| Issue |
Aquat. Living Resour.
Volume 31, 2018
|
|
|---|---|---|
| Article Number | 21 | |
| Number of page(s) | 6 | |
| DOI | https://doi.org/10.1051/alr/2018010 | |
| Published online | 31 August 2018 | |
Research Article
Effect of increase in temperature on the survival and growth of Macrobrachium amazonicum (Palaemonidae) in the Amazon
1
Universidade Federal do Amapá (UNIFAP), Programa de Pós-Graduação em Biodiversidade e Biotecnologia ( PPG-BIONORTE),
Macapá,
AP, Brazil
2
Embrapa Amapá,
Macapá,
AP, Brazil
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Catharina Vinagre
Received:
8
March
2018
Accepted:
29
June
2018
Abstract
Macrobrachium amazonicum is a shrimp species distributed in freshwater habitats of Neotropical regions and is of great importance for the Amazonian economy. This study evaluated the effects of temperature increase on the survival and growth of M. amazonicum. For this, we distributed 360 M. amazonicum juveniles in 70 L tanks, and carried out a 90-day experiment with three treatments (T0: 28 ± 0.5 °C, or room temperature; T1: 30 ± 0.2 °C; T2: 32 ± 0.2 °C), using 4 replicate tanks each with 30 individual shrimp. Culture-tanks were connected to a recirculation system with biofiltration and constant aeration. Animals were fed twice a day using shrimp pelleted commercial food. After 90 days of trial, the total length and body mass gain of the animals cultured at room temperature was 78% and 433%, respectively. The specific growth rate, condition factor, weight gain, and length and survival of animals cultured at 30 and 32 °C were lower than those cultivated at 28 °C, and feed conversion was higher. Therefore, water temperature of 30 and 32 °C may compromise growth and survival of M. amazonicum during cultivation, none of the extreme temperatures may be recommended in practice.
Key words: Cultivation / growth / shrimp / survival / temperature
© EDP Sciences 2018
1 Introduction
According to current predictions, global temperature would increase 1.7–3.9 °C by 2100 in South America; however, the warming of the Amazon region will be greater (Junk, 2013). Climate change in the Amazon is expected to cause changes in rainfall patterns, which in turn causes long periods of drought and an overall reduction of water availability for the basin. This reduction is expected to increase water temperature in the Amazon River system and, consequently, cause drastic changes in the lifespan of organisms (Oberdorff et al., 2015). Climate change will affect aquatic organisms of the Amazon that must adapt to the new climatic regimes, migrate to thermally more suitable places or become extinct (Junk, 2013; Oberdorff et al., 2015). Therefore, climate change presents a great challenge to the sustainability of aquaculture (Ahmed et al., 2014). In addition, lowlands and floodplains, which serve as feeding grounds and nurseries for many species (Junk, 2013), will experience a drastic reduction due to climate change, affecting the biodiversity of Amazonian ecosystems.
In the Amazon, lowlands and floodplain areas present a variety of habitats that enable the existence of a diverse fauna (Isaac-Nahum, 2006; Junk, 2013). Aquatic fauna in this region is highly influenced by seasonal variations of precipitation and temperature levels. All these factors cause the region to concentrate a great fishing potential, highly influenced by seasonality (Isaac-Nahum, 2006; Maciel and Valenti, 2009; Junk, 2013; Freire et al., 2012; Nóbrega et al., 2014; Lima and Santos, 2014). Despite difficulties in quantifying the aquatic biodiversity of the region, many species are already threatened by over-exploitation of natural stocks.
Changes in environmental temperatures also greatly influence survival, growth, reproduction and disease susceptibility in freshwater shrimp species (Manush et al., 2004; Ahmed et al., 2014; Boock et al., 2016; Crisp et al., 2017), as this directly affects food intake and the metabolism of animals, causing stress. Survival and molting of Macrobrachium borellii and Palaemonetes argentinus were reduced at temperatures of 15 and 30 °C (Montagna, 2011). The growth of Macrobrachium rosenbergii increases in temperatures of 24–29 °C, and decreases in temperature of 34.0 °C (Habashy and Hassan, 2011), which lead to a higher food and oxygen consumption (Niu et al., 2003). Macrobrachium species have a wide of temperature tolerance that vary from 13 a 43 °C (Manush et al., 2004). In coastal regions of northeastern Brazil, adult and juvenile M. amazonicum populations occur in freshwater ecosystems with temperatures ranging from 26 to 31 °C (Rocha et al., 2015).
The decapod Macrobrachium amazonicum (shrimp-of-the-Amazon) has a wide distribution that includes several river basins, such as Orinoco, Amazonas, São Francisco, Araguaia-Tocantins, Paraná, Paraguay, and rivers of the northeastern, eastern, and northern coast of Brazil. In addition, this species has been recorded in Panamá, Costa Rica and Nicaragua, extending its distribution to Central America (Maciel and Valenti, 2009; Rocha et al., 2015). In the Amazonian river basin, M. amazonicum constitute a part of the diet of urban and riverine human populations, and play important ecological roles in aquatic ecosystems as a component of the trophic web (Maciel and Valenti, 2009; Lima and Santos, 2014). Therefore, this shrimp has great economic importance for artisanal fishing in the Amazon and in many water reservoirs from northeastern Brazil (Maciel and Valenti, 2009). This species also has a great potential for food production and important characteristics for animal husbandry, such as rapid growth and rusticity to handling (Maciel and Valenti, 2009; Lima and Santos, 2014). However, little is known about its culture, and the potential effects of increased temperature on its growth and survival have not been studied. Thus, the aim of this study was to investigate the effects of increases in water temperature on the survival and growth of M. amazonicum.
2 Materials and methods
2.1 Animals and experimental conditions
This 90-day experiment was conducted at the Larviculture Laboratory of Embrapa Amapá (0°0′50.07″S; 51°5′8.58″O) in Macapá, State of Amapá, Brazil. A total of 360 Macrobrachium amazonicum juveniles (length of 29.12 ± 4.13 mm and weight total of 0.22 ± 0.09 g) were used, which came from a laboratory larviculture, from females collected in the Igarapé da Fortaleza basin (0°1′35.75″S; 51°8′16.40″O), Macapá, State of Amapá (Brazil). Post-larvae and juveniles were kept in black tanks, following previous recommendations (Maciel & Valenti, 2014).
The M. amazonicum juveniles underwent three treatments with different temperatures: T0: room temperature, 28.0 ± 0.5 °C (control); T1: 30.0 ± 0.2 °C; T2: 32.0 ± 0.2 °C. We used 4 replicates per treatment, and included 30 animals in each replicate. These 360 shrimps were distributed in 12 tanks with a capacity of 70 liters of fresh water, coupled to a recirculation system with biological filtration and constant aeration. Temperature was maintained constant (± 0.2 °C) in T1 and T2 but was left to naturally vary in the control treatment, using thermostats (Roxin® model HT 1900 of 100 W, China). Temperature in the tanks with controlled values was increased in 0.5 °C every two days starting from room temperature, to enable thermal acclimation. The animals were fed twice daily (8 a.m. and 5 p.m.) with a shrimp pelleted commercial diet (35% crude protein), with a daily supply of 5% of the biomass, divided into two equal portions (i.e., 2.5% of biomass in each portion). During 90 days, every 15 days all individuals were removed from each tank for biometry, and were then returned to their respective experimental units. Corresponding biomass was adjusted, and 50% of the water from the culture tanks was changed.
Water temperature, pH, dissolved oxygen and conductivity were determined daily using a multiparameter analyzer (Horiba®, model U-52G, Tokyo, Japan). Tanks were then cleaned in order to remove food wastes and feces, recording observations concerning molting and dead animals. Total ammonia levels were measured every three days using a photometer ammonia gauge (Hanna® model HI 96715C, Romania).
2.2 Procedures to evaluate growth parameters
The total length (mm) and body weight (g) of all shrimps were measured after 90 days to evaluate husbandry parameters such as gross feed conversion rate, body weight gain (%), length gain (%), weight gain (g), daily growth (%/day), average final weight (g), average length (mm) and survival (%). These parameters are described as (Cavalli et al., 2004; Ning et al., 2007):
Gross feed conversion rate = amount of feed (g)/total weight gain (g);
Weight gain (g) = final average weight − initial average weight;
-
Specific growth rate = [(average final weight − average initial weight) * 100]/days of experiment;
-
Growth rate in total length (%) = (average of the final total length − average of the initial total length/average of the initial total length) * 100;
-
Survival rate (%) = (number of alive animals at the end of the experiment/initial number of animals) * 100.
The mass-to-length ratio was calculated using the equation W = aLb, where W is the weight (g), L is the total length (mm), and a and b are constants. These constants were estimated by means of a linear regression of the transformed equation: W = loga + b x logL. The length (mm) and body weight (g) data were also used to calculate the condition factor (Kn), following recommendations from Le-Cren (1951).
2.3 Statistical analysis
Normality and homoscedasticity of the data were previously tested using the Shapiro-Wilk and Bartlett tests, respectively. The data did not follow a normal distribution; thus the Kruskal-Wallis test, and Dunn test were used to assess differences among medians (Zar, 2010). All analyses were performed using R software v. 3.2.4 (R Core Team, 2015).
3 Results
Significant differences (P < 0.01) were detected in pH, dissolved oxygen and electrical conductivity between treatments (Tab. 1), caused by the increase of temperature.
Feed conversion rates of M. amazonicum individuals under temperatures of 30 °C and 32 °C were higher than the observed for individuals at room temperature (28 °C). Weight and length gain, specific growth rate, survival rate and the condition factor were lower in the treatments compared with the control group (Tab. 2).
The survival rate differed (P < 0.01) between the treatments across the 90-day experiment. Animals who were kept at 30 °C and those at 32 °C presented a survival rate 10% and 110% lower, respectively, compared with those who were kept at room temperature (28 °C) (Fig. 1).
The final total length and body weight of the animals at 30 and 32 °C was lower compared with control animals (Fig. 2).
Water quality parameters in tanks of Macrobrachium amazonicum juveniles during 90-day of cultivation at different temperature treatments.
Performance parameters of Macrobrachium amazonicum juveniles after 90 days of cultivation at different temperature treatments (T0: 28 ± 0.5 °C, T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C).
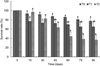 |
Fig. 1 Variation in the survival rate of Macrobrachium amazonicum juveniles across the 90-day culture at different temperatures (T0: 28 ± 0.5 °C, T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C). Mean values followed by the same letters between column indicate no difference according to Dunn test (P < 0.01). |
 |
Fig. 2 Initial and final length and body weight (mean values) of Macrobrachium amazonicum juveniles after 90 days of cultivation at different temperatures (T0: 28 ± 0.5 °C; T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C). Mean values followed by the same letters (lowercase) did indicate no difference for initial length and weight, and mean values followed by the same letters (uppercase) indicate no difference for final length and weight according to the Dunn test (P > 0.05). |
4 Discussion
Our results indicate that higher temperatures used for cultivating M. amazonicum, triggered a reduction in pH, dissolved oxygen and electric conductivity levels. Metabolic activities of Macrobrachium spp. are controlled by temperature and oxygen consumption increases with increasing temperature (Niu et al., 2003; Manush et al., 2004). However, in M. amazonicum, increase of temperature did not affect ammonia levels. Two factors contributed to this stability in ammonia levels: the efficiency of the biofilters used and the periodical exchange of 50% of the water from the tanks. The ammonia nitrogen is a variable that provides a rough estimate of the potential pollution in a culture tank. However, a large amount of oxygen can be used by nitrifying bacteria to oxidize ammonia into nitrate. Therefore, a fraction of dissolved oxygen will be consumed in the decomposition process of organic matter and nitrification (Keppeler et al., 2012). The values of these parameters were similar to values previously used in the cultivation of M. amazonicum (Aya-Baquero and Velasco-Santamaría, 2013; Kimpara and Santos, 2013; Maciel and Valenti, 2014), which is a species adapted to the high temperatures of the Amazon. Optimum temperature for shrimps is in the range of 28–32 °C and dissolved oxygen concentration of 5–8 mg/L. Therefore, considering that the values of water quality suited to grow M. amazonicum have not yet been established (Maciel and Valenti, 2014), values of temperature (28–29 °C), pH (7.6–7.8), dissolved oxygen (7.5–7.9 mg L−1), total ammonia (≤ 0.04 mg L−1) and electrical conductivity (150–155 μ Scm−1) found may be recommended as standard values for the intensive cultivation of this freshwater shrimp species.
Temperature is one of the most important factors that controls growth rate, food intake, feed conversion and survival rate of shrimp, and may affect disease resistance. This is one of the important factors that determine economic performance in shrimp farming (Moraes-Riodades et al., 2006; Ahmed et al., 2014; Boock et al., 2016), because the metabolic rate increases and survival with optimum temperature (García-Guerrero, 2010). Thus, for commercially cultured crustaceans, the effect of water temperatures on larval growth rates are often incorporated into planning tools, such as degree-hours or degree-days calculations (Crisp et al., 2017). Rising temperature up to certain limit favors aquaculture by reducing the time required to produce marketable sized animal and producing more generations per year. Nevertheless, high temperature may affect the health of aquatic animals by increasing metabolic rates and subsequent oxygen demand, as well as increasing proliferation, invasiveness and virulence of bacteria and other pathogens that causes pathophysiological disturbances in shrimps (Manush et al., 2004).
Increased production of food should not only consider the intensification of production practices, but should also ensure the sustainable use of natural resources. Thus, the focus should be the development of technologies and management procedures that increase the productivity of underperforming systems, reducing the negative impacts and enhancing positive impacts (Boock et al., 2016). In this study, the production performance of M. amazonicum was better when the animals were cultured at temperature of 28 °C; because temperatures above this threshold reduced body weight gain and growth, specific growth rate, body condition and survival rate, increasing feed conversion rates. Similarly, for Macrobrachium borellii and Palaemonetes argentinus, survival rate was reduced at temperatures of 15 and 30 °C (Montagna, 2011). The growth of Macrobrachium rosenbergii increased at 24–29 °C, and decreased at 34 °C (Habashy and Hassan, 2011). Therefore, these species are sensitive to small changes of environmental temperature. Negative impact on survival of Metapenaeus dalli at the highest temperature of 32.6 °C was reported (Crisp et al., 2017).
In aquaculture, growth is usually measured in body weight gain, and the cultivation with optimum economic performance is the one with lower feed conversion. In this study, feed conversion rates of juvenile shrimps for the three temperatures ranged from 2.7 to 2.9, being lower than those reported by Marques et al. (2010) for M. amazonicum (4.1–4.2) also grown in net cages. Metabolic activities of Macrobrachium species are controlled by temperature (Manush et al., 2004), which affects the food consumption (Niu et al., 2003). The condition factor, a quantitative indicator of body condition in response to food, reproductive and environmental conditions (Le-Cren, 1951; Deekae and Abowei, 2010; Rocha et al., 2015) was lower in M. amazonicum at temperatures 30 and 32, due to the reduction of the body weight. As the growth efficiency was affected by these water temperatures; probably, they contribute for a lower food consumption. However, Madlen (2010) showed that growth of M. rosenbergii declined in high temperature due to failure of enzymatic function. Therefore, as temperature is the most important limiting factor for metabolic functions in crustaceans, but particularly in shrimps directly determining metabolic rate and growth (Manush et al., 2004; Montagna, 2011; Crisp et al., 2017), it can change the energy flow (Montagna, 2011) and maybe food assimilation, affecting the weight and body condition.
Macrobrachium species have wide minimum and maximum temperature (Manush et al., 2004), in dependence of life stage. Survival of M. amazonicum larvae is better at 28 °C than at 30 °C (Maciel and Valenti, 2009). In this study, the highest survival rate of M. amazonicum juveniles was 78% at temperatures around 28 °C, while the survival rate was 65% at 30 °C and only 36% at 32 °C. The survival rates recorded at temperatures of 28 °C and 30 °C may be considered high for a water recirculation system, because the survival rate estimated for cultivation in nurseries of M. amazonicum is above 60% (Maciel and Valenti, 2009). Macrobrachium amazonicum is abundant in natural environments and may represent about 80% of the microcrustaceans' biomass in certain biotopes, such as the Amazonian lakes (Maciel and Valenti, 2009).
5 Conclusions
Temperature increase reduces the levels of dissolved oxygen, pH and conductivity, negatively affecting the survival, growth and fattening of M. amazonicum juveniles. As water temperature of 30 and 32 °C may compromise growth and survival of M amazonicum during cultivation, none of the extreme temperature is therefore recommended in practice.
Conflicts of interest
All authors declare that they have no competing interests.
Acknowledgments
Our acknowledgments to Elane Tavares Lobo, Evandro Freitas dos Santos, Osiel Amoras de Araújo junior, Sting Silva Duarte, and Taina Martins de Carvalho for their help in this study. This work was funded by the research foundation of the state of Amapá (Fundação de Amparo à Pesquisa do Estado do Amapá–FAPEAP) and a CNPq productivity research grant (#303013/2015-0) to Dr. M. Tavares-Dias.
References
- Ahmed N, Bunting SW, Rahman S, Garforth CJ. 2014. Community-based climate change adaptation strategies for integrated prawn-fish-rice farming in Bangladesh to promote social-ecological resilience. Rev Aquac 6: 20–35. DOI:10.1111/raq.12022 [CrossRef] [Google Scholar]
- Aya-Baquero E, Velasco-Santamaría Y. 2013. Fecundidad y fertilidad de Macrobrachium amazonicum (Héller, 1862) (Decapoda, Palaemonidae) del Piedemonte Llanero Colombiano. Rev MVZ Cordoba 18: 3773–3780. [CrossRef] [Google Scholar]
- Boock MV, Marques HL, Mallasen M, Barros HP, Moraes-Valenti P, Valenti WC. 2016. Effects of prawn stocking density and feeding management on rice–prawn culture. Aquaculture 451: 480–487. DOI:10.1016/j.aquaculture.2015.10.009 [CrossRef] [Google Scholar]
- Cavalli RO, Zimmermann S, Speck RC. 2004. Growth and feed utilization of the shrimp Farfantepenaeus paulensis fed diets containing different marine protein sources. Ciên Rural 34: 891–896. [CrossRef] [Google Scholar]
- Crisp JA, Partridge GJ, D'Souza FML, Tweedley JR, Moheimani NR. 2017. Effects of temperature and salinity on larval survival and development of the western school prawn Metapenaeus dalli. Int Aquat Res 9: 1–10. DOI:10.1007/s40071-016-0151-0 [CrossRef] [Google Scholar]
- Deekae SN, Abowei JSN. 2010. Macrobrachium macrobrachion (Herklots, 1851) length–weight relationship and Fulton's condition factor in Luubara creek, Ogoni Land, Niger Delta, Nigeria. Int J Anim Vet Adv 2: 155–162. [Google Scholar]
- Freire JL, Marques CB, Silva BB. 2012. Estrutura populacional e biologia reprodutiva do camarão-da-amazônia Macrobrachium amazonicum (Heller, 1862) (Decapoda: Palaemonidae) em um estuário da região nordeste do Pará, Brasil. Brazilian J Aquat Sci Techol 16: 65–76. [CrossRef] [Google Scholar]
- García-Guerrero MU. 2010. Effect of temperature on consumption rate of main yolk components during embryo development of the prawn Macrobrachium americanum (Crustacea: Decapoda: Palaemonidae). J World Aquac Soc 41: 84–92. DOI:10.1111/j.1749-7345.2009.00336.x [CrossRef] [Google Scholar]
- Habashy MM, Hassan MMS. 2011. Effects of temperature and salinity on growth and reproduction of the freshwater prawn, Macrobrachium rosenbergii (Crustacea- Decapoda) in Egypt. IJESE 1: 83–90. [Google Scholar]
- Isaac-Nahum VJ. 2006. Explotação e manejo dos recursos pesqueiros do litoral amazônico: um desafio para o futuro. Cienc Cult 58: 33–36. [Google Scholar]
- Junk WJ. 2013. Current state of knowledge regarding South America wetlands and their future under global climate change. Aquat Sci 75: 113–131. DOI:10.1007/s00027-012-0253-8 [CrossRef] [Google Scholar]
- Keppeler EC, Moraes-Valenti PMC, Pereira LV. 2012. Temporal variation in the water quality of ponds and effluent of grow-out ponds of Amazon River prawn Macrobrachium amazonicum. Rev Peru Biol 19: 299–306. [Google Scholar]
- Kimpara JM, Santos AO. 2013. Effect of water exchange and mechanical aeration on grow-out of the Amazon river prawn in ponds. J World Aquac Soc 44: 845–852. DOI:10.1111/jwas.12080 [CrossRef] [Google Scholar]
- Le-Cren ED. 1951. The length–weight relationship and seasonal cycle in gonad weight and conditions in the perch (Perca fluviatilis). J Anim Ecol 20: 201–219. [CrossRef] [Google Scholar]
- Lima JF, Santos TS. 2014. Aspectos econômicos e higiênico-sanitários da comercialização de camarões de água doce em feiras livres de Macapá e Santana, Estado do Amapá. Biota Amaz 4: 1–8. [CrossRef] [Google Scholar]
- Maciel CR, Valenti WC. 2014. Effect of tank colour on larval performance of the Amazon river prawn Macrobrachium amazonicum. Aquac Res 45: 1041–1050. DOI:10.1111/are.12048 [CrossRef] [Google Scholar]
- Maciel CR, Valenti WC. 2009. Biology, fisheries, and aquaculture of the Amazon river prawn Macrobrachium amazonicum: a review. Nauplius 17: 61–79. [Google Scholar]
- Marques HLA, Lombardi JV, Mallasen M, Barros HP, Boock MV. 2010. Stocking densities in cage rearing of Amazon river prawn (Macrobrachium amazonicum) during nursery phases. Aquaculture 307: 201–205. DOI:10.1016/j.aquaculture.2010.07.035 [CrossRef] [Google Scholar]
- Montagna MC. 2011. Effect of temperature on the survival and growth of freshwater prawns Macrobrachium borellii and Palaemonetes argentinus (Crustacea, Palaemonidae). Iheringia Série Zool 101: 233–238. DOI:10.1590/S0073-47212011000200011 [CrossRef] [Google Scholar]
- Moraes-Riodades, PMC, Kimpara JM, Valenti WC. 2006. Effect of the Amazon river prawn Macrobrachium amazonicum culture intensification on ponds hydrobiology. Acta Limnol 18, 311–319. [Google Scholar]
- Manush SM, Pal AK, Chatterjee N, Das T, Mukherjee SC. 2004. Thermal tolerance and oxygen consumption of Macrobrachium rosenbergii acclimated to three temperatures. J Thermal Biol 29: 15–19. [CrossRef] [Google Scholar]
- Ning QJ, Fu SG, Xu XJ, He JT. 2007. A new and practical application of JH antagonist KK-42 to promoting growth of shrimp Penaeus schmitti. Aquaculture 270: 422–426. DOI:10.1016/j.aquaculture.2007.04.078 [CrossRef] [Google Scholar]
- Niu C, Lee D, Goshima S, Nakao S. 2003. Effects od temperature on food consumptio, growth and oxigen consumption of freshwater prawn Macrobrachium rosenberguii (De Man, 1879) postlarvae. Aquat Res 34: 501–506. [CrossRef] [Google Scholar]
- Nóbrega PSV, Bentes B, Martinelli-lemos JM. 2014. Population structure and relative growth of the Amazon shrimp Macrobrachium amazonicum (Heller, 1862) (Decapoda: Palaemonidae) on two islands in the fluvial-estuarine plain of the Brazilian Amazon. Nauplius 22: 13–20. DOI:10.1590/S0104-64972014000100002 [CrossRef] [Google Scholar]
- Oberdorff T, Jézéquel C, Campero M, Carvajal-Vallejos F, Cornu JF, Dias MS, Duponchelle F, Maldonado-Ocampo JA, Ortega H, Renno JF, Tedesco PA. 2015. How vulnerable are Amazonian freshwater fishes to ongoing climate change? J Appl Ichthyol 31: 4–9. DOI:10.1111/jai.12971 [CrossRef] [Google Scholar]
- R Core Team. 2015. R Core Team. R A Lang. Environ. Stat. Comput. R Found. Stat. Comput. Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org/ [Google Scholar]
- Rocha SS, Silva RLS, Santos JL, Oliveira G. 2015. Length–weight relationship and condition factor of Macrobrachium amazonicum (Heller, 1862) (Decapoda, Palaemonidae) from a reservoir in Bahia, Brazil. Nauplius 23: 146–158. DOI:10.1590/S0104-64972015002308 [CrossRef] [Google Scholar]
- Zar JH. Biostatistical analysis, Prentice Hall, New Jersey USA, 2010 [Google Scholar]
Cite this article as: Bastos AM, Lima JF, Tavares-Dias M. 2018. Effect of increase in temperature on the survival and growth of Macrobrachium amazonicum (Palaemonidae) in the Amazon. Aquat. Living Resour. 31: 21
All Tables
Water quality parameters in tanks of Macrobrachium amazonicum juveniles during 90-day of cultivation at different temperature treatments.
Performance parameters of Macrobrachium amazonicum juveniles after 90 days of cultivation at different temperature treatments (T0: 28 ± 0.5 °C, T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C).
All Figures
 |
Fig. 1 Variation in the survival rate of Macrobrachium amazonicum juveniles across the 90-day culture at different temperatures (T0: 28 ± 0.5 °C, T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C). Mean values followed by the same letters between column indicate no difference according to Dunn test (P < 0.01). |
| In the text | |
 |
Fig. 2 Initial and final length and body weight (mean values) of Macrobrachium amazonicum juveniles after 90 days of cultivation at different temperatures (T0: 28 ± 0.5 °C; T1: 30 ± 0.2 °C, T2: 32 ± 0.5 °C). Mean values followed by the same letters (lowercase) did indicate no difference for initial length and weight, and mean values followed by the same letters (uppercase) indicate no difference for final length and weight according to the Dunn test (P > 0.05). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


