| Issue |
Aquat. Living Resour.
Volume 36, 2023
|
|
|---|---|---|
| Article Number | 33 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/alr/2023029 | |
| Published online | 20 December 2023 | |
Research Article
Reproduction patterns of the bloody cockle Senilia senilis (Linnaeus 1758) in the Sine-Saloum inverse estuary
1
IRD, Univ Brest, Ifremer, CNRS, LEMAR, 29280, Plouzané, France
2
Université Cheikh Anta Diop Dakar, département de biologie animale, Dakar, Sénégal
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Pauline Kamermans
Received:
15
September
2023
Accepted:
24
November
2023
Abstract
Understanding the reproductive biology of a species is an important means of determining the renewal capacity of natural stocks, especially in the case of heavily exploited species. It is a fundamental element in supporting the implementation of management measures. Here, we studied the bloody cockle (S. senilis) in the Sine-Saloum, with the aim of describing its seasonal and spatial reproductive cycle. S. senilis reproduction was studied over an annual cycle at two sites chosen for their contrasting situations along the upstream-downstream gradient. The reproductive cycle was studied by histological analysis of a pool of individuals maintained in-situ and sampled throughout the year. Our results showed that gamete maturation is asynchronous within and between individuals. Gametogenesis mostly occurred in October. The maturation stage showed a seasonal pattern with continuous reproduction throughout the year, with two preferred periods between May and July and December and February. The reproductive cycle is highly dependent on temperature and salinity variations, resulting in a seasonal cycle and spatial heterogeneity. The temperature induces gametogenesis and salinity synchronizes the spawning periods.
Key words: Artisanal fishery / bivalve / reproduction / monsoon / inverse estuary / histology
© B. Sané et al., Published by EDP Sciences 2023
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
The Sine-Saloum delta is a mangrove ecosystem located along the Atlantic Ocean in West Africa, in Senegal (Fig. 1; 14° 11′ N, 16° 15′ O). The only freshwater input is rainfall during the monsoon season, which runs from July to October. It is subject to spatial and seasonal variations in salinity. During the dry season, from November to June, the highest salinities are recorded upstream due to evaporation (Descroix et al., 2020). This justifies its qualification as an inverse estuary (Pages and Citeau, 1990). The amplitude of seasonal salinity variability increases with distance from the ocean. The lowest salinity of 40 is recorded during the rainy season upstream, at Kaolack, around 100 km from the mouth of the delta, while in the dry season it can exceed 100 (Descroix et al., 2020).
This ecosystem is home to a wide diversity of molluscs such as oysters (Crassostrea tulipa), “Yett” (Cymbium spp.), “Touffa” (Murex spp., Thais spp.) and bloody cockle “Pagne” (Senilia senilis) (Camara et al., 2017). The bloody cockle S. senilis (Linnaeus, 1758) is a key resource for local communities. This species has been harvested artisanally for millennia, as evidenced by the presence of numerous shell middens (Carré et al., 2022; Hardy et al., 2016). This shellfish is fished mainly by women, historically for food purposes. Today, the accelerated exploitation of this bivalve no longer follows a logic of self-consumption, but is also part of a marketing chain, and as such constitutes an important source of income for women of the Sine-Saloum delta. Agricultural activities have been abandoned in favor of fishing due to the salinization of the soil (Dia, 2003). Fishing yields have been declining for a decade and the size of the individuals caught tends to decrease due to the pressure and over-salting of estuarine waters. In addition to these various constraints, the bloody cockle is also impacted by the effects of climate change, such as the disruption of rainfall patterns and rising water temperatures (Diouf and Sarr, 2014).
From a sustainable development perspective, understanding the reproductive biology of this species is an important means of determining the renewal capacity of natural stocks and the parameters that can affect population renewal. It is a fundamental element in supporting the implementation of management measures. The description of the reproductive cycle of S. senilis has not yet been the subject of specific work in the Sine-Saloum delta. The mode of reproduction and the effect of environment parameters remains uncertain. While some authors, such as Yoloye (1974), support hermaphroditism, others, such as Yankson (1982), demonstrate the opposite.
The reproduction of marine bivalves is favored by optimal environmental parameters and can be compromised under stressful conditions (Vázquez et al., 2021). Reproduction is especially influenced by environmental parameters such as temperature, salinity and food availability (Broom, 1983). Food modulates gamete quality and quantity (Utting and Millican, 1997). Temperature is involved in germ cell maturation and spawning initiation (Fabioux et al., 2005; Hadfield and Anderson, 1988). In tropical environments, seasonal fluctuations in water temperature are less important, but salinity variations can be significant and periodic, especially in ecosystems subject to the influence of the monsoon (Baron and Clavier, 1992).
Some oyster species of the genus Crassostrea spawn at the start of the monsoon season when water salinity decreases (Sühnel et al., 2023; Barman et al., 2022; Paixão et al., 2013; Stephen, 1980). A similar pattern is also observed in the Anadara genus, which is phylogenetically related to the bloody cockle (Manjarrés-Villamil et al., 2013; Broom, 1983). We can therefore ask whether a similar phenology is observed in the bloody cockle S. senilis due to salinity fluctuations occurring in the Sine-Saloum delta.
The present study aims (1) describing the seasonal reproductive cycle of S. senilis and (2) assessing the existence of a spatial contrast in the Sine-Saloum delta. The reproductive cycle was studied in-situ by histology over 1 yr period at two sites chosen for their contrasting situations along the upstream-downstream gradient and representative of the main fishing grounds. The obtained results make it possible to describe spatial and temporal patterns for various descriptors such as sex ratio, reproductive investment and the evolution of maturity stages. Finally, the measurement of key environmental parameters enables us to propose scenarios for the environmental determinism of the reproductive cycle of S. senilis in the Sine-Saloum region.
2 Material and methods
2.1 Study site
The study sites were chosen close to two villages in the Sine-Saloum delta: Niodior and Falia. These two sites, which are artisanal shellfish fishing areas, show contrasting oceanic influences: the village of Niodior, located downstream opposite the Sangomar breach, is significantly influenced by oceanic waters, whereas the village of Falia, located upstream in the heart of the mangrove swamp, is less influenced by oceanic waters (Fig. 1). Both experimental sites are located in the intertidal zone. Water level at high tide on both sites is between 80 cm and 100 cm, depending on neap or spring tides. Sane et al., unpublished data. In the following, the two sites will be referred to as “upstream site” for Falia and “downstream site” for Niodior.
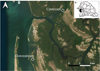 |
Fig. 1 (A) Location of the Sine-Saloum delta on the map of Senegal and (B) location of the experimental monitoring study sites. |
2.2 Experimental system
At each site, an intertidal mudflat has been identified to house an experimental park. An area of 16 m2 was marked out with wire mesh and wooden stakes, and the surface was completely cleared of all bloody cockles. At each site, 500 individuals with a shell height (antero-posterior dimension) between 22 mm and 24 mm were sampled in the vicinity and seeded in the park. This size class was selected to correspond to mature individuals, the size of first maturity being estimated at 20 mm (Tito De Morais, 2011; Yankson, 1982). The densities obtained were therefore 31 ind.m−2, which ensures that there is no effect of density on individual performances (Diouf and Sarr, 2014). Mortalities were comparable between the two sites, although a precise measurement was not made due to the difficulties in identifying dead individuals. We did not replace individuals that died during the experiment.
2.3 Monitoring strategy
Sampling was carried out every three to six weeks between March 2021 and March 2022. More frequent (three weeks) sampling was carried out between July and September, corresponding to the intense monsoon period. For each sampling, 10 individuals were randomly caught from the seeded individuals at each experimental park. It should be noted that due to the limited number of individuals at the end of the experiment, two dates could not be sampled at the upstream site (Falia) at the end of the period (February and March). The height and total fresh mass of each individual sampled were measured. The bloody cockles were then carefully opened with a scalpel and the flesh of the animals was fixed individually in Davidson's solution for 48 h (Howard and Smith, 1983). Samples were then transferred to a 70% ethanol solution until histological process.
2.4 Histological analyses
As the gonad is diffused around the digestive gland in this species (Yankson, 1982), a cut was made through the body mass to obtain a cross-section of the gonad, digestive gland and muscular tissues of the foot (Fig. 2A). The tissue section was then placed in a cassette and dehydrated in ethanol baths of increasing concentration (80, 95, 95, 95, 100, 100, 100%). Samples were then cleaned with xylene before being embedded in paraffin wax. Five-micrometer thick sections were cut with a microtome and mounted on glass slides. The sections were then rehydrated in series of decreasing concentration ethanol (100, 100, 96, 70%), stained with aqueous hematoxylin-Eosin Y and covered with a coverslip (Fabioux et al., 2005). The sections were then digitized under a microscope (ZEISS Stemi 508) at x60 equipped with a camera (AxioCam MRc5) (Fig. 2B).
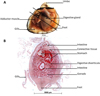 |
Fig. 2 (A) Anatomy of the S. senilis bloody cockle: black lines show the cut section; (B) Microscopic illustration of the transversal cut section. |
2.5 Sex-ratio determination
In order to differentiate the sex of each individual, the gonadal structure was examined to identify the presence of male or female germ. Individuals showing both distinct male and female germ cells were classified as hermaphrodites, while those whose sex could not be distinguished due to the absence of sexually differentiated germ cells were classified as indeterminate.
2.6 Reproductive investment
Reproductive investment is estimated by the gonadal index (GI), which represents the ratio of gonadal area to total histological section area. This is a commonly used method for assessing maturity in bivalves (e.g. Fabioux et al., 2005; Wilson, 1987; Barber and Blake, 1983). Indeed, the surface area occupied by the gonad increases as the gonadal tubules develop during the gametogenesis process (Broquard, 2019). Surface areas were determined by image analysis using ImageJ software (Schneider et al., 2012). The gonad and the entire section were delimited by manual contouring, and surfaces were extracted automatically enabling the calculation of GI, such as:

2.7 Determination of maturity stage
In order to more accurately describe the annual dynamics of bloody cockle reproduction, we studied the evolution of the germ cell stages in the gonadal region at both sampling sites and for the entire period sampled. A classification of the observed stages of development of germ cells was established from all the samples, based on analysis of the structure and morphology of the male and female germ cells. Thus, 5 reproductive stages were identified (Tab. 1 and Fig. 3). The different stages of maturation were identified on the basis of the work of Suwanjarat et al. (2009) and Freites et al. (2010) and adapted according to the histological observations made.
Histological examination showed an asynchronous evolution of gametogenesis between different regions of the gonad of the same individual, with tubules at different stages of development. Based on the 5 defined stages, the maturity level of each individual was determined by image analysis, identifying the contribution of each reproductive stage to the total gonad surface area. For each histological section, the area occupied by each stage was delimited by a polygon using ImageJ software (illustrated in Fig. 4). The proportion of each stage was calculated as the ratio between the surface area of one stage and the sum of the surface area of all stages. This analysis was carried out for the 10 individuals of each sampling time, at each site. Results were expressed as the average proportion of each stage for all individuals over a sampling date.
Description and classification criteria for the reproductive stages of Senilia senilis.
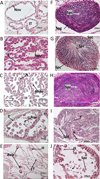 |
Fig. 3 Photographs of gonadal structures illustrating the 5 stages of maturation identified for males (A-E) and females (F-J) of Senilia senilis: (A) and (F) early gametogenesis (stage 1), (B) and (G) active gametogenesis (stage 2), (C) and (H) ripeness (stage 3), (D) and (I) partial spawning (stage 4), (E) and (J) total spawning or rest (stage 5). Legends: ET empty tubule; H hemocyte; N nucleus; Nov non-mature oocyte; Mov mature oocyte; Rov residual oocyte; Spg spermatogonia; Spc spermatocyte; Spz spermatozoid; T tubule. |
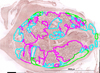 |
Fig. 4 Example of surface delimitation of the different maturation stages for one S. senilis histological section. Each color represents a stage: pink corresponds to the stage of ripeness (stage 3), blue to partial spawning (stage 4) and green to total spawning or rest (stage 5). |
2.8 Measurement of environmental parameters
Throughout the sampling period, water column temperature (±0.1 °C), pressure (±0.2 cmH2O) and conductivity (±4 ms.cm−1) were recorded by a CTD-Divers probe (vanEssen Instruments) placed a few centimeters from the sediment. Salinity was obtained by converting conductivity using the gsw_SP_from_C package on R software (Hill et al., 1986). In addition to the CTD probe, a Wimo probe (NKE Instrumentation), equipped with a salinity and temperature sensor, was placed at the upstream park. For this study, measurements were recorded every 10 min. Due to significant fouling episodes on the sensors, particularly at the upstream site, only usable data were retained, combining data from both sensors. Data processing was carried out to retain only the temperatures recorded during immersion, based on pressure measurements.
Immersion time was determined using pressure measurements. Immersion periods were identified by applying a threshold to exclude periods of emersion. An adjustment was made to the emersion time to account for the 15 cm distance between the sensor and the sediment. The immersion rate was computed as the ratio of immersion time to the total recording duration.
2.9 Statistical analyses
The Chi2 test was used to test if the sex ratio of the studied populations at the two sites showed a sex ratio different from 1:1. All analyses were performed using R software (R Core Team, 2020).
3 Results
3.1 Sex-ratio
Out of a total of 190 individuals analyzed, 8 were classified as indeterminate and 1 as hermaphroditic at the downstream site (Niodior) and 9 as indeterminate and 3 as hermaphroditic at the upstream site (Falia).
At the upstream site, the percentage of females and males were 51.9% and 49.1%, respectively, but the sex-ratio was not significantly different from 1:1 (Chi-2 test, p-value = 0.3). The downstream site showed 65.3% females versus 34.7% males. The sex ratio significantly favors females (Chi-2 test, p-value = 0.002).
3.2 Seasonal and spatial patterns
3.2.1 Environmental parameters
The recorded immersion rate from July 2021 to March 2022 at the downstream location showed a lower value of 67% in comparison to the 74% measured upstream. During the monitoring year, water temperatures appeared very similar at both sites, with the same seasonal pattern. The highest temperatures were recorded during the monsoon season between June and November. They exceeded 28 °C during this period, with a maximum of 31.8 °C (±0.6 °C) recorded in October (Fig. 4A). The dry-season period from November to March was marked by the lowest temperatures at both sites, with a sharp drop recorded in December: 3 °C in 2 days. The minimum temperature recorded during this period was 21.4 °C (±0.6 °C) (Fig. 4A).
Salinity also showed a seasonal pattern marked by two periods of high salinity decrease: an initial low salinity period between April and July, and a second, more significant during the monsoon season, between August and November (Fig. 4B). Salinity varied between 23.8 (±0.2) and 38.08 (±0.2) downstream and between 18.4 (±0.6) and 38.6 (±0.6) upstream. The amplitude of salinity variation was greater upstream, around 20.2 compared with 14.2 downstream. The return period to initial salinities appeared longer upstream than downstream (Fig. 4B).
3.2.2 Reproductive investment
An increase in mean gonadal index was recorded during the monsoon period between July and October for both sites (Fig. 4C). The maximum GI percentage was reached in October during the period of low salinity and high temperature. This phase of increase was followed by a period of sharp decrease in GI from October to November. The minimum GI (18%) was reached in December at both upstream and downstream sites. Between March and July, the variation in gonad index was relatively low, even during periods of low salinity. The area occupied by gonads thus varied between 18% and 40% downstream and between 18% and 30% upstream (Fig. 4C).
3.2.3 Maturation stages
Figure 4D represents the frequency of each maturation stage at each date downstream and upstream. Qualitative analysis of the maturation stage showed a seasonal pattern characterized by total or partial spawning events (stages 4 and 5) throughout the year, but with two main peaks (Fig. 4D). The first peak, less significant in terms of total spawning, occurs during the first low salinity period, between May and July. This spawning event was followed by an increase in gametogenesis (stage 2) and maturity (stage 3) stages during the monsoon, between July and October, at both sites. The maximum number of mature tubules was recorded in October. However, the percentage of mature tubules was lower upstream (20%) than downstream (50%). The second spawning peak occurred after the monsoon, between December and February, when the temperature dropped and salinity returned to its initial value recorded in July. The percentage of tubules on total spawning or rest (stage 5) during this period was more important downstream than upstream. In December, 98% of tubules downstream were in the total spawning or rest stage, whereas it was only 75% upstream.
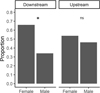 |
Fig. 5 Proportion of males and females at each sampling site: ns indicates a non-significant difference between males and females (Chi-2 test, p > 0.05); * indicates a significant difference between males and females (Chi-2 test, p < 0.05). |
 |
Fig. 6 Variation of (A) temperature, (B) salinity, (C) gonad index and (D) frequency of each maturation stage upstream and downstream, measured between March 2021 and March 2022. |
4 Discussion
4.1 The sex-ratio
In some bivalves, the predominance of males or females may be due to variations in environmental conditions (Chahouri et al., 2022). Yankson (1982) reported that in a population of bloody cockles from Ghana, the sex ratio was balanced in the open lagoon and significantly in favor of females at salinities >50 and surface water temperatures of 32-34 °C. Another example, in pearl oysters Pinctada mazatlanica and Pinctada margaritifera, showed a balanced sex ratio when the animals were in natural conditions, but under farmed conditions, the sex ratio was in favor of males (Saucedo and Southgate 2008). As in the latter's work, we showed a very low percentage of individuals with simultaneous hermaphroditism (4/200 individuals), for whom both male and female gonads were active and at different stages of development. Our results show that the upstream sex ratio is balanced between males and females. In contrast, the sex ratio was unbalanced in favor of females at the downstream site. The balanced sex-ratio seems in line with results reported for other species of the Anadara genus in various locations: A. granosa in Malaysia (Broom, 1983), A. antiquata in the Philippines (Toral-Barza and Gomez, 1985), in India (Narasimham, 1988), A. trapezia in Australia (Hadfield and Anderson, 1988), A. inaequivalvis on the southeast coast of the Black Sea (Sahin et al., 2006). This has also been observed for S. senilis by Yankson (1982) in Ghana, but our observations seem to contradict his observations, which indicate that more restrictive conditions, with higher salinities and temperatures, favored a sex ratio dominated by females. Our observations indicate that more oceanic conditions promote females in the Sine-Saloum delta. However, this observation needs to be validated. The main aim of our work was not to study the sex ratio. A broader sampling protocol would be needed to be representative of the population and to confirm or not our observations.
We have to note that there is an uncertainty about the reproduction mode, protandrous hermaphroditism or gonochoric, of S. senilis. Yoloye (1974) claimed that S. senilis was protandrous hermaphrodite. He showed in Lagos lagoon system that all individuals start life as males and close to 75% changed to females at 6 months old and about 19 mm long. He also noted that most individuals undergo only one sex change during their life cycle. In contrast, Yankson (1982) showed strong evidence that A. senilis, like A. granosa and A. trapezia, was basically gonochoristic. Our results are inconclusive on this point, although they seem to confirm the observations made by Yankson (1982). Firstly, we used animals between 22 and 24 mm long, considered as sexually mature and we found more than 30% of males in each sampling site, which does not appear in accordance with the Yoloye hypothesis (Yoloye, 1974). We also found no evidence of transition or sex reversal. Although the immersion time is slightly longer upstream, growth disparities exist between the two sites, with individuals upstream growing more slowly than those downstream (Diouf and Sarr, 2014 and unpublished personal data). These variations result in a situation where upstream individuals are older for an equivalent size. Therefore, it appears that the individuals monitored downstream are generally younger, further reinforcing our disagreement with the protandrous hermaphroditism hypothesis (especially due to the higher prevalence of females downstream).
4.2 Reproductive phenology
For the present work, we established a classification of maturity stages for S. senilis, which was not available in the literature. Total spawning and resting stages were combined, as resting was poorly or not at all visible. The gametogenesis stage was subdivided into two stages: early gametogenesis and advanced gametogenesis, depending on the presence or absence of spermatozoa in the tubule. Our work thus provides a classification of germ cell developmental stages for S. senilis that can be used for future work on the species. The reproductive cycle of S. senilis showed asynchronous maturation of the gonads and between individuals. In the vast majority of individuals, within a single individual, the gonad comprises tubules at different stages of gametogenesis advancement. For this reason, we have considered the average percentage of tubules at each stage for each date.
The results of the maturation cycle of S. Senilis at both sites showed that spawning takes place throughout the year. This is typical behavior for bivalves in tropical zones (Mahmoud and Yassien, 2022; Manjarrés-Villamil et al., 2013; Narasimham, 1988; Toral-Barza and Gomez, 1985). This is consistent with the results of Le Loeuff and Zabi (1993), who reported the year-round presence of S. senilis larvae in the Lagos lagoon in Nigeria. The year-round presence of bloody cockle larvae was also reported by Afinowi (1976) in Ghana and Nigeria. In addition to this typical behavior, we observed two main spawning peaks at both sites: the first, smaller peak between May and July, and the second, larger peak between December and February. This type of behavior has also been described for tropical bivalves that reproduce year-round with one or two peak clutches associated with specific environmental conditions (Ghribi et al., 2017; Sahin et al., 2006).
Temperature and salinity are very important factors influencing the reproductive cycle of bivalves. In tropical zones, the most intense spawning periods are frequently triggered by an increase in temperature (Rodríguez-Jaramillo et al., 2022; Jahangir et al., 2014; Ren et al., 2003; Massapina et al., 1999). In this study, the two periods of spawning were identified at temperatures around 26 °C and 24 °C (May and December), which do not correspond with the maximum temperatures observed (> 30 °C), so temperature does not seem to be the trigger for spawning of the bloody cockle in the Sine-Saloum delta. On the other hand, the two most intense spawning periods coincided with two periods of desalination. This observation therefore tends to confirm the hypothesis that the bloody cockle's reproductive cycle is influenced by variations in salinity. These results suggest an inverse relationship between water salinity and spawning in S. senilis, and are consistent with data from Okera (1976), according to which the bloody cockle reproduces after the monsoon in Sierra Leone. Freshwater intake during the monsoon season is also considered an influential factor on reproductive mechanisms in Anadara granosa in Malaysia (Broom, 1983) and India (Manjarrés-Villamil et al., 2013).
A salinity depression leads to growth and maturation of the gametes in A. granosa (Broom, 1983). Reproduction in this species occurs mainly when low-salinity conditions are maintained for a long period (Broom, 1982). It has also been shown that in Cerastoderma edule, an intertidal species that can live in salinities ranging from 18 to 40. Salinity changes of 5 to 20 and 10 to 25 in March and May, respectively, induced massive spawning followed by gonad recovery (Vázquez et al., 2021). These results differ, from those found by Debenay et al. (1994), who reported a maximum spawning period during the rainy season in the Mbodjiène lagoon in Senegal. In the present study, significant gametogenesis activity was recorded upstream and downstream during the monsoon season between July and October.
Qualitative analysis of maturation stages is very often coupled with more quantitative analysis. The most widely used method in the Anadara genus is the calculation of the condition index (Freites et al., 2010; Narasimham, 1988). Reproductive investment is widely used in oysters (Fabioux et al., 2005). Our results indicate that the period of gametogenesis and maturation observed from the identification of maturity stages coincides well with the phase of increasing gonadal index between July and October. This validates a methodological approach for this species, which is technically simple to implement. However, the percentage occupied by gonads was lower at the first low salinity period, between April and May, than during monsoon, between July and October. The difference between these two spawning episodes suggests that salinity is not the only driver of reproduction in this species. Indeed, between April and May, as the proportion of mature tubules is lower, there are quantitatively few spawning events, even during desalination. During this period, temperatures are still low. However, during the monsoon season, when temperatures are higher (>30 °C), significant gametogenesis activity was recorded upstream and downstream. Partial spawning also increases, then in October, when the majority of tubules are mature, the minimum salinity causes almost all the tubules to “empty”. High temperatures therefore seem to act as a primary driver, allowing the majority of individuals to mature, culminating in significant spawning when environmental conditions become unfavorable. In this way, high temperature synchronizes maturation and salinity triggers oviposition in S. senilis in the Sine-Saloum delta.
A comparison between the two sites shows that the surface area occupied by gonads is always smaller upstream, with the exception of September when gonad indices are equal. Spawning effort is therefore greater downstream than upstream. However, studying the effect of phytoplankton availability could help to understand this difference. It has been shown in some bivalves that food can improve fertility and gamete quality (Utting and Millican 1997) and reproductive investment (Enriquez-Diaz et al., 2009). This food availability could be seasonally limited in an inverse estuary (Villanueva, 2015). Initial data on phytoplankton communities' dynamics suggest a seasonal pattern, with densities increasing at the end of the monsoon season (Thomas and al., unpublished data). This would coincide with the period of highest reproductive investment that we have highlighted, offering favorable conditions for larval development and recruitment.
5 Conclusion
This work on S. senilis provided an initial description of the reproductive cycle of this bloody cockle in the Sine-Saloum delta. It appeared that reproduction takes place all year round, with two preferred periods between May and July and December and February. The results also showed a reproductive cycle marked by a strong dependence on salinity variations, resulting in a seasonal cycle and spatial heterogeneity. The study also enabled us to establish a standardized classification method for future studies with described stages, and a simple image analysis method for this species. In addition to qualitative analysis using maturity stages, quantitative analysis of reproductive effort by determining the gonadal index (GI) would also appear to be a simple tool for monitoring purposes, in addition to the condition index. With a view to assessing the effectiveness of the observed clutches, the monitoring of recruitment dynamics would enable us to identify whether there are more favorable periods for recruitment or whether it takes place all year round. Next step will also have to be focused on trophic aspects in order to highlight how food variability may drive reproductive patterns. The information provided by this study is a valuable contribution to our knowledge of the ecology of a key species in West African coastal ecosystems. A better understanding of the reproductive dynamics of S. senilis is a fundamental element to feed modelling approaches at individual and population scales, which are key steps in supporting management measures.
Acknowledgements
This work was supported by the French National Research Agency within the framework of the IROCWA project (ANR-19-CE32-0003-01 grant). BS benefited from an ARTS grant from IRD. Authors thank Nelly Le Goic for her advice and help with histological analysis and Barama Djouane (Fallou Galass) and Abdou Karim Houmenou for their availability and help in the field. We also thank Jeanne Elisabeth Diouf for sharing and discussing the subject.
References
- Afinowi M. 1976. The biology of Anadara senilis and Gryphaea (Crassostrea) gasar in West African waters, in: CIFA Tech. Pap. FAO, p. 29. [Google Scholar]
- Barber BJ, Blake NJ. 1983. Growth and reproduction of the bay scallop, Argopecten irradians (Lamarck) at its southern distributional limit. J Exp Mar Biol Ecol 66: 247–256. [CrossRef] [Google Scholar]
- Barman AC, Wong NLWS, Abd Karim MM. 2022. Reproductive cycle of the oyster Crassostrea (Magallana) saidii (Wong and Sigwart, 2021) from Southeast Asia. Aquac Fish. https://doi.org/10.1016/j.aaf.2022.05.007 [Google Scholar]
- Baron J, Clavier J. 1992. Etude des populations de bivalves intertidaux sur le littoral sud-ouest de Nouvelle-Calédonie. [Google Scholar]
- Broom MJ. 1982. Structure and seasonality in a Malaysian mudflat community. Estuar Coast Shelf Sci 15: 135–150. [CrossRef] [Google Scholar]
- Broom MJ. 1983. Gonad development and spawning in Anadara granosa (L.) (Bivalvia: Arcidae). Aquaculture 30: 211–219. [CrossRef] [Google Scholar]
- Broquard C. 2019. Le déterminisme sexuel de l'huître Crassostrea gigas: du phénotype aux facteurs moléculaires sous-jacents. [Google Scholar]
- Camara A, Hardy K, Dioh E, Gueye M, Piqué R, Carré M, Sall M, Diouf MW. 2017. Amas et sites coquilliers du delta du Saloum (Sénégal) : Passé et présent. L'Anthropologie, Patrimoine et changements climatiques depuis un million d'années. Troisièmes Journées Francophones 121: 204–214. [Google Scholar]
- Carré M, Quichaud L, Camara A, Azzoug M, Cheddadi R, Ochoa D, Cardich J, Pérez A, Salas-Gismondi R, Thébault J, Thomas Y. 2022. Climate change, migrations, and the peopling of Sine-Saloum mangroves (Senegal) in the past 6000 years. Quat Sci Rev 293: 107688. [CrossRef] [Google Scholar]
- Chahouri A, Ouchene H, Yacoubi B, Moukrim A, Banaoui A. 2022. Reproductive cycle of two marine sentinel bivalve species (Donax trunculus and Scrobicularia plana) in the Agadir bay, Southern Morocco. Reg Stud Mar Sci 56: 102611. [Google Scholar]
- Debenay JP, Tack DL, Ba M, Sy I. 1994. Environmental conditions, growth and production Anadara senilis (Linnaeus, 1758) in a Senegal lagoon. J Mollus Stud 60: 113–121. [CrossRef] [Google Scholar]
- Descroix L, Sané Y, Thior M, Manga S-P, Ba BD, Mingou J, Mendy V, Coly S, Dièye A, Badiane A, Senghor M-J, Diedhiou A-B, Sow D, Bouaita Y, Soumaré S, Diop A, Faty B, Sow BA, Machu E, Montoroi J-P., Andrieu J, Vandervaere J-P. 2020. Inverse estuaries in West Africa: evidence of the rainfall recovery? Water 12: 647. [CrossRef] [Google Scholar]
- Dia IMM. 2003. Elaboration et mise en oeuvre d'un plan de gestion intégrée: la réserve de biosphère du delta du Saloum, Sénégal [WWW Document]. URL https://www.iucn.org/fr/resources/publication/elaboration-et-mise-en-oeuvre-dun-plan-de-gestion-integree-la-reserve-de (accessed 9.1.23). [Google Scholar]
- Diouf M, Sarr A. 2014. Renforcement de la résilience climatique des communautés et en particulier pour les regroupements de femmes des villages de Dionewar, Niodior et Falia dans le Delta du Saloum au Sénégal (Projet EFA, rapport final.). UCAD, IUPA. [Google Scholar]
- Enriquez-Diaz M, Pouvreau S, Chavez-Villalba J, Pennec ML. 2009. Gametogenesis, reproductive investment, and spawning behavior of the Pacific giant oyster Crassostrea gigas: evidence of an environment-dependent strategy. Aquac Int 17: 491–506. [CrossRef] [Google Scholar]
- Fabioux C, Huvet A, Le Souchu P, Le Pennec M, Pouvreau S. 2005. Temperature and photoperiod drive Crassostrea gigas reproductive internal clock. Aquaculture 250: 458–470. [CrossRef] [Google Scholar]
- Freites L, Montero L, Arrieche D, Babarro J, Saucedo P, Cordova C, García N. 2010. Influence of environmental factors on the reproductive cycle of the eared ark Anadara notabilis (Röding, 1798) In Northeastern Venezuela. J Shellfish Res 29: 69–75. [CrossRef] [Google Scholar]
- Ghribi F, Bello G, Zupa R, Passantino L, Santamaria N, El Cafsi M, Corriero A. 2017. Reproductive and tissue plasticity in Arca noae (Bivalvia: Arcidae). Eur Zoo J 84: 473–487. [CrossRef] [Google Scholar]
- Hadfield A, Anderson D. 1988. Reproductive cycles of the bivalve molluscs Anadara trapezia (Deshayes), Venerupis crenata Lamarck and Anomia descripta Iredale in the Sydney region. Mar Freshwater Res 39: 649. [CrossRef] [Google Scholar]
- Hardy K, Camara A, Piqué R, Dioh E, Guèye M, Diadhiou HD, Faye M, Carré M. 2016. Shellfishing and shell midden construction in the Saloum Delta, Senegal. J Anthropol Archaeol 41: 19–32. [CrossRef] [Google Scholar]
- Howard DW, Smith CS. 1983. Histological techniques for marine bivalve mollusks. [Google Scholar]
- Jahangir S, Siddiqui G, Ayub Z. 2014. Temporal variation in the reproductive pattern of blood cockle Anadara antiquata from Pakistan (northern Arabian Sea). Turk J Zool 38: 263–272. [CrossRef] [Google Scholar]
- Le Loeuff P, Zabi GSF. 1993. Revue des connaissances sur la faune benthique des milieux margino-littoraux d'Afrique de l'Ouest. Troisième partie: discussion et conclusions. [Google Scholar]
- Mahmoud MAM, Yassien MH. 2022. The gametogenic cycle and spawning of the Short-Necked Clam, Paphia undulata Born, 1778 from Timsah Lake, Suez Canal, Egypt (preprint). In Review. [Google Scholar]
- Manjarrés-Villamil AE, Lucero-Rincón CH, Gualteros WO, Cantera-Kintz JR, Gil-Agudelo DL. 2013. Abundancia y Madurez Sexual de Anadara Similis en el Manglar de Luisico, Bahía Málaga, Pacífico Colombiano*. Boletín de Investigaciones Marinas y Costeras − Invemar 42: 215–231. [Google Scholar]
- Massapina C, Joaquim S, Matias D, Devauchelle N. 1999. Oocyte and embryo quality in Crassostrea gigas (Portuguese strain) during a spawning period in Algarve, South Portugal. Aquat Living Resour 12: 327–333. [CrossRef] [Google Scholar]
- Narasimham KA. 1988. Biology of the blood clam Anadara granosa (Linnaeus) in Kakinada Bay. J Mar Biol Assoc India 30: 137–150. [Google Scholar]
- Okera W. 1976. Observations on some population parameters of exploited stocks of Senilia senilis (=Arca senilis) in Sierra Leone. Mar Biol 38: 217–229. [CrossRef] [Google Scholar]
- Pages J, Citeau J. 1990. Rainfall and salinity of a Sahelian estuary between 1927 and 1987. J Hydrol 113: 325–341. [Google Scholar]
- Paixão L, Ferreira MA, Nunes Z, Fonseca-Sizo F, Rocha R. 2013. Effects of salinity and rainfall on the reproductive biology of the mangrove oyster (Crassostrea gasar): implications for the collection of broodstock oysters. Aquaculture 380-383: 6–12. [CrossRef] [Google Scholar]
- Ren J, Marsden I, Ross A, Schiel D. 2003. Seasonal variation in the reproductive activity and biochemical composition of the Pacific oyster (Crassostrea gigas) from the Marlborough Sounds, New Zealand. N Z J Mar Freshw Res 37: 171–182. [CrossRef] [Google Scholar]
- Rodríguez-Jaramillo C, García-Corona JL, Zenteno-Savín T, Palacios E. 2022. The effects of experimental temperature increase on gametogenesis and heat stress parameters in oysters: Comparison of a temperate-introduced species (Crassostrea gigas) and a native tropical species (Crassostrea corteziensis). Aquaculture 561: 738683. [CrossRef] [Google Scholar]
- Sahin C, Düzgüneş E, Okumus İ. 2006. Seasonal variations in condition index and gonadal development of the introduced blood cockle Anadara inaequivalvis (Bruguiere, 1789) in the southeastern black sea coast. Turk J Fish Aquat Sci 6 (2): 155–163. [Google Scholar]
- Saucedo PE, Southgate PC. 2008. Reproduction, development and growth. Pearl Oyster 131–186. [CrossRef] [Google Scholar]
- Schneider CA, Rasband WS, Eliceiri KW. 2012. NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9: 671–675. [CrossRef] [PubMed] [Google Scholar]
- Stephen D. 1980. The reproductive biology of the Indian oyster Crassostrea madrasensis (Preston): I. Gametogenic pattern and salinity. Aquaculture 21: 139–146. [CrossRef] [Google Scholar]
- Sühnel S, José Lagreze-Squella F, Legat JFA, Puchnick-Legat A, Strand Å, Sühnel Lagreze S, Manoel Rodrigues de Melo C. 2023. Effects of salinity on the reproductive cycle of the mangrove oyster Crassostrea tulipa in hatchery conditions. Aquac Res 2023: e7409585. [Google Scholar]
- Suwanjarat J, Pituksalee C, Thongchai S. 2009. Reproductive cycle of Anadara granosa at Pattani Bay and its relationship with metal concentrations in the sediments 9. [Google Scholar]
- Tito De Morais L. 2011. Mise en place d'un plan de gestion pour l'exploitation durable des coquillages des îles du Saloum (Research Report). IRD Dakar. [Google Scholar]
- Toral-Barza L, Gomez ED. 1985. Reproductive cycle of the cockle Anadara antiquata L. in calatagan, Batangas, Philippines. J Coast Res 1: 241–245. [Google Scholar]
- Utting SD, Millican PF. 1997. Techniques for the hatchery conditioning of bivalve broodstocks and the subsequent effect on egg quality and larval viability. Aquaculture 155: 45–54. [CrossRef] [Google Scholar]
- Vázquez E, Woodin SA, Wethey DS, Peteiro LG, Olabarria C. 2021. Reproduction under stress: acute effect of low salinities and heat waves on reproductive cycle of four ecologically and commercially important bivalves. Front Mar Sci 8. [Google Scholar]
- Villanueva MC. 2015. Contrasting tropical estuarine ecosystem functioning and stability: a comparative study. Estuar Coast Shelf Sci 155: 89–103. [CrossRef] [Google Scholar]
- Wilson JH. 1987. Temporal and spatial distribution of Ostrea edulis larvae in Kilkerian Bay, CO. Galway. [Google Scholar]
- Yankson K. 1982. Gonad maturation and sexuality in the West African bloody cockle Anadara senilis (L.). J Mollus Stud 48: 294–301. [CrossRef] [Google Scholar]
- Yoloye V. 1974. The sexual phases of the West African bloody cockle Anadara senilis (L.). J Mollus Stud 41: 25–27. [CrossRef] [Google Scholar]
Cite this article as: Sané B, Diouf M, Jean F, Flye-Sainte-Marie J, Kerhervé M, Fabioux C, Thomas Y. 2023. Reproduction patterns of the bloody cockle Senilia senilis (Linnaeus 1758) in the Sine-Saloum inverse estuary. Aquat. Living Resour. 36: 33
All Tables
Description and classification criteria for the reproductive stages of Senilia senilis.
All Figures
 |
Fig. 1 (A) Location of the Sine-Saloum delta on the map of Senegal and (B) location of the experimental monitoring study sites. |
| In the text | |
 |
Fig. 2 (A) Anatomy of the S. senilis bloody cockle: black lines show the cut section; (B) Microscopic illustration of the transversal cut section. |
| In the text | |
 |
Fig. 3 Photographs of gonadal structures illustrating the 5 stages of maturation identified for males (A-E) and females (F-J) of Senilia senilis: (A) and (F) early gametogenesis (stage 1), (B) and (G) active gametogenesis (stage 2), (C) and (H) ripeness (stage 3), (D) and (I) partial spawning (stage 4), (E) and (J) total spawning or rest (stage 5). Legends: ET empty tubule; H hemocyte; N nucleus; Nov non-mature oocyte; Mov mature oocyte; Rov residual oocyte; Spg spermatogonia; Spc spermatocyte; Spz spermatozoid; T tubule. |
| In the text | |
 |
Fig. 4 Example of surface delimitation of the different maturation stages for one S. senilis histological section. Each color represents a stage: pink corresponds to the stage of ripeness (stage 3), blue to partial spawning (stage 4) and green to total spawning or rest (stage 5). |
| In the text | |
 |
Fig. 5 Proportion of males and females at each sampling site: ns indicates a non-significant difference between males and females (Chi-2 test, p > 0.05); * indicates a significant difference between males and females (Chi-2 test, p < 0.05). |
| In the text | |
 |
Fig. 6 Variation of (A) temperature, (B) salinity, (C) gonad index and (D) frequency of each maturation stage upstream and downstream, measured between March 2021 and March 2022. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


