| Issue |
Aquat. Living Resour.
Volume 39, 2026
|
|
|---|---|---|
| Article Number | 10 | |
| Number of page(s) | 13 | |
| DOI | https://doi.org/10.1051/alr/2026003 | |
| Published online | 09 April 2026 | |
Research Article
Introduction pathways and ecological impacts of Nile tilapia in a large tropical reservoir of India: implications for management
1
ICAR- Central Inland Fisheries Research Institute, Barrackpore, Kolkata, India
2
Vadodara Research Centre of ICAR - Central Inland Fisheries Research Institute, Gujarat, India
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
16
September
2025
Accepted:
18
March
2026
Abstract
This study examines the early invasion of Nile tilapia (Oreochromis niloticus) in Panam Reservoir, Gujarat, marking the first record of an exotic fish species establishing itself in this large reservoir system. Using Rapid Rural Appraisal and participatory approaches, the introduction pathway was traced to an accidental escape from cage culture initiated in 2018, with a major loss event in 2019. Initially seen sporadically until 2022, tilapia quickly expanded its presence, accounting for over 20% of the total catch by 2023 and ranking third among landed species. Ecological evaluations showed significant dietary overlap with native fishes, an association with eutrophic conditions, and traits such as trophic plasticity and high reproductive rates, indicating strong invasive potential. Although tilapia has increased overall fish biomass and contributed to local protein supply through higher landings, it presents ecological and economic threats by displacing native species and reducing the value of indigenous fisheries. The study highlights the need for strict biosecurity measures, regulated enclosure culture, and integrated management strategies to balance productivity with biodiversity conservation.
Key words: Invasion / exotic species / large reservoir / tilapia / enclosure culture
Handling Editor: Victor Frossard
© A.M. Sajina et al., Published by EDP Sciences 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Biodiversity underpins the structure, functioning, and resilience of ecosystems, yet it is facing an unprecedented global decline (Hooper et al., 2005). Among the primary drivers of this decline, biological invasions by exotic species have emerged as a major threat to native biodiversity and ecosystem stability. These invasions can disrupt food webs, alter species interactions, and impair ecosystem services essential to human well-being (Leprieur et al., 2008; Douthwaite et al., 2018; Rice et al., 2021; Kang et al., 2023; Nambeye et al., 2025). However, the ecological mechanisms and consequences of invasions vary considerably across taxa, ecosystems, and spatial scales, making it crucial to understand context-specific invasion dynamics.
In freshwater systems, unauthorized introductions of nonnative fishes are a persistent challenge, particularly in developing countries where aquaculture and capture fisheries are closely intertwined. Understanding the pathways of such introductions is essential for formulating effective prevention and management strategies (Azzurro et al., 2014; Cucherousset and Olden, 2011, 2020). Nonnative fishes can alter the trophic structure of aquatic communities, outcompete native species, and introduce cascading ecological effects with far-reaching consequences (Britton et al., 2010; Eloranta et al., 2014; Matsuzaki et al., 2013).
The term “tilapia” refers to a group of tropical freshwater fish within the Cichlidae family, comprising species from the genera Oreochromis, Tilapia, and Sarotherodon, which are native to regions of Africa and the southwestern Middle East. Since the 1930s, these fish have been deliberately introduced to various parts of the world for multiple purposes, including controlling aquatic vegetation and insect populations, serving as bait in capture fisheries, being kept in aquariums, and more prominently, for their value as a food source through aquaculture (Canonico et al., 2005). Tilapias, particularly Oreochromis niloticus, the Nile tilapia, are among the most widely introduced freshwater fishes due to their high reproductive rate, broad environmental tolerance, and economic value. Often dubbed the “aquatic chicken” (Coward and Little, 2001), tilapias have become a cornerstone of aquaculture in many countries, supporting both food security and rural livelihoods. However, their unregulated introduction into open waters has sparked ecological concerns. Tilapia invasions have been linked to biodiversity loss, altered ecosystem functioning, and degradation of native fish communities, especially in tropical and subtropical freshwater systems (Canonico et al., 2005; Shuai et al., 2019; Shuai and Li, 2022; Yongo et al., 2023; Xiong et al., 2023).
Reservoirs, being man-made and inherently dynamic ecosystems, are particularly susceptible to biological invasions (Havel et al., 2005; Moss, 2008). Shaped by both natural processes and human interventions, including species introductions, they serve as important interfaces between aquaculture and inland capture fisheries (Panikkar and Khan, 2008). In India, reservoirs hold considerable potential for enhancing fish production (van Zwieten et al., 2011), but this potential is increasingly threatened by the spread of exotic species that disrupt ecological balance (Panikkar et al., 2021). The ecological complexity of reservoirs, with their variable hydrology, diverse fish communities, and differing management practices, calls for a better understanding of invasion dynamics to support adaptive and sustainable fisheries management. Case studies from tropical regions highlight these concerns. For instance, Hamid et al. (2023) reported the escape of O. niloticus from aquaculture facilities into the natural waters of the Temengor Reservoir in Malaysia, raising concerns about ecological competition with native species. Similarly, a study by Johnson et al. (2022) documented a sharp rise in the catch share of O. niloticus in the Halali Reservoir, Madhya Pradesh, during the decade from 2008 to 2018, underscoring its rapid proliferation and the associated decline of commercially valuable native fish species.
This growing concern underscores the urgent need to evaluate the ecological outcomes of fish invasions and to predict the conditions under which exotic species, such as tilapia, establish themselves, proliferate, and impact native biodiversity. The present study addresses this knowledge gap by investigating the early-stage invasion of Nile tilapia in Panam Reservoir, Gujarat, examining its introduction pathway, ecological interactions, and implications for sustainable reservoir fisheries.
2 Material and methods
2.1 Study site and sampling
The Panam Reservoir, located in Gujarat, India (latitude 23.000°N; longitude 73.700°E to 73.800°E), was selected as the study site, as it is a relatively under-studied large freshwater reservoir of regional importance. Constructed on the River Panam, a tributary of the River Mahi, the reservoir's development began in 1971, with gradual filling during the late 1970s and early 1980s and full completion in 1999. The reservoir has a catchment area of 2312 km2 and a surface area of 89.80 km2 at full reservoir level and serves as an important source of irrigation water, drinking water, and inland fisheries while supporting diverse aquatic biota. The surrounding landscape is predominantly rural, comprising dispersed villages, agricultural fields, and patches of dry deciduous forest, with no major urban centers or industrial activities in the immediate vicinity.
Three sampling stations (S1–S3) were selected to represent the longitudinal gradient of the reservoir and associated environmental heterogeneity (Fig. 1). Site selection was based on hydrological regime, position along the reservoir continuum, and representativeness of dominant habitat conditions. Station S1 was located in the dam-side lentic zone, characterized by relatively deeper and hydrologically stable conditions, with observed depths ranging from approximately 13 to 14 m during sampling. Station S2 was established in the transitional mid-reservoir zone, where lacustrine and riverine influences overlap and water depths ranged from about 6 to 12 m. Station S3 was positioned in the upper lotic, riverine zone, characterized by shallow depths (∼3.5–5 m) and stronger fluvial influence. All stations were located away from the immediate shoreline to ensure comparable open-water sampling conditions.
Seasonal sampling was carried out over a two-year period (2022–2023) to capture intraannual and interannual variability in environmental conditions and aquatic biota. Sampling was conducted during four distinct seasonal phases typical of the region: pre-monsoon (March–May), southwest monsoon (June–September), post-monsoon (October–November), and winter (December–February). Each station was sampled once during each season in both years. Water quality parameters including water temperature, dissolved oxygen, pH, specific conductivity, total dissolved solids, total alkalinity, total hardness, phosphate-P, nitrate-N, and silicate-Si were measured seasonally across zones following standard methods prescribed by APHA (2017).
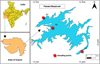 |
Fig. 1 Map showing the location of Panam Reservoir in Gujarat, India, with designated fish sampling points. |
2.2 Fish diversity and catch structure analysis
Fish diversity and catch structure of Panam Reservoir were assessed through seasonal fish landing surveys conducted during 2022–2023 at established landing centers representing the lentic, transitional, and lotic zones of the reservoir. Due to the limited number of active landing sites, these centers were considered representative of overall fishing activity. Surveys were conducted for one full day per season, during which daily catches of operating fishers were examined, and all landed fish were identified to species level using standard taxonomic keys. Observed total length (TL) of representative individuals in the landings was noted during surveys to provide indicative size information, although systematic length–frequency sampling was not undertaken.
Species richness was compiled across seasons to document fish diversity, with particular attention to small indigenous fish species. Information on self-recruitment of commercially important native species and the use of riverine (lotic) sections as breeding areas was derived from Rapid Rural Appraisal, key informant interactions, and fishers' observations, supported by repeated seasonal occurrence of juveniles in landings from the upper reaches. These observations are qualitative and are presented to provide ecological context rather than quantitative estimates of recruitment. Catch structure was analyzed based on species-wise relative contribution to total landings, and seasonal and interannual changes in catch composition were evaluated to identify shifts in dominance patterns, with emphasis on the emergence and increasing contribution of O. niloticus between 2022 and 2023.
2.3 Dietary overlap studies
Dietary overlap between O. niloticus and native fishes was examined using specimens obtained from the seasonal landing surveys described in Section 2.2, thereby directly linking trophic analysis to observed catch composition. Based on dominance in landings and reported feeding ecology, four native species, viz., Labeo rohita, Labeo calbasu, Systomus sarana, and Cirrhinus reba, were selected for comparison.
Thirty individuals of O. niloticus and each selected native species were collected from fishers' landings during 2022–2023, pooled across seasons, and transported on ice to the laboratory. Gut contents were analyzed using the visual estimation method (Hyslop, 1980; Manko, 2016), wherein the relative volume of each dietary category was visually estimated under a low-power microscope and expressed as a percentage of total gut content. Dietary overlap was quantified using Schoener's index (Schoener, 1970): C = 1–0.5 Σ |pᵢ − qᵢ|, where pᵢ and qᵢ represent the proportional contribution of the ith food item in the diets of the two species being compared. Values greater than 0.6 indicate substantial dietary overlap.
2.4 Phytoplankton analysis
Phytoplankton sampling was carried out at the three sampling stations following the seasonal framework described in Section 2.1. Each station was sampled once per season in each year (2022–2023). At each station, phytoplankton samples were collected using two complementary approaches. Integrated water samples were obtained from the upper water column (0–50 cm) using a Van Dorn sampler, from which a 1-L subsample was used for quantitative analysis. In addition, net samples were collected by filtering surface water through a 20 µm mesh plankton net to enhance representation of larger and colonial taxa. All samples were immediately preserved with Lugol's iodine solution (1% final concentration) for microscopic examination (APHA, 2017). Qualitative identification was performed under a compound microscope (40 × –100 × ) using standard taxonomic keys and monographs (Prescott, 1978; Bellinger & Sigee, 2010). Quantitative enumeration was conducted using a Sedgwick–Rafter counting chamber, and phytoplankton abundance was expressed as individuals per liter. Taxonomic identification was based on diagnostic morphological characteristics, including cell size and shape, colony formation, and surface ornamentation. For a descriptive assessment of community composition, phytoplankton data from both sampling years were pooled, as the analysis aimed to characterize overall community structure and dominance patterns rather than interannual variability.
2.5 Pathway analysis and ecosystem impact assessment
The invasion pathways of Oreochromis niloticus and its associated ecosystem impacts were investigated using an integrated approach combining Rapid Rural Appraisal (RRA), fishery information, and interpretation of field-based ecological data generated under the sampling framework described above.
Rapid Rural Appraisal (Dunn, 1994; Pido et al., 1997) was undertaken to reconstruct the introduction history and subsequent spread of O. niloticus in Panam Reservoir. The RRA comprised semi-structured interviews, key informant interactions, and group discussions with stakeholders directly associated with reservoir fisheries. Respondents included approximately 25 active fishers, 6 local fish traders, and 4 fisheries managers or cooperative representatives drawn from major fishing villages and landing centers around the reservoir, namely Panam, Hareda, Bhunidra, Nasirpur, Mahelan, Chhogada, Boriavi, Gajipur, and Valaiya, ensuring spatial coverage across dam-side, transitional, and riverine sections. Participants were selected using purposive sampling to capture both spatial and experiential diversity.
Key informant interactions were conducted with experienced senior fishers, cooperative office bearers, and officials possessing long-term knowledge of fisheries management practices and cage culture operations in the reservoir. In addition, two group discussions were held at major landing sites, with group sizes ranging from more than 20 to over 30 participants, to validate individual responses and triangulate information on potential introduction pathways, including escape from cage aquaculture, accidental release, or intentional stocking. RRA activities were carried out continuously between May 2022 and December 2023, with one day of each seasonal field visit dedicated to stakeholder interactions.
Ecological implications of tilapia establishment were interpreted by integrating stakeholder-derived information with patterns observed in water quality, phytoplankton composition, and fish catch structure, as described in subsequent sections. Fish assemblage-environment relationships were analyzed using Canonical Correspondence Analysis (CCA) in PAST. The CCA included the 10 most abundant species from different ecological groups, including O. niloticus, the focal species of this study. Abundance data from three seasons (pre-monsoon, monsoon, and post-monsoon) and three habitat zones (lotic, transitional, and dam-side) were considered. Water quality variables were log(x + 1) transformed, and fish abundance data were square-root transformed prior to CCA.
3 Results
3.1 Fish diversity and fisheries
A total of 41 fish species were documented from the Panam Reservoir during the study period, reflecting moderate biodiversity in the aquatic ecosystem (Tab. 1). The fishery is co-dominated by carps and catfishes, which together constitute the bulk of commercial landings (Fig. 2). A substantial number of small indigenous fish species, such as Puntius spp., Pethia spp., Rasbora spp., Chanda nama, Parambassis spp., Macrognathus pancalus, Glossogobius giuris, and Channa punctata, were consistently recorded in landings but remain underutilized and are not actively targeted by prevailing fishing practices. The capture of large-sized individuals across several species is reflected in the observed mean total length values recorded during landing surveys (Tab. 1). Besides, many commercially valuable native species exhibit self-recruiting populations, particularly utilizing the lotic zones of the reservoir as breeding grounds. Information on self-recruitment and use of riverine (lotic) sections as breeding areas is derived from Rapid Rural Appraisal, key informant interactions, and repeated seasonal observations of juveniles in landings from the upper reaches of the reservoir. These observations are qualitative in nature and are presented to provide ecological context rather than quantitative estimates of recruitment or productivity.
 |
Fig. 2 Comparative percentage composition of major fish groups in the catch from Panam Reservoir during 2022 and 2023. |
List of fish species recorded from Panam Reservoir, their family, fishery group/category, and observed mean total length (TL ± SD) in landings (2022–2023).
3.2 Shifting catch structure
In the Panam Reservoir, recent fish catch records indicate a significant shift in species composition, notably marked by the establishment and rapid proliferation of O. niloticus. Prior to 2022, this species was either unrecorded or appeared negligible in fish landings, indicating its minimal presence within the reservoir's ichthyofaunal community and its insignificant contribution to the fishery. However, in 2022, its presence became more noticeable, contributing approximately 5% to the total fish catch. This trend continued in 2023, with O. niloticus rapidly rising to become the third most abundant species in the harvest, following Labeo rohita and Sperata seenghala. Figure 2 presents a comparative visual representation of species composition in 2022 and 2023, illustrating the rapid expansion of O. niloticus in the reservoir. Concurrently, a decline is observed in the shares of major and medium carps, minor cyprinids, and other native species, while indigenous catfishes show only marginal fluctuation.
3.3 Phytoplankton composition
The phytoplankton composition of Panam Reservoir based on pooled seasonal data from 2022 to 2023 indicated a highly eutrophic condition, with Cyanophyceae overwhelmingly dominant, contributing nearly 83% of the total assemblage. Within this group, Oscillatoria (11,700–17,100 cells L−1) and Microcystis (6800–21,000 cells L−1 across two species) were most abundant, followed by Anabaena (up to 1300 cells L−1), Pseudoanabaena (510–1470 cells L−1), Merismopedia (1600 cells L−1), Calothrix (1500 cells L−1), Aphanocapsa (300 cells L−1), and Gomphosphaeria (420 cells L−1). Bacillariophyceae contributed only a minor share, represented chiefly by Aulacoseira (590–1710 cells L−1) along with Cymbella, Navicula, Cyclotella, Synedra, Fragillaria, Gomphonema, and others in low densities (≤240 cells L−1). Chlorophyceae exhibited moderate diversity, with Spirogyra (410–1690 cells L−1), Scenedesmus (650 cells L−1), Chlorella (560 cells L−1), Pediastrum (130–260 cells L−1 across three species), Kirchneriella (220 cells L−1), Tetraedron (160 cells L−1), and Pandorina (120–160 cells L−1) as the principal representatives. Euglenophyceae occurred sparsely, with Trachelomonas (450 cells L−1) and Euglena (30 cells L−1), while Dinophyceae was represented by Ceratium (20–210 cells L−1) and Peridinium (10–30 cells L−1) (Fig. 3).
 |
Fig. 3 Phytoplankton composition in Panam Reservoir. Left: Major classes showing Cyanophyceae dominance. Right: Genera composing the Cyanophyceae community are dominated by Anabaena and Microcystis. |
3.4 Pathways of tilapia invasion
This represents the first recorded case of exotic fish invasion in Panam Reservoir, marked by the establishment of Nile tilapia. The inferred introduction pathway is based on concordant information obtained through RRA (summarized semi-quantitatively in Tab. 2), key informant interactions, and group discussions conducted across multiple fishing villages and landing centers around the reservoir.
The pathway analysis indicates that the introduction occurred through a combination of intentional and unintentional routes. O. niloticus was deliberately introduced in 2018 for cage culture aimed at food fish production. However, in 2019, a major accidental loss reportedly occurred when the entire stock escaped into the reservoir due to structural failure and/or extreme environmental conditions affecting the cage systems. This escape event was independently recalled by multiple stakeholder groups, including fishers from dam-side and mid-reservoir zones, suggesting an unintentional release into open waters.
Accordingly, the introduction pathway is best classified as mixed: intentional within cage-based aquaculture but unintentional with respect to release into the reservoir ecosystem. The high level of agreement among respondents and interview modes lends confidence to this reconstructed pathway despite the qualitative nature of the evidence. The subsequent establishment and proliferation of tilapia highlight the ecological risks associated with aquaculture-related species introductions, particularly in open-water systems.
Summary of RRA responses on invasion pathway and impacts of O. niloticus in Panam Reservoir.
3.5 Dietary overlap analysis
The results of dietary overlap analysis revealed significant potential for resource competition between Nile tilapia and native fishes in Panam Reservoir. Table 3 shows that tilapia shared major dietary components, namely phytoplankton, zooplankton, macrophytes, detritus, zoobenthos, and insects, with native species such as L. rohita, L. calbasu, S. sarana, and C. reba. Gut content analysis indicated that tilapia exhibited an omnivorous diet, dominated by phytoplankton (31%), detritus (19%), macrophytes (15%), zoobenthos (15%), zooplankton (10%), and insects (10%). Schoener's index values (Fig. 4) highlighted the highest dietary similarity with L. calbasu (0.74) and C. reba (0.70), suggesting considerable competition for shared resources, particularly detritus, zoobenthos, and insects.
 |
Fig. 4 Schoener's index values showing dietary overlap between Nile tilapia and native fishes in Panam Reservoir. |
 |
Fig. 5 CCA biplot depicting the association of environmental variables with the distribution of the ten most abundant fish species in Panam Reservoir. |
Gut content composition (%) of Nile tilapia and selected native fishes from the reservoir.
3.6 Ordination of fish abundance and reservoir water quality
The ordination of dominant fish species and environmental variables revealed clear gradients structuring the assemblage in Panam Reservoir. The first two canonical axes together explained 63.39% of the total inertia, with Axis 1 accounting for 35.35% and Axis 2 28.04% of the variation (Fig. 5). Axis 1 represented a gradient from relatively mineralized waters characterized by higher TDS, conductivity, and total hardness to nutrient-rich and oxygenated conditions associated with nitrate, phosphate, and dissolved oxygen. Within this ordination space, the invasive O. niloticus was positioned closer to vectors representing hardness, TDS, and conductivity, suggesting a tendency to occur in waters with relatively higher ionic content. In contrast, the species is located farther from dissolved oxygen, nitrate-N, and phosphate-P, indicating a comparatively weaker association with nutrient-related gradients (Fig. 5). However, the permutation test showed that the overall relationship was not statistically significant (Trace p = 0.18), suggesting only a weak association between environmental gradients and fish distribution patterns and that the distribution of O. niloticus may also be shaped by broader ecological factors such as habitat adaptability and trophic plasticity.
4 Discussion
Despite being impounded in the late 1970s to early 1980s, the Panam Reservoir has remained largely unstudied since its formation. Limited historical data exists, with Srivastava (1985) being one of the few to report on its fish productivity, estimated at a modest 6 kg/ha/year. This lack of historic scientific attention highlights a significant scope for comprehensive ecological and fisheries studies in this important reservoir. The relatively large mean total length observed across several fish species suggests favorable growth conditions in the reservoir, though these observations are indicative and not based on structured length–frequency analysis.
The increasing dominance of O. niloticus in the catch structure of Panam Reservoir, accompanied by a concurrent decline in major and medium carps, minor cyprinids, and other native species, with only marginal fluctuation in indigenous catfishes, indicates an emerging community shift. This pattern is supported by multiple lines of evidence in the present study: (i) RRA indicating cage culture escape as the likely introduction pathway; (ii) CCA showing its association with low dissolved oxygen and nutrient-rich zones; (iii) gut content analysis revealing broad trophic overlap with native species; and (iv) field observations suggesting that macrophyte-rich habitats facilitate its spread, collectively highlighting important ecological and fisheries management implications.
This pattern aligns with observations from other reservoirs where tilapia invasions have resulted in restructuring of native fish assemblages and fisheries yield. For example, in Tangxi Reservoir, the proliferation of non-native tilapia coincided with a marked decline in bighead carp and alteration of community structure (Peng et al., 2021). Aquaculture escape has repeatedly been identified as a primary introduction pathway. Similar establishment following cage culture escape has been documented in Temengor Reservoir, Malaysia (Hamid et al., 2023), and dispersal into river networks from aquaculture facilities has been reported in Mississippi (Lowe et al., 2012).
Recent evidence further indicates that post-escape spread is not solely passive. O'Mara et al. (2024) demonstrated, using otolith strontium isotope analysis, that tilapia actively move across habitats soon after introduction, facilitating rapid establishment within catchments. Together, these findings support two key dimensions of tilapia invasion reflected in Panam Reservoir: aquaculture-driven escape as the entry pathway and rapid dispersal as the mechanism of establishment. These dynamics emphasize the need for pathway-focused surveillance and early detection in reservoir ecosystems.
4.1 Driving factors for successful tilapia invasion in indian reservoirs
Tilapia exhibit several biological traits that contribute to their success as invaders in aquatic systems beyond their natural range. These traits include reproductive strategies, tolerance to diverse environmental conditions, and a low trophic level feeding habit (Canonico et al., 2005; Deines et al., 2016). Their breeding success is further supported by behaviors such as nest building, mouthbrooding, and brood guarding (Russell et al., 2012). Additionally, tilapia can modify their reproductive strategies to match environmental conditions, for example, by producing more broods with reduced brood efficiency during unfavorable seasons (Reardon and Chapman, 2008). During nesting, they may display aggressive behavior toward native fish species (Bradford et al., 2011).
Based on the results of the present study and supported by evidence from other documented tilapia invasions, several ecological and biological factors appear to have driven the successful establishment of O. niloticus in Panam Reservoir. These include tolerance to suboptimal environmental conditions revealed by CCA, the availability of macrophyte-dominated habitats, pronounced trophic plasticity and dietary overlap with native fishes, reduced ecological resistance reflected in the altered fish assemblage, exploitation of reservoir-created niches, and flexible reproductive strategies. Together, these factors explain the rapid establishment and expansion of this invasive species in the reservoir, as discussed below.
(i) High tolerance to a broad range of environmental conditions: The CCA outputs in the present study highlighted a clear association of O. niloticus with zones characterized by lower dissolved oxygen and elevated nutrient levels. This ordination pattern reflects tilapia's well-known tolerance to diverse environmental conditions (Philippart and Ruwet, 1982; Zambrano et al., 2006). Many Indian reservoirs undergo seasonal fluctuations and suboptimal conditions, such as reduced oxygen and high turbidity, which can limit native species (Sugunan, 1995; Sarkar et al., 2018). Tilapia's physiological adaptations allow it not only to survive but also to proliferate under such conditions, providing a distinct competitive advantage and enabling colonization of disturbed or marginal habitats within the reservoir
(ii) Extensive macrophyte-dominated habitats: Field observations in Panam Reservoir showed dense submerged macrophyte beds, particularly in shallow transitional zones where tilapia abundance was highest. These vegetated areas provide favorable microhabitats for nesting, shelter, and foraging, enhancing reproductive success and juvenile survival of O. niloticus. Such habitat complexity likely facilitated the localized establishment and spread of the species. Similar habitat preferences for tilapia have been reported elsewhere (O'Mara et al., 2024), supporting the pattern observed in Panama. The dominance of submerged vegetation in these zones thus appears to have played an important role in creating conditions conducive to tilapia invasion.
-
(iii) Trophic plasticity and dietary overlap with native fishes: Gut content analysis in the present study demonstrated that O. niloticus in Panam Reservoir consumed phytoplankton, zooplankton, macrophytes, detritus, zoobenthos, and insects, resulting in substantial dietary overlap with native fishes (Schoener's index 0.62–0.74). This wide dietary spectrum reflects the pronounced trophic plasticity of tilapia, a trait widely recognized as a key driver of its invasive success (Bowen, 1982; Canonico et al., 2005). Such generalist feeding behavior allows the species to persist and proliferate even under conditions where native, more specialized fishes may struggle to compete for resources.
The phytoplankton composition of Panam Reservoir indicated dominance of cyanobacteria, a condition under which tilapia are known to thrive (Kamujunke et al., 2002; Chen et al., 2006; Bwanika et al., 2006). Although cyanobacteria are often problematic due to their mechanical and toxic effects on grazers and fish (Landsberg, 2002; Bednarska, 2006), adult O. niloticus are capable of ingesting large colonial forms such as Microcystis aeruginosa and tolerating associated cyanotoxins, a capacity not commonly observed in juveniles or zooplankton (Bury et al., 1995; Wilson et al., 2006; Tillmanns et al., 2008). Under eutrophic conditions, cyanobacteria may constitute a major portion of the diet of adult tilapia (Getachew, 1987; Bwanika et al., 2006), with similar feeding patterns reported from other invaded systems.
Morphological adaptations, including an elongated gut and an efficient filter-feeding apparatus, further enhance tilapia's ability to utilize cyanobacterial blooms (Moriarty, 1973; Moriarty and Moriarty, 1973a,b; Sanderson et al., 2001). This feeding mechanism allows ingestion of filamentous and colonial cyanobacteria along with smaller phytoplankton and detrital particles (Temesgen et al., 2022). Tilapia are also known to switch to animal-based food sources such as zooplankton, insect larvae, and fish eggs under low-visibility conditions (Bwanika et al., 2006), highlighting their omnivorous tendencies.
Given these traits, the high abundance of Cyanophyceae observed in Panam Reservoir likely provided a favorable trophic niche for O. niloticus. The ability to assimilate dominant genera such as Microcystis and Anabaena (Getachew, 1987; Bwanika et al., 2006; Semyalo, 2009), combined with flexible utilization of plant, detrital, and animal food sources, appears to have facilitated the successful establishment and rapid proliferation of tilapia in the reservoir.
(iv) Reduced ecological resistance reflected in the altered fish assemblage structure: The altered fish assemblage structure observed in Panam Reservoir suggests reduced ecological resistance, a condition that favors the establishment of invasive species. The concept of biotic resistance proposes that ecosystems with high native biodiversity are less susceptible to invasions (Elton, 1958; Levine et al., 2004). However, in many Indian reservoirs, including Panam, anthropogenic pressures such as habitat modification, overfishing, pollution, and hydrological alterations associated with damming have contributed to a decline in native fish diversity (Sarkar et al., 2008). This reduction in native species diversity likely created ecological vacancies and weakened competitive and predatory controls, enabling opportunistic invaders like O. niloticus to establish and expand more effectively (Canonico et al., 2005; Kolar and Lodge, 2001). Similar patterns of invasion under reduced biotic resistance have been reported from other modified freshwater systems (Zambrano et al., 2006), supporting the observations from Panam Reservoir.
(v) Exploitation of vacant ecological niches created by reservoir transformation: The lentic and transitional character of Panam Reservoir, created by damming of the river system, appears to have provided ecological niches favorable for tilapia establishment. Reservoir formation alters flow regimes, habitat structure, and nutrient dynamics, often leading to the decline of river-adapted native species and colonization by lentic-tolerant taxa (Agostinho et al., 2008; Gomes and Miranda, 2001). In Panama, the dominance of O. niloticus in lentic and vegetated transitional zones, along with the observed reduction in native assemblage structure, suggests exploitation of such altered ecological space. Nutrient accumulation, macrophyte colonization, and habitat heterogeneity typical of reservoirs (Baran et al., 2007; Zhang et al., 2017a) likely created favorable conditions that were readily utilized by this invasive species. Similar exploitation of reservoir-created niches by non-native fishes has been documented in other impounded systems.
(vi) Flexible reproductive strategies and life history trait plasticity: The rapid increase in tilapia dominance observed in Panam Reservoir is also consistent with the species' well-known life history plasticity. Tilapia exhibits prolific breeding, early maturity, parental care, and the ability to reproduce multiple times within a year (Trewavas, 1983; Philippart and Ruwet, 1982). These traits enable populations to establish quickly and persist under fluctuating environmental conditions. In altered ecosystems such as reservoirs, where habitat disturbance and reduced competition occur, such reproductive flexibility provides a strong advantage (Canonico et al., 2005). These characteristics likely contributed to the swift establishment and expansion of O. niloticus in Panama following its escape from cage culture.
4.2 Potential impacts of tilapia invasion
The substantial dietary overlap observed in Panam Reservoir (Schoener's index 0.62–0.74) indicates that the broad trophic adaptability of O. niloticus provides a competitive advantage with the potential to destabilize native food web structures. Our findings are consistent with reports from the Pearl River, China, where tilapia invasion compressed the trophic niches of native species and reduced their isotopic diversity (Shuai and Li, 2022). In Panama, this overlap suggests a risk of displacement for native fishes, particularly those with specialized feeding habits. Similar ecological consequences have been observed in Halali Reservoir, Madhya Pradesh, where a significant reduction in the catch of Labeo catla, L. rohita, and C. mrigala followed the introduction of tilapia (Johnson et al., 2022).
Beyond resource competition, tilapia's benthic foraging behavior enhances nutrient release and sediment disturbance, which reduces water clarity, suppresses benthic algal growth, and promotes eutrophication (Zhang et al., 2017b; Yongo et al., 2023). Field observations in Panama, including increased turbidity reported through RRA and dominance in macrophyte-rich transitional zones, support the likelihood of such habitat alterations. These changes can degrade habitat structure and impair ecological functions. The reported decline in native carps in Panama, reflected in catch structure and RRA responses, may represent cascading effects of these ecological disturbances. Similar patterns were reported from Temengor Reservoir, where Nile tilapia escapees from aquaculture were associated with reduced abundance and niche overlap with native fishes (Hamid et al., 2023).
Despite these ecological concerns, RRA responses and landing observations indicated that tilapia has contributed to increased overall fish biomass and improved year-round protein availability to local communities. However, the dominance of a low-value species has reduced the economic return from fisheries due to the decline of high-value native fishes. While consistent catches support food security for small-scale fishers, economic returns remain limited due to poor market value.
There remains potential to improve tilapia profitability through value chain development, including processing, branding, and diversification. Its hardiness and adaptability also make it suitable for integrated fish farming in rural areas. As emphasized by Costa-Pierce (2003), aquaculture escape is a major pathway for tilapia invasions, with many feral populations traceable to culture facilities. The pathway reconstructed through RRA in Panama strongly reflects this pattern. The presence of hybrid stocks further complicates identification and regulation, underlining the need for molecular tools and stricter containment measures to prevent uncontrolled establishment in natural waters.
4.3 Management of tilapia invasions in indian reservoirs
Managing O. niloticus invasions in Indian reservoirs, such as Panam, is challenging due to the species' high adaptability, prolific breeding, and ecological resilience. While eradication is nearly impossible once established, population suppression can be achieved through targeted netting, trapping, and community-based fishing, especially in localized zones (Costa-Pierce, 2003). Installing barriers to prevent spread into sensitive habitats can strengthen these efforts, though they demand sustained labor and monitoring. Biological control using native predators like catfish may offer additional regulation but requires caution to avoid unintended impacts on native species (Gozlan et al., 2010).
Long-term strategies must focus on ecological restoration and strict policy enforcement. Enhancing water quality, reducing nutrient loads, reintroducing native species, and restoring vegetation can reduce tilapia's dominance and support native biodiversity (Kolding et al., 2008; Padilla and Williams, 2004). Regulatory measures, such as licensing, escape-proof aquaculture systems, and restrictions on live fish sales, are critical to prevent further spread (De Silva et al., 2009). Promoting confined tilapia culture in biosecure systems can balance economic benefits with ecological safety (FAO, 2022). Ultimately, an integrated approach involving habitat restoration, controlled harvest, policy support, and community awareness is essential to mitigate tilapia's impact and ensure the sustainability of India's reservoir ecosystems and dependent livelihoods.
Given the ecological conditions observed in Panam Reservoir, targeted and site-appropriate management interventions are essential. Drawing on recommendations by Johnson et al. (2022) from their work in Halali Reservoir and aligning with the ecological potential of Panam Reservoir, a combined strategy of direct removal, habitat protection, stocking, and biosecurity is proposed. These measures aim not only to limit tilapia populations but also to strengthen native fish communities and promote sustainable fisheries development and can be listed as follows:
Increase targeted fishing pressure on tilapia using gill nets with mesh sizes designed to capture adults (150–250 mm total length) to reduce the breeding population.
Implement focused removal of juveniles from shallow and marginal areas using small-mesh nets to limit recruitment and future population growth.
Enhance annual stocking of Indian major carps and indigenous minor carps to replenish native biodiversity and increase biotic resistance to tilapia dominance.
Reduce nutrient loading and manage water quality to prevent eutrophic conditions that support tilapia proliferation.
Protect and restore lotic habitats (riverine sections) critical for the breeding of native self-recruiting species.
Strengthen biosecurity measures for aquaculture operations, ensuring escape-proof cage systems and enforcing restrictions on live tilapia sales and transport.
Support confined, biosecure tilapia culture where economically viable, balancing production with ecological safeguards.
5 Conclusion
This study was conducted during the early stages of a Nile tilapia invasion in Panam Reservoir, Gujarat, providing timely insights into its ecological and socio-economic impacts. The findings emphasise tilapia's strong reproductive ability, trophic flexibility, and habitat adaptability, which have helped it to establish and spread quickly. Significant dietary overlap with native species and its association with eutrophic conditions indicate a growing threat to native biodiversity and ecosystem stability.
While the species boosts fish biomass and provides more protein for local communities, its spread has led to a decline in high-value native fish, creating economic issues for traditional fisheries. The study highlights the urgent need for integrated management strategies that include ecological monitoring, targeted removal, habitat restoration, and policy regulation. Early action is essential to reduce long-term impacts and develop a sustainable approach that balances conservation with local livelihoods.
Acknowledgements
The authors are grateful to ICAR, New Delhi and the Director, ICAR-CIFRI for the approval and support to conduct the research. The authors thank the institute personnel who assisted during the research. The cooperation rendered by the fishers and local authority at sampling sites during sample collection is gratefully acknowledged.
Funding
The work was carried out under an institutionally funded project ‘RWF/20-23/10: Understanding spatio-temporal variations of reservoir ecosystems and developing improved fisheries management strategies for different eco-regions: a new perspective’ of ICAR- Central Inland Fisheries Research Institute, Barrackpore, India.
Data availability statement
The data supporting the findings of this study are included within the article. Additional data are available from the corresponding author upon reasonable request.
References
- Agostinho AA, Pelicice FM, Gomes LC. 2008. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol 68: 1119–1132. [Google Scholar]
- APHA. Standard methods for the examination of water and wastewater, 23rd edn. Washington DC: American Public Health Association, 2017. [Google Scholar]
- Azzurro E, Tuset VM, Lombarte A, Maynou F, Simberloff D, Rodríguez‐Pérez A, Solé RV. 2014. External morphology explains the success of biological invasions. Ecol Lett 17: 1455–1463. [Google Scholar]
- Baran E, Jantunen T, Chong CK. 2007. Values of Inland Fisheries in the Mekong River Basin. WorldFish Center, Phnom Penh, 2007, p. 76. [Google Scholar]
- Bednarska A. 2006. Adaptive changes in morphology of Daphnia filter appendages in response to food stress. Polish J Ecol 54: 663–668. [Google Scholar]
- Bellinger EG, Sigee DC. Freshwater Algae: Identification and Use as Bioindicators. Wiley-Blackwell, Oxford, 2010. [Google Scholar]
- Bowen SH. 1982. Feeding, digestion and growth—qualitative considerations. pp. 141–156, in: Pullin RSV, Lowe-McConnell RH (Eds.), The Biology and Culture of Tilapias. ICLARM Conf Proc 7: 432. [Google Scholar]
- Bradford M, Kroon FJ, Russell DJ. 2011. The biology and management of Tilapia mariae (Pisces: Cichlidae) as a native and invasive species: a review. Mar Freshw Res 62: 902–917. [Google Scholar]
- Britton JR, Davies GD, Harrod C. 2010. Trophic interactions and consequent impacts of the invasive fish Pseudorasbora parva in a native aquatic food web: a field investigation in the UK. Biol Invas 12: 1533–1542. [Google Scholar]
- Bury NR, Eddy FB, Codd GA. 1995. The effects of the cyanobacterium Microcystis aeruginosa, the cyanobacterial hepatotoxin microcystin–LR, and ammonia on growth rate and ionic regulation of brown trout. J Fish Biol 46: 1042–1054. [Google Scholar]
- Bwanika GN, Chapman LJ, Kizito Y, Balirwa J. 2006. Cascading effects of introduced Nile Perch (Lates niloticus) on the foraging ecology of Nile tilapia (Oreochromis niloticus). Ecol Freshw Fish 15: 470–481. [Google Scholar]
- Canonico GC, Arthington A, McCrary JK, Thieme ML. 2005. The effects of introduced tilapias on native biodiversity. Aquat Conserv: Mar Fresh Ecosyst 15: 463–483. [Google Scholar]
- Chen J, Xie P, Zhang D, Ke Z, Yang H. 2006. In situ studies on the bioaccumulation of microcystins in the phytoplanktivorous silver carp (Hypophthalmichthys molitrix) stocked in Lake Taihu with dense toxic Microcystis blooms. Aquaculture 261: 1026–1038. [Google Scholar]
- Costa-Pierce BA. 2003. Rapid evolution of an established feral tilapia (Oreochromis spp.): the need to incorporate invasion science into regulatory structures. Biol Invasions 5: 71–84. [CrossRef] [Google Scholar]
- Coward K, Little DC. 2001. Culture of the ‘aquatic chicken’: present concerns and future prospects. Biologist (London, England) 48: 12–16. [Google Scholar]
- Cucherousset J, Olden JD. 2011. Ecological impacts of non-native freshwater fishes. Fisheries 36, 215–230. [CrossRef] [Google Scholar]
- Cucherousset J, Olden JD. 2020. Are domesticated freshwater fish an underappreciated culprit of ecosystem change? Fish Fish 21: 1253–1258. [Google Scholar]
- De Silva SS, Nguyen TTT, Turchini GM, Amarasinghe US, Abery NW. 2009. Alien species in aquaculture and biodiversity: a paradox in food production. Ambio 38: 24–28. [Google Scholar]
- Deines AM, Wittmann ME, Deines JM, Lodge DM. 2016. Tradeoffs among ecosystem services associated with global tilapia introductions. Rev Fish Sci Aquac 24: 178–191. [Google Scholar]
- Douthwaite RJ, Jones EW, Tyser AB, Vrdoljak SM. 2018. The introduction, spread and ecology of redclaw crayfish Cherax quadricarinatus in the Zambezi catchment. Afr J Aquat Sci 43: 353–366. [Google Scholar]
- Dunn T. 1994. Rapid Rural Appraisal: a description of the methodology and its application in teaching and research at Charles Sturt University. Rural Soc. 4: 30–36. [Google Scholar]
- Eloranta AP, Nieminen P, Kahilainen KK. 2014. Trophic interactions between introduced lake trout (Salvelinus namaycush) and native Arctic charr (S. alpinus) in a large Fennoscandian subarctic lake. Ecol Freshw Fish 24:181–192. [Google Scholar]
- Elton CS. The Ecology of Invasions by Animals and Plants. Methuen, London, 1958. [Google Scholar]
- FAO. 2022. Promoting sustainable aquaculture for food security and economic development. Harare. https://doi.org/10.4060/cc0324en [Google Scholar]
- Getachew T. 1987. A study on a herbivorous fish. Oreochromis niloticus L., diet and its quality in two Ethiopian Rift valley lakes, Awasa and Zwai. J Fish Biol 30: 439–449. [Google Scholar]
- Gomes LC, Miranda LE. 2001. Riverine characteristics dictate composition of fish assemblages and limit fisheries in reservoirs of the upper Paraná River basin. Regul Rivers: Res Manag 17: 67–76. [Google Scholar]
- Gozlan RE, Britton JR, Cowx I, Copp GH. 2010. Current knowledge on non-native freshwater fish introductions. J Fish Biol 76: 751–786. [CrossRef] [Google Scholar]
- Hamid MA, Sah ASRM, Idris I, Nor SAM, Mansor M. 2023. Impacts of tilapia aquaculture on native fish diversity at an ecologically important reservoir. PeerJ 11: e15986. [Google Scholar]
- Havel JE, Lee CE, Vander Zanden JM. 2005. Do reservoirs facilitate invasions into landscapes? BioScience 55: 518–525. [Google Scholar]
- Hooper DU, Chapin III FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawton JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setälä H, Symstad AJ, Vandermeer J, Wardle DA. 2005. Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75: 3–35. [Google Scholar]
- Hyslop EJ. 1980. Stomach contents analysis—a review of methods and their application. J Fish Biol 17: 411–429. [CrossRef] [Google Scholar]
- Johnson C, Sarkar UK, Koushlesh SK, Das AK, Das BK, Naskar BK. 2022. Fish assemblage, ecosystem status and potential impact of Nile Tilapia in Halali Reservoir of Central India. Environ Dev Sustain 24: 7753–7775. [Google Scholar]
- Kamjunke N, Schmidt K, Pflugmacher S, Mehner T. 2002. Consumption of cyanobacteria by roach (Rutilus rutilus): useful or harmful to the fish? Freshw Biol 47: 243–250. [Google Scholar]
- Kang B, Vitule JR, Li S, Shuai F, Huang L, Huang X, Fang J, Shi X, Zhu Y, Xu D, Yan Y. 2023. Introduction of non‐native fish for aquaculture in China: a systematic review. Rev Aquac 15: 676–703. [Google Scholar]
- Kolar CS, Lodge DM. 2001. Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204. [Google Scholar]
- Kolding J, van Zwieten P, Mkumbo O, Silsbe G, Hecky R. Are the Lake Victoria fisheries threatened by exploitation or eutrophication? Towards an ecosystem based approach to management, in: Bianchi G, Skjoldal HR (Eds.), The ecosystem Approach to Fisheries. CABI , Oxfordshire, UK, 2008, pp. 309–354. [Google Scholar]
- Landsberg JH. 2002. The effects of harmful algal blooms on aquatic organisms. Rev Fish Sci 10: 113–390. [Google Scholar]
- Leprieur F, Beauchard O, Blanchet S, Oberdorff T, Brosse S. 2008. Fish invasions in the world's river systems: when natural processes are blurred by human activities. PLoS Biol 6: e322. [CrossRef] [Google Scholar]
- Levine JM, Adler PB, Yelenik SG. 2004. A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7: 975–989. [Google Scholar]
- Lowe MR, Wu W, Peterson MS, Brown-Peterson NJ, Slack WT, et al. 2012. Survival, growth and reproduction of non-native Nile Tilapia II: fundamental niche projections and invasion potential in the Northern Gulf of Mexico. PLOS One 7: e41580. [Google Scholar]
- Manko P. 2016. Stomach content analysis in freshwater fish feeding ecology. Prešov: University of Prešov. ISBN 978-80-555-1613-4. [Google Scholar]
- Matsuzaki SS, Sasaki T, Akasaka M. 2013. Consequences of the introduction of exotic and translocated species and future extirpations on the functional diversity of freshwater fish assemblages. Glob Ecol Biogeogr 22: 1071–1082. [Google Scholar]
- Moriarty DJW. 1973. The physiology of digestion of blue-green algae in the cichlid fish, Tilapia nilotica. J Zool 171: 25–39. [Google Scholar]
- Moriarty CM, Moriarty DJW. 1973a. Quantitative estimation of the daily ingestion of phytoplankton by Tilapia nilotica and Haplochromis nigripinnis in Lake George, Uganda. J Zool Lond 171: 15–23. [Google Scholar]
- Moriarty DJW, Moriarty CM. 1973b. The assimilation of carbon from phytoplankton by two herbivorous cichlid fishes: Tilapia nilotica and Haplochromis nigripinnis. J Zool Lond 171: 41–45. [Google Scholar]
- Moss B. 2008. The kingdom of the shore: achievement of good ecological potential in reservoirs. Freshwat Rev 1: 29–42. [Google Scholar]
- Nambeye E, Katapa C, Mulenga BC, Mudenda HG, Eilitta M, Rice MA. 2025. The bio-invasion and population dynamics of Cherax quadricarinatus in Zambian waters. Afr J Aquat Sci 50: 88–98. [Google Scholar]
- O'Mara K, Venarsky M, Marshall J, Stewart-Koster B. 2024. Diet-habitat ecology of invasive tilapia and native fish in a tropical river catchment following a tilapia invasion. Biol Invasions 26: 489–504. [Google Scholar]
- Padilla DK, Williams SL. 2004. Beyond ballast water: aquarium and ornamental trades as sources of invasive species in aquatic ecosystems. Front Ecol Environ 2: 131–138. [Google Scholar]
- Panikkar P, Khan MF. 2008. Comparative mass-balanced trophic models to assess the impact of environmental management measures in a tropical reservoir ecosystem. Ecological Model 212(3–4): 280–291. [Google Scholar]
- Panikkar P, Khan MF, Sarkar UK, Das BK. 2021. Changing foodwebs of Indian aquatic ecosystems under the threats of invasive species: an overview. Aquat Ecosyst Health Manag 24(2), 24–32. DOI: 10.14321/aehm.024.02.06 [Google Scholar]
- Peng L, Xue X, Liao J, Zhao J, Tang Q, Lin Q, Zhang Q, Han B-P. 2021. Potential impact of population increases of non-native tilapia on fish catch and plankton structure: a case study of Tangxi Reservoir in southern China. Aquat Invasions 16: 329–348. [Google Scholar]
- Philippart JC, Ruwet JC. Ecology and distribution of tilapias, in: Pullin RSV, Lowe-McConnell RH (Eds.), The Biology and Culture of Tilapias. International Center for Living Aquatic Resources Management (ICLARM), Manila 1982, pp. 15–59. (ICLARM Conference Proceedings, 7). [Google Scholar]
- Pido MD, Pomeroy RS, Garces LR, Carlos MB. 1997. A rapid appraisal approach to evaluation of community‐level fisheries management systems: framework and field application at selected coastal fishing villages in the Philippines and Indonesia. Coast Manag 25: 183–204. [Google Scholar]
- Prescott GW. How to Know the Freshwater Algae, 3rd ed. W. M.C. Brown Company Publishers, Dubuque, 1978. [Google Scholar]
- Reardon EE, Chapman LJ. 2008. Reproductive seasonality in a swamp‐locked African cichlid. Ecol Freshw Fish 17: 20–29. [Google Scholar]
- Rice MA. 2021. Intensive fishing effort and market controls as management tools for invasive aquatic species: a review. Asian Fish Sci 34: 383–392. [Google Scholar]
- Russell DJ, Thuesen PA, Thomson FE. 2012. Reproductive strategies of two invasive tilapia species Oreochromis mossambicus and Tilapia mariae in northern Australia. J Fish Biol 80(6): 2176–2197. doi: 10.1111/j.1095-8649.2012.03267.x. [Google Scholar]
- Sanderson SL, Cheer AY, Goodrich JS, Graziano JD, Callan WT. 2001. Crossflow filtration in suspension-feeding fishes. Nature 412(6845): 439-441. [CrossRef] [PubMed] [Google Scholar]
- Sarkar UK, Pathak AK, Lakra WS. 2008. Conservation of freshwater fish resources of India: new approaches, assessment and challenges. Biodivers. Conserv 17(10): 2495–2511. [Google Scholar]
- Sarkar UK, Sandhya KM, Mishal P, Karnatak G, Lianthuamluaia, Kumari S, Panikkar P, Palaniswamy R, Karthikeyan M, Mol SS, Paul TT. 2018. Status, prospects, threats, and the way forward for sustainable management and enhancement of the tropical Indian reservoir fisheries: an overview. Rev Fish Sci Aquac 26: 155–175. [Google Scholar]
- Schoener TW. 1970. Nonsynchronous spatial overlap of lizards in patchy habitats. Ecology 51: 408–418. [Google Scholar]
- Semyalo R. 2009. The effect of cyanobacteria on the growth, survival, and behaviour of tropical fish (Nile tilapia) and zooplankton (Daphnia lumholtzi), PhD thesis, University of Bergen, Norway. [Google Scholar]
- Shuai F, Li J, 2022. Nile tilapia (Oreochromis niloticus Linnaeus, 1758) invasion caused trophic structure disruptions of fish communities in the south China river—Pearl River. Biology 11: 1665. [Google Scholar]
- Shuai F, Li X, Liu Q, Zhu S, Wu Z, Zhang Y. 2019. Nile tilapia (Oreochromis niloticus) invasions disrupt the functional patterns of fish community in a large subtropical river in China. Fish Manag Ecol 26: 578–589. [Google Scholar]
- Srivastava A. Inland Fish Marketing in India. Indian Institute of Management, Ahmedabad, 1985. [Google Scholar]
- Sugunan VV. Reservoir fisheries of India. FAO Fisheries Technical Paper. No. 345. Rome: FAO, 1995, p. 423. [Google Scholar]
- Temesgen M, Getahun A, Lemma B, Janssens GPJ. 2022. Food and feeding biology of Nile Tilapia (Oreochromis niloticus) in Lake Langeno, Ethiopia. Sustainability 14: 974. [Google Scholar]
- Tillmanns AR, Wilson AE, Pick FR, Sarnelle O. 2008. Meta-analysis of cyanobacterial effects on zooplankton population growth rate: species-specific responses. Fundam Appl Limnol 171: 285–295. [Google Scholar]
- Trewavas E. Tilapiine fishes of the genera Sarotherodon, Oreochromis and Danakilia. London: British Museum (Natural History), 1983. [Google Scholar]
- van Zwieten PA, Béné C, Kolding J, Brummett R, Valbo-Jørgensen J. 2011. Review of tropical reservoirs and their fisheries: the cases of Lake Nasser, Lake volta and Indo-Gangetic Basin reservoir. FAO Fisheries & Aquaculture Technical Paper. Food and Agriculture Organization of the United Nations, Rome. [Google Scholar]
- Wilson AE, Sarnelle O, Tillmanns AR. 2006. Effects of cyanobacterial toxicity and morphology on the population growth of freshwater zooplankton: meta-analyses of laboratory experiments. Limnol Oceanogr 51: 1915–1924. [Google Scholar]
- Xiong W, Guo C, Gozlan RE, Liu J. 2023. Tilapia introduction in China: economic boom in aquaculture versus ecological threats to ecosystems. Rev Aquacult 15: 179–197. [Google Scholar]
- Yongo E, Zhang P, Mutethya E, Zhao T, Guo Z. 2023. The invasion of tilapia in South China freshwater systems: a review. Lakes Reserv Res Manag 28: e12429. [Google Scholar]
- Zambrano L, Martínez-Meyer E, Menezes N, Peterson AT. 2006. Invasive potential of common carp (Cyprinus carpio) and Nile tilapia (Oreochromis niloticus) in American freshwater systems. Can J Fish Aquat Sci 63: 1903–1910. [Google Scholar]
- Zhang J, Deng R, Liang H, Yang J, 2017a. A study of well test data interpretation model for waterbearing reservoirs with phase redistribution. IOP Conf Ser: Earth Environ Sci 64: 012019. [Google Scholar]
- Zhang X, Mei X, Gulati RD. 2017b. Effects of omnivorous tilapia on water turbidity and primary production dynamics in shallow lakes: implications for ecosystem management. Rev Fish Biol Fisheries 27: 245–254. [Google Scholar]
Cite this article as: Sajina AM , Vaisakh G, Lohit Kumar K, Kumari S, Das AK, Manna RK, Solanki JK, Ali Y, Das BK. 2026. Introduction pathways and ecological impacts of Nile tilapia in a large tropical reservoir of India: implications for management. Aquat. Living Resour. 39: 10. https://doi.org/10.1051/alr/2026003
All Tables
List of fish species recorded from Panam Reservoir, their family, fishery group/category, and observed mean total length (TL ± SD) in landings (2022–2023).
Summary of RRA responses on invasion pathway and impacts of O. niloticus in Panam Reservoir.
Gut content composition (%) of Nile tilapia and selected native fishes from the reservoir.
All Figures
 |
Fig. 1 Map showing the location of Panam Reservoir in Gujarat, India, with designated fish sampling points. |
| In the text | |
 |
Fig. 2 Comparative percentage composition of major fish groups in the catch from Panam Reservoir during 2022 and 2023. |
| In the text | |
 |
Fig. 3 Phytoplankton composition in Panam Reservoir. Left: Major classes showing Cyanophyceae dominance. Right: Genera composing the Cyanophyceae community are dominated by Anabaena and Microcystis. |
| In the text | |
 |
Fig. 4 Schoener's index values showing dietary overlap between Nile tilapia and native fishes in Panam Reservoir. |
| In the text | |
 |
Fig. 5 CCA biplot depicting the association of environmental variables with the distribution of the ten most abundant fish species in Panam Reservoir. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


