marine conservation (e.g., fish nursery seagrass areas), inclusive marine spatial planning, and participatory governance involving small-scale fisher communities in a heavily-used coastal area. This study underscores the potential of boat tracking and catch surveys as powerful tools for small-scale fisheries assessment through participatory research. Further anthropological research is needed to deepen our understanding of social interactions driving the informal spatial fishing behaviours and arrangements.
Acknowledgments
This work was funded by the French institute for sustainable development (IRD) as part of the IH.SM-IRD research partnership (JEAI-ACOM), the MIKAROKA international research laboratory, the Laboratory of Excellence CORAIL, France (GEOFIA and MANJONO grants), and the Critical Ecosystem Partnership Fund (CEPF-IH.SM, grant N° 66341). We thank Dufrehino, Tsipy Romano, and Jean-Jacques Marcelin for their help with data collection. We thank local assistants, the fokontany chiefs, and the fishers who volunteered to participate in the survey. We thank Emma Gibbons from Reef Doctor for her kind and careful proofreading of the original manuscript. Fish photo credit (Fig. 6): Anissa Volanandiana.
Data availability statement
No new data/codes were created or analyzed in this study.
References
- Acheson JM. The Lobster Gangs of Maine, University Press of New England, 1988. [Google Scholar]
- Acheson JM, Gardner RJ. 2004. Strategies, conflict, and the emergence of territoriality: the case of the Maine lobster industry. Am Anthropol 106: 296–307. [Google Scholar]
- Andrefouët S, Guillaume M, Delval A, Rasoamanendrika F, Blanchot J, Bruggemann H. 2013. Fifty years of changes in reef flat habitats of the Grand R, cif of Toliara (SW Madagascar) and the impact of gleaning. Coral Reefs 32: 757–768 [Google Scholar]
- Andrialovanirina N, Ponton D, Behivoke F, Mahafina J, Léopold M. 2020. A powerful method for measuring fish size of small-scale fishery catches using Image J. Fish Res 223:105425. [Google Scholar]
- Baker-Médard M, Kroger E. 2023. Troubling the waters: gendered dispossession, violence, and sea cucumber aquaculture in Madagascar. Soc Nat Resour 0: 1–28. [Google Scholar]
- Basurto X, Gutierrez NL, Franz N, Mancha-Cisneros MDM, Gorelli G, Aguión A, Funge-Smith S, Harper S, Mills DJ, Nico G, Tilley A, Vannuccini S, Virdin J, Westlund L, Allison EH, Anderson CM, Baio A, Cinner J, Fabinyi M, Hicks CC, Kolding J, Melnychuk MC, Ovando D, Parma AM, Robinson JPW, Thilsted HS. 2025. Illuminating the multidimensional contributions of small-scale fisheries. Nature 637: 875–884. [Google Scholar]
- Bauchot M-L, Bianchi G. Fiches FAO d’identification des espèces pour les besoins de la pêche. Guide des poissons commerciaux de Madagascar (espèces marines et d’eaux saumâtres). FAO, Rome, 1984. [Google Scholar]
- Beckensteiner J, Boschetti F, Thébaud O. 2023. Adaptive fisheries responses may lead to climate maladaptation in the absence of access regulations. npj Ocean Sustain 2: 1–5. [Google Scholar]
- Begossi A. 2001. Mapping spots: fishing areas or territories among islanders of the Atlantic Forest (Brazil). Reg Environ Change 2: 1–12. [Google Scholar]
- Behivoke F, Etienne M-P, Guitton J, Randriatsara RM, Ranaivoson E, Léopold M. 2021. Estimating fishing effort in small-scale fisheries using GPS tracking data and random forests. Ecol Indic 123:107321. [CrossRef] [Google Scholar]
- Bennett NJ, Govan H, Satterfield T. 2015. Ocean grabbing. Mar Policy 57: 61–68. [Google Scholar]
- Berkes F, Turner NJ. 2006. Knowledge, learning and the evolution of conservation practice for social-ecological system resilience. Human Ecol 34: 479–494. [Google Scholar]
- Blythe JL, Gill DA, Claudet J, Bennett NJ, Gurney GG, Baggio JA, Ban NC, Bernard ML, Brun V, Darling ES. 2023. Blue justice: a review of emerging scholarship and resistance movements. Cambridge Prisms: Coastal Futures 1: e15. [Google Scholar]
- Brooks ME, Kristensen K, Van benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Maechler M, Bolker BM. 2017. Balances speed and flexibility among packages for zeroinflated generalized linear mixed modeling. The R Journal 9: 378–400. [Google Scholar]
- Bruggemann JH, Rodier M, Guillaume MMM, Andréfouët S, Arfi R, Cinner JE, Pichon M, Ramahatratra F, Rasoamanendrika F, Zinke J, McClanahan TR. 2012. Wicked social-ecological problems forcing unprecedented change on the latitudinal margins of coral reefs: the case of Southwest Madagascar. Ecol Soc 17:47. [Google Scholar]
- Burgos C, Gil J, del Olmo LA. 2013. The Spanish blackspot seabream (Pagellus bogaraveo) fishery in the Strait of Gibraltar: spatial distribution and fishing effort derived from a smallscale GPRS/GSM based fisheries vessel monitoring system. Aquat Living Resour 26: 399–407. [Google Scholar]
- Calenge C. 2006. The package “adehabitat” for the R software: A tool for the analysis of space and habitat use by animals. Ecol Modell 197: 516–519. [Google Scholar]
- Cantoni E, Flemming JM, Welsh AH. 2017. A random-effects hurdle model for predicting bycatch of endangered marine species. Annals Appl Stat 11: 2178–2199. [Google Scholar]
- Chollett I, Perruso L, O’Farrell S. 2022. Toward a better use of fisheries data in spatial planning. Fish Fish 23: 1136–1149. [Google Scholar]
- Close CH, Brent Hall G. 2006. A GIS-based protocol for the collection and use of local knowledge in fisheries management planning. J Environ Manag 78: 341–352. [Google Scholar]
- Cohen PJ, Allison EH, Andrew NL, Cinner J, Evans LS, Fabinyi M, Garces LR, Hall SJ, Hicks CC, Hughes TP, Jentoft S, Mills DJ, Masu R, Mbaru EK, Ratner BD. 2019. Securing a just space for small-scale fisheries in the blue economy. Front Mar Sci 6:171. [Google Scholar]
- Couclelis H. 2003. The certainty of uncertainty: GIS and the limits of geographic knowledge. Trans GIS 7: 165–175. [Google Scholar]
- Cripps G, Gardner CJ. 2016. Human migration and marine protected areas: insights from Vezo fishers in Madagascar. Geoforum 74: 49–62. [Google Scholar]
- Engelhard GH. On the need to study fishing power change: challenges and perspectives, in: K. Schwerdtner Máñez, B. Poulsen (Eds.), Perspectives on Oceans Past. Dordrecht, Springer Netherlands, 2016, pp. 89–101. [Google Scholar]
- Fernández-Boán M, Freire J, Parma AM, Fernández L, Orensanz JM. 2013. Monitoring the fishing process in the sea urchin diving fishery of Galicia. ICES J Mar Sci 70: 604–617. [Google Scholar]
- Fox J, Weisberg S. An R Companion to Applied Regression, SAGE Publications, 2018. [Google Scholar]
- Fricke R, Mahafina J, Behivoke F, Jaonalison H, Léopold M, Ponton D. 2018. Annotated checklist of the fishes of Madagascar, southwestern Indian Ocean, with 158 new records. Fish Taxa 3. [Google Scholar]
- Galotto-Tébar MM, Pomares-Padilla A, Czerwinski IA, Gutiérrez-Estrada JC. 2022. Is the vessel fishing? Discrimination of fishing activity with low-cost intelligent mobile devices through traditional and heuristic approaches. Expert Syst Appl 200:117091. [Google Scholar]
- Gerritsen H, Lordan C. 2011. Integrating vessel monitoring systems (VMS) data with daily catch data from logbooks to explore the spatial distribution of catch and effort at high resolution. ICES J Mar Sci 68: 245–252. [CrossRef] [Google Scholar]
- Gill DA, Blythe J, Bennett N, Evans L, Brown K, Turner RA, Baggio JA, Baker D, Ban NC, Brun V, Claudet J, Darling E, Franco AD, Epstein G, Estradivari, Gray NJ, Gurney GG, Horan RP, Jupiter SD, Lau JD, Lazzari N, Lestari P, Mahajan SL, Mangubhai S, Naggea J, Selig ER, Whitney CK, Zafra-Calvo N, Muthiga NA. 2023. Triple exposure: reducing negative impacts of climate change, blue growth, and conservation on coastal communities. One Earth 6:118130. [Google Scholar]
- Gill DA, Cheng SH, Glew L, Aigner E, Bennett NJ, Mascia MB. 2019. Social synergies, tradeoffs, and equity in marine conservation impacts. Ann Rev Environ Resour 44: 347–372. [Google Scholar]
- Grantham HS, Wilson KA, Moilanen A, Rebelo T, Possingham HP. 2009. Delaying conservation actions for improved knowledge: how long should we wait? Ecol Lett 12:293301. [Google Scholar]
- Grati F, Azzurro E, Scanu M, Tassetti AN, Bolognini L, Guicciardi S, Vitale S, Scannella D, Carbonara P, Dragičević B, Ikica Z, Palluqi A, Marčeta B, Ghmati H, Turki A, Cherif M, Bdioui M, Jarboui O, Benhadjhamida N, Mifsud J, Milone N, Ceriola L, Arneri E. 2022. Mapping small-scale fisheries through a coordinated participatory strategy. Fish Fish 23: 773–785. [Google Scholar]
- Guillemot N, Léopold M, Cuif M, Chabanet P. 2009. Characterization and management of informal fisheries confronted with socio-economic changes in New Caledonia (South Pacific). Fish Res 98: 51–61. [Google Scholar]
- Guitton J, Soumah M. 2023. GPSMonitoring: A R package for small scale fisheries monitoring using GPS logger. Softw Impacts 17:100573. [Google Scholar]
- Hallén P, Bornmalm L, Alexandersson H. 2023. The power of fishing: the introduction of steam and motorization in Scandinavian fishing, 1880–1950. Int J Marit Hist 35: 454–474. [Google Scholar]
- Hartig F. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models, 2024. [Google Scholar]
- Herinirina PA, Bigot L, Frouin P, Volandrae HTE, Randriatsara RM, Behivoke F, Ranivoarivelo L, Léopold M. 2023. Seagrass macroinvertebrate bycatches support mosquito net trawl fishery in Madagascar. Aquat Living Resour 36:18. [Google Scholar]
- Hind EJ. 2014. A review of the past, the present, and the future of fishers’ knowledge research: a challenge to established fisheries science. ICES J Mar Sci 72: 341–358. [Google Scholar]
- House J, Kleiber D, Steenbergen DJ, Stacey N. 2023. Participatory monitoring in community-based fisheries management through a gender lens. Ambio 52: 300–318. [Google Scholar]
- Janßen H, Bastardie F, Eero M, Hamon KG, Hinrichsen H-H, Marchal P, Nielsen JR, Le Pape O, Schulze T, Simons S, Teal LR, Tidd A. 2018. Integration of fisheries into marine spatial planning: Quo vadis? Estuarine, Coastal and Shelf Science, Vectors of change in the marine environment 201: 105–113. [Google Scholar]
- King TJ. Formal and informal territoriality in ocean management, in: T.J. King, G. Robinson (Eds.), At Home on the Waves: Human Habitation of the Sea from the Mesolithic to Today, Berghahn Books, 2019, pp. 332–348. [Google Scholar]
- Laroche J, Ramananarivo N. 1995. A preliminary survey of the artisanal fishery on coral reefs of the Tulear Region (southwest Madagascar). Coral Reefs 14: 193–200. [CrossRef] [Google Scholar]
- Lavitra T, Moridy F, Rabearison M, Rodine C, Rakotomahazo C, Nomenisoa A, Ranivoarivelo N, Rasolofonirina R, Rakotoarimanana A, Franberg C, Troell M, Eeckhaut I, Boleslas G, Todinanahary G. 2024. Local perceptions of the socioeconomic and environmental impacts of sea cucumber farming in southwestern Madagascar. SPC Beche-de-mer Inf Bull 44: 48–58. [Google Scholar]
- Le Guyader D, Ray C, Gourmelon F, Brosset D. 2017. Defining high-resolution dredge fishing grounds with Automatic Identification System (AIS) data. Aquat Living Resour 30:39. [Google Scholar]
- Lee J, South AB, Jennings S. 2010. Developing reliable, repeatable, and accessible methods to provide high-resolution estimates of fishing-effort distributions from vessel monitoring system (VMS) data. ICES J Mar Sci 67: 1260–1271. [Google Scholar]
- Léopold M, Guillemot N, Rocklin D, Chen C. 2014. A framework for mapping small-scale coastal fisheries using fishers’ knowledge. ICES J Mar Sci 71: 1781–1792. [Google Scholar]
- Maka O, Pascal B, Todinanahary G. 2022. Wild sea cucumber fishery in southwestern Madagascar. SPC Beche-de-mer Inf Bull 42: 36–45. [Google Scholar]
- McClanahan TR. 2015. Biogeography versus resource management: how do they compare when prioritizing the management of coral reef fish in the south-western Indian Ocean? J Biogeogr 42: 2414–2426. [Google Scholar]
- McCluskey SM, Lewison RL. 2008. Quantifying fishing effort: a synthesis of current methods and their applications. Fish Fish 9: 188–200. [Google Scholar]
- Méndez-Espinoza D, Ojeda-Ruiz MÁ, Marín-Monroy EA, Jiménez-Esquivel V, Cota-Nieto JJ. 2020. Participatory research to understand spatio-temporal dynamics of small-scale fleets: the C. bellicosus fishery in Magdalena Bay, Baja California Sur, Mexico. Ocean Coast Manag 198:105369. [Google Scholar]
- Muallil RN, Mamauag SS, Cababaro JT, Arceo HO, Aliño PM. 2014. Catch trends in Philippine small-scale fisheries over the last five decades: the fishers’ perspectives. Mar Policy 47:110117. [Google Scholar]
- Muttenzer F. 2015. The social life of sea-cucumbers in Madagascar: migrant fishers display of a livelihood ethos 27: 101–121. [Google Scholar]
- Navarrete Forero G, Miñarro S, Mildenberger TK, Breckwoldt A, Sudirman, Reuter H. 2017. Participatory boat tracking reveals spatial fishing patterns in an Indonesian artisanal fishery. Front Mar Sci 4. [Google Scholar]
- Palmer JL, Armstrong C, Akbora HD, Beton D, Çağlar Ç, Godley BJ, Metcalfe K, Özkan M, Snape RTE, Broderick AC. 2024. Behavioural patterns, spatial utilisation and landings composition of a small-scale fishery in the eastern Mediterranean. Fish Res 269:106861. [Google Scholar]
- Papageorgiou M, Tourapi C, Nikolaidis G, Petrou A, Moutopoulos DK. 2024. Profiling the Cypriot fisheries sector through the lens of fishers: a participatory approach between fishers and scientists. Fishes 9:308. [Google Scholar]
- Pellowe KE, Leslie HM. 2020. The interplay between formal and informal institutions and the potential for co-management in a Mexican small-scale fishery. Mar Pol 121: 104179. [Google Scholar]
- Prince J, Hordyk A. 2019. What to do when you have almost nothing: A simple quantitative prescription for managing extremely data-poor fisheries. Fish Fish 20: 224–238. [Google Scholar]
- Ranaivomanana HS, Jaquemet S, Ponton D, Behivoke F, Randriatsara RM, Mahafina J, Léopold M. 2023. Intense pressure on small and juvenile coral reef fishes threatens fishery production in Madagascar. Fish Manag Ecol 30: 494–506. [Google Scholar]
- Randriatsara RM, Behivoke F, Ramahatratra F, Mahafina J, Lamy T, Léopold M. 2025. Mapping place names for small-scale fisheries evaluation. Aquat Living Resour 38:13. [Google Scholar]
- Rufino MM, Mendo T, Samarão J, Gaspar MB. 2023. Estimating fishing effort in small-scale fisheries using high-resolution spatio-temporal tracking data (an implementation framework illustrated with case studies from Portugal). Ecol Indic 154:110628. [Google Scholar]
- Scholz AJ, Steinback C, Kruse SA, Mertens M, Silverman H. 2011. Incorporation of spatial and economic analyses of human-use data in the design of marine protected areas. Conserv Biol 25: 485–492. [Google Scholar]
- Stephenson RL, Paul S, Pastoors MA, Kraan M, Holm P, Wiber M, Mackinson S, Dankel DJ, Brooks K, Benson A. 2016. Integrating fishers’ knowledge research in science and management. ICES J Mar Sci: J Conseil 73: fsw025. [Google Scholar]
- Taquet M, Diringer A. Fishes of the Indian Ocean and Red Sea. Editions Quae, 2012. [Google Scholar]
- Tassetti AN, Ferrà C, Fabi G. 2019. Rating the effectiveness of fishery-regulated areas with AIS data. Ocean Coast Manag 175: 90–97. [Google Scholar]
- Tassetti AN, Galdelli A, Pulcinella J, Mancini A, Bolognini L. 2022. Addressing gaps in smallscale fisheries: a low-cost tracking system. Sensors 22:839. [Google Scholar]
- Thiault L, Collin A, Chlous F, Gelcich S, Claudet J. 2017. Combining participatory and socioeconomic approaches to map fishing effort in small-scale fisheries. PLOS ONE 12: e0176862. [Google Scholar]
- Thorson JT, Shelton AO, Ward EJ, Skaug HJ. 2015. Geostatistical delta-generalized linear mixed models improve precision for estimated abundance indices for West Coast groundfishes. ICES J Mar Sci 72: 1297–1310. [Google Scholar]
- Thoya P, Pérez-Jorge S, Okemwa GM, Mwamlavya H, Tuda A, Wambiji N, Maina J. 2020. Spatial patterns and environmental risks of ringnet fishing along the Kenyan coast. Afr J Mar Sci 42: 23–33. [Google Scholar]
- Tilley A, Dam Lam R, Lozano Lazo D, Dos Reis Lopes J, Freitas Da Costa D, De Fátima Belo M, Da Silva J, Da Cruz G, Rossignoli C. 2024. The impacts of digital transformation on fisheries policy and sustainability: Lessons from Timor-Leste. Environ Sci Policy 153:103684. [Google Scholar]
- Tilley A, Lopes JDR, Wilkinson SP. 2020. PeskAAS: A near-real-time, open-source monitoring and analytics system for small-scale fisheries. PLOS ONE 15: e0234760. [Google Scholar]
- Trouillet B. 2019. Aligning with dominant interests: the role played by geo-technologies in the place given to fisheries in marine spatial planning. Geoforum 107: 54–65. [Google Scholar]
- Turner RA, Gray T, Polunin NVC, Stead SM. 2013. Territoriality as a driver of fishers’ spatial behavior in the Northumberland lobster fishery. Soc Nat Resour 26: 491–505. [Google Scholar]
- Watson JT, Haynie AC, Sullivan PJ, Perruso L, O’Farrell S, Sanchirico JN, Mueter FJ. 2018. Vessel monitoring systems (VMS) reveal an increase in fishing efficiency following regulatory changes in a demersal longline fishery. Fish Res 207: 85–94. [Google Scholar]
- Wickham H. ggplot2: Elegant Graphics for Data Analysis, Springer-Verlag New York, 2016. [Google Scholar]
- Wickham H, François R, Henry L, Muller K, Vaughan D. dplyr: A Grammar of Data Manipulation, 2023. [Google Scholar]
- Wilen JE. 2004. Spatial management of fisheries. Mar Resour Econ 19: 7–19. [Google Scholar]
- Zeller D, Harper S, Zylich K, Pauly D. 2015. Synthesis of underreported small-scale fisheries catch in Pacific island waters. Coral Reefs 34: 25–39. [Google Scholar]
Cite this article as: Behivoke F, D’Agata S, Guitton J, Etienne M-P, Randriatsara RM, Ranaivoson E, Mahafina J, Léopold M. 2026. Fine-scale fishery patterns reveal challenges and opportunities for coastal management and conservation in Madagascar. Aquat. Living Resour. 39: 6. https://doi.org/10.1051/alr/2025020
All Tables
Fishery and survey characteristics per village and gear type in the study area. The total number of fishing boats (% sampled in brackets), GPS tracks, catch landing records, and participatory catch records are shown. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8).
Total and relative distribution of estimated fishing effort (in h) by village and gear type in the study area. The extent of total fishing area and exclusive fishing area for each village (in km2) are indicated. The proportion of fishing area by village is estimated compared to the total fishing area (218 km2). The proportion of exclusive fishing area by village (%) is estimated compared to the total fishing area for each village. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8).
Distribution of fishing pressure and fish catches across habitat types in the study area. Data include total fishing area (% of total per habitat between brackets), low-intensity fishing area (% of total per habitat between brackets), and average fishing pressure (standard deviation between brackets) in each habitat. Data about fish catches include average and median catch rates (standard deviation between brackets) and total and relative (% of total between brackets) catches in each habitat.
Accessibility to fishing grounds by village in the bay of Toliara as derived from the fishing tracks of the observational dataset. Fishing trip characteristics include mean and median travel time (in h), distance (in km), and speed (in km·h−1) to fishing grounds along with standard deviations (SD), and the minimum and maximum distance travelled (in km). Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Ankilibe (7), and Sarodrano (8).
All Figures
 |
Fig. 1 Survey area in the bay of Toliara, southwestern Madagascar. Reef geomorphology units are shown as distinct colours. Numbers correspond to the villages with Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8). Recent fishing settlements are shown in italics. |
| In the text | |
 |
Fig. 2 Sampling and analytical procedure of fishery data used for characterizing the spatial distribution of fishing effort and catches in the coral reef fishery. |
| In the text | |
 |
Fig. 3 Spatial distribution of fishing effort by fishing gear type and in total (F) in the coral reef fishery in the study area. The fishing villages are indicated: Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8). |
| In the text | |
 |
Fig. 4 Spatial distribution of annual fishing effort by village (A-H; in kg·ha−1) and spatial extent of villages’ fishing grounds (I) in the study area. Shared fishing areas (I, in grey) and villages’ exclusive fishing areas (I, in distinct colours) are shown. Numbers refer to Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8) villages’ fishing areas. |
| In the text | |
 |
Fig. 5 Relative contribution (%) of the 75 reef fish families in estimated annual catches in the study area. The seven dominant families (62% of total catches) are shown (in pink): Labridae, Scaridae, Acanthuridae, Lethrinidae, Gobiidae, and Mullidae. |
| In the text | |
 |
Fig. 6 Distribution of annual catch rates (kg·ha−1) of the dominant target reef fish families (AG) and in total (H) in the study area. Numbers refer to Ambohitsabo (1), Besakoa (2), Mahavatsy 2 (3), Ankiembe-bas (4), Mahavatsy 1 (5), Ankiembe-haut (6), Ankilibe (7), and Sarodrano (8) villages. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


