| Issue |
Aquat. Living Resour.
Volume 31, 2018
|
|
|---|---|---|
| Article Number | 3 | |
| Number of page(s) | 9 | |
| DOI | https://doi.org/10.1051/alr/2017040 | |
| Published online | 23 November 2017 | |
Research Article
Age, growth, mortality and movement patterns of shortjaw tapertail anchovy, Coilia brachygnathus, in the channel connecting Dongting Lake and the Yangtze River in central China
1
The Key Laboratory of Aquatic Biodiversity and Conservation of Chinese Academy of Sciences, Institute of Hydrobiology, Chinese Academy of Sciences,
430072
Wuhan, PR China
2
University of Chinese Academy of Sciences,
100049
Beijing, PR China
3
Yellow River Basin Water Environment Monitoring Center,
450004
Zhengzhou, PR China
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Handling Editor: Richard Nash
Received:
24
March
2017
Accepted:
23
October
2017
Abstract
Shortjaw tapertail anchovy, Coilia brachygnathus, is one of the anchovy species that exclusively lives in freshwaters, which is the dominant species in Dongting Lake. In order to explain its success as a dominant species in Dongting Lake, the basic biological knowledge of this species was studied from July 2013 to July 2015 in the channel connecting Dongting Lake and the Yangtze River. A total of 11 064 samples of C. brachygnathus were collected, and their standard length ranged between 6.8 and 35.4 cm. The length–weight relationship was W = 0.0026 L3.057, and von Bertalanffy growth curve was Lt = 36.41(1 − e−0.28(t+0.8)). The exploitation rate (E) of 0.49, indicated no overfishing on this species. Its movement patterns were highly significantly and positively correlated with the rate of variation in water level, discharge and water temperature. When water level (discharge and temperatures) were rising (mainly March to July), C. brachygnathus migrated into Dongting Lake, peaking in May. When the water level (discharge and temperatures) were falling (mainly October to December), the migratory behaviour changed to exiting Dongting Lake, peaking in October. The present study suggests, fast generation time, low exploitation rate and the river–lake movements (migrations) are the possible reasons that made C. brachygnathus a dominant species in this area.
Key words: Coilia brachygnathus / age / growth / mortality / movement patterns / Dongting lake
© EDP Sciences 2017
1 Introduction
Anchovies are geographically widespread fish species in both marine and freshwater ecosystems. Because of the significant biomass, they are of great economic and ecological importance. Most marine anchovy species, such as the European (Engraulis encrasicolus) and Japanese (E. japonicus) anchovies, have been widely studied (Bacha and Amara, 2009; Islam and Tanaka, 2009). However, few studies are available on freshwater anchovy species (Liu, 2008; Zhang et al., 2013).
Shortjaw tapertail anchovy, Coilia brachygnathus, belonging to the family Engraulidae, is one of the anchovy species that exclusively lives in freshwater environments (Whitehead et al., 1988). It is a small-bodied pelagic fish that mainly occurs in the middle and lower Yangtze River basin, China (Anonymous, 1976; Whitehead et al., 1988; Wu et al., 2015). Previous studies revealed that C. brachygnathus has a high growth rate and short life span, and it can reach a length of 200 mm within 6 months (Tang, 1987; Liu, 2008). C. brachygnathus reach maturity within 5–6 months and spawn once a year from April to July (Liu, 2008). This species is of great importance in freshwater ecosystems as it links low and high levels of the food web, and is important as a predator of zooplankton, shrimp and small fish and as prey for larger fishes and river dolphin (Anonymous, 1976; Luo, 2006; Zhang et al., 2013). It was once considered as a “land-locked” type of anadromous Coilia ectenes, but Wang et al. (2015a) found that the mitogenome of C. brachygnathus displayed a novel gene order arrangement compared with published C. ectenes.
Over the past few decades, fish resources have declined severely in Yangtze floodplain, owing to the river–lake disconnection and overfishing (Xie, 2003; Zhang and Li, 2007). However, in contrast to the decline of many fish species, C. brachygnathus has become one of the dominant species in Dongting Lake and other similar lakes that still connect to the Yangtze River directly (Hu et al., 2011; Ru and Liu, 2013; Wu et al., 2015). This leads to the interesting question of why this species has become dominant.
Research on the life history characteristics, such as growth, mortality and movement patterns, is important for improving current understanding of fish population (Lagler, 1972; Wang et al., 2013; Rolls et al., 2013), thereby facilitating its management and conservation. The main objectives of this study were to: (1) provide estimates of age, growth and mortality parameters for C. brachygnathus in Dongting Lake area; (2) determine the movement patterns of C. brachygnathus between Dongting Lake and the Yangtze River; and (3) analyze the possible reasons that C. brachygnathus becoming a dominant species in Dongting Lake area and give advice to fish resources conversion.
2 Materials and methods
2.1 Study site
Our study was conducted in the only outlet channel connecting East Dongting Lake and the Yangtze River (Fig. 1). It is about 7 km long and 1 km mean width, and the sampling sites was close to the middle reach of the channel. Silt, nutrients, and fishes go freely between Dongting Lake and the Yangtze River through this channel. This site has a very large seasonal fluctuation in water-level, varying by more than 10 m (Dou and Jiang, 2000).
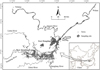 |
Fig. 1 A map of Dongting Lake and the sampling site in the channel connecting Dongting Lake and the Yangtze River. |
2.2 Sample collection
Sampling was carried out monthly from July 2013 to November 2014 and bimonthly from January 2015 to July 2015. In order to reduce possible sampling bias by single size fishing net, six trinal gillnets of three sizes (approximately 100 m length, 2.5 m width, 35, 65, and 85 mm stretch mesh sizes of inner wall) were used in this study. The nets were stretched across the water body for 12 h starting at dusk between 17:00–19:00 h each day, and taken out in the morning. The nets were maintained by two fixed sticks from the west bank to the middle of the channel, and nets were placed more than 200 m from each other. Sampling duration was 3–7 days per month depending on the weather.
When any gillnet was taken out of the water, all entangled C. brachygnathus were counted and their movement direction of entering or existing the lake were judged (see below). The standard length (SL), and weight (W) were recorded (to the nearest 1 mm and 0.1 g). The length–weight relationship of C. brachygnathus individuals was estimated using the equation, W = aSLb, where W is the total weight (g), SL is the standard length (cm), and a and b the constants to be determined. As sexes could not be identified externally, both the sexes were treated together. Student's t-test was applied to verify whether the b value was significantly different from the isometric expected value of 3 (Pauly, 1984). To make the data comparable, the same sampling approach was conducted in all investigation periods. The catches from all trinal gillnets in one month were combined to represent the total catch of each month. Monthly variations of catches were measured with the catch per unit effort (CPUE), which was standardized to the number of individuals catch per day.
Five environmental factors were measured during the sampling period. Water temperature (WT, °C) and dissolved oxygen concentration (DO, mg/L) were measured using a multi-probe logger. Flow velocity (FV, m/s) was measured once a day using an electric flowmeter (LS1206B; Nanjing, China) in front of the nets. The daily water level (WL, m a.s.l) and discharge (DC, m3/s) data were obtained from the Chenglingji Hydrologic Station, which was located near our sampling site. Mean values of variables of each sampling period were used as the value of each month, and the values of the variation rate of these environmental variables was calculated as the difference of the mean value of the adjacent month, which can describe the intensity of the variation of these environmental variables.
2.3 Age and growth
Scales from random samples of 563C. brachygnathus individuals were used for age determination. The scales (cycloid scale) were taken from between the end of the pectoral fin and the beginning of the dorsal fin. Each scale was read twice by one reader using a binocular microscope without prior information on weight, length, or capture time. The first and second counts were performed with an interval of at least two weeks. Only when both counts by the same reader were in agreement, then the counts were accepted. If the estimated numbers of bands differed, the scale was recounted, and the final count was accepted as the agreed number. If the third count did not match either of the previous two counts, the specimen was discarded (Wang et al., 2015b). The number of narrow bands of scale was recorded as annulus (Bagenal and Tesch, 1978) (Fig. 2).
To validate the first growth increment, a scale radius of 25 young-of-the-year (<13 cm) fish specimens was measured. These fish were captured during spring surveys, when the first annulus should be laid down (Campana, 2001). The marginal increment ratio (MIR) was used to examine the periodicity of narrow zone formation in scales. The monthly MIR changes were estimated with the formula: MIR = (R − Rn)/(Rn − Rn−1), where R is the scale radius, Rn is the radius of the last complete zone, and Rn-1 is the radius of the penultimate complete zone (Haas and Recksiek, 1995). FishBC 3.0 was used for measuring the radius. Photographs were taken using the Leica Application Suite (version 15) with a CCD (charge coupled device) connected to the microscope and the computer (Wang et al., 2013). The coefficient of variation (CV) was calculated to estimate the relative precision of age interpretations (Chang, 1982; Campana et al., 1995).
Von Bertalanffy growth curves, based on length-at-age from all age readings, were fitted by non-linear regression: Lt = L∞(1 − e−k(t−t0)), where Lt is the length at age t, L∞ is the asymptotic length, k is the growth coefficient, and t0 is the age at length 0. The growth performance index (Ø) was calculated based on the growth parameter estimates by the equation: ϕ = logk + 2logL∞ (Moreau et al., 1986). The index was used to compare growth parameters obtained in this study with those reported by other authors.
 |
Fig. 2 A scale of Coilia brachygnathus specimen aged 3 (20.9 cm SL). Black line is the axis for measuring the radius and arrows highlight the annuli. |
2.4 Mortality
The total instantaneous annual mortality (Z) was estimated using the length converted catch curve method provided in the FiSAT software (Gayanilo et al., 2005). To obtain an independent estimate of natural mortality rate (M), the empirical equation of Pauly (1980) was used: logM = − 0.0066 − 0.279logL∞ + 0.6543logk + 0.4634logT (Pauly, 1980), here, M = natural mortality; k = growth coefficient; L∞ = asymptotic length (in cm); T = the mean annual temperature (in °C), which is assumed to reflect the local lake surface temperature (in the present study, T = 17.5 °C). The fishing mortality (F) was calculated as F = Z − M, and the exploitation rate (E) was determined as 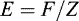 (Ricker, 1975).
(Ricker, 1975).
2.5 Movement patterns
Movement direction for each individual was judged by the orientation of its head in the net (Fernandes, 1997). Two main directions were recorded: (1) entering Dongting Lake and (2) exiting Dongting Lake toward the Yangtze River. In order to describe the principal direction of movement for the whole group, we utilised a “movement index” (MI), which was calculated as: 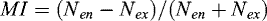 (Zitek et al., 2004; Reichard and Jurajda, 2007), where, Nen is the number of C. brachygnathus individuals entering Dongting Lake; Nex is the number exiting Dongting Lake toward the Yangtze River. The value of MI varies between +1 and −1. The positive value represents the tendency of C. brachygnathus individuals entering Dongting Lake and vice versa. According to the absolute value, the intensity of movement tendencies could be estimated. Pearson rank correlation was used to investigate the correlation of the variation rates of environmental factors on MI, and all statistical analyses were performed using Excel 2010 and SPSS (Version 18.0, SPSS, Inc.).
(Zitek et al., 2004; Reichard and Jurajda, 2007), where, Nen is the number of C. brachygnathus individuals entering Dongting Lake; Nex is the number exiting Dongting Lake toward the Yangtze River. The value of MI varies between +1 and −1. The positive value represents the tendency of C. brachygnathus individuals entering Dongting Lake and vice versa. According to the absolute value, the intensity of movement tendencies could be estimated. Pearson rank correlation was used to investigate the correlation of the variation rates of environmental factors on MI, and all statistical analyses were performed using Excel 2010 and SPSS (Version 18.0, SPSS, Inc.).
3 Results
3.1 Overview
During the two year study period, a total of 11064 C. brachygnathus individuals were captured. In terms of abundance, C. brachygnathus dominated the catches during almost all the study period, with the monthly relative abundance in number of the total catches reached 39.38 ± 17.31% (mean ± SD). All of the C. brachygnathus individuals (6.8–35.4 cm SL, mean ± SD = 16.4 ± 4.3 cm) were measured for length–weight relationship analyses. The relationship between standard length and weight provided a good fit for C. brachygnathus data and the regression equation was described as W = 0.0026SL3.057 (r2 = 0.968, n = 11064). The regression coefficient b was not significantly different from the theoretical value 3 (t-test; P>0.05). Individuals between 10 and 22 cm size (90.70%) dominated the population, with two obvious peaks at 11–12 and 18–19 cm (Fig. 3).
Monthly variations of the relative abundances of C. brachygnathus were observed in our investigated site. High CUPE values occurred two times a year, May–August and October–December in every year (Fig. 4).
 |
Fig. 3 Standard length frequency distributions of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015. |
 |
Fig. 4 CPUE (Catch per Unit Effort) of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015. Vertical bars: standard errors. |
3.2 Age and growth
To determine the first annulus, the young-of-the-year specimens (<13 cm) were sampled from April to June (n = 25). The scales of young-of-the-year samples all showed absence of narrow band, and their scale radius ranged from 1.12 to 1.81 mm (mean ± SD = 1.42 ± 0.17 mm). The total marginal increment ratio (MIR) fell sharply in April, and then increased gradually in the next few months (Fig. 5). Thus, the marginal increment analysis revealed that a single annulus was formed during April–June. In the 563 scales counted twice, 519 scales (92.18%) showed complete agreement; the other 44 scales (7.82%) had only a 1-year difference between the two counts. When the 44 scales were reanalyzed, all were in agreement with one of the counts, and the totals mean CV was 2.16%. Age structure of the C. brachygnathus population was simple, with 1–4 years, and the dominant age groups were 2 and 3 years, representing 34.3 and 44.0% of the total (Tab. 1).
The VBG parameters were estimated as: L∞ = 36.41 cm, k = 0.28 year−1, and age at Zero length was estimated as −0.8 year, which gave the von Bertalanffy growth equation for this species as: Lt = 36.41(1 − e−0.28(t+0.8)) (Fig. 6). The derived growth performance index (Ø) of C. brachygnathus was 2.57.
 |
Fig. 5 Mean monthly marginal increment ratio (MIR, Error bars = SD) of Coilia brachygnathus scales. Sample sizes indicated above the data points. |
Standard length and weight distribution of Coilia brachygnathus in different age groups determined by scales reading.
 |
Fig. 6 Von Bertalanffy growth curve of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River with observed standard length at age estimated from 563 scales readings. |
3.3 Mortality
The total mortality (Z) of C. brachygnathus estimated by the length converted catch curve was 1.17 (with a 95% confidence interval of 1.06–1.27) year−1. While, the estimated natural mortality (M) was 0.59 year−1, the estimated fishing mortality (F) stood at 0.58 year−1. With the values of M and F known, the exploitation rate was then computed as E = 0.49.
3.4 Movement patterns
Movement of C. brachygnathus individuals between Dongting Lake and the Yangtze River appeared to peak twice, as described by the MI. The first movement occurred mainly from March to July, and reached a maximum usually in May, indicating that more C. brachygnathus individuals were moving in than going out of Dongting Lake, especially in May; the second occurred from August to November, and reached the minimum value usually in October, indicating the movement in this period mainly exiting Dongting Lake, especially in October. This phenomenon is very evident in 2014, when the environmental factors fluctuate normally in Dongting Lake. While in late 2013 and early 2014, the environmental factors fluctuated frequently, movement of the C. brachygnathus was not so typical but still showed the similar overall pattern (Fig. 7). Pearson rank correlation indicated that the variations in water level, discharge and water temperature were highly significantly and positively correlated with the MI (Tab. 2). In March, as the water level (discharge and temperatures) began to rise, C. brachygnathus initiated migration in to the lake, and this tendency peaked in May. After that, migratory tendencies entering the lake declined with the lower variation rate of water level (discharge and temperatures). When the water level (discharge and temperatures) began to fall, the migratory tendencies switched from entering to exiting the lake. The migratory tendencies exiting lake increased and peaked usually at October, and then weaken with the lower variation rate of water level (discharge and temperatures) (Fig. 7).
 |
Fig. 7 Movement index (MI) of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015, and water level is given as example to illustrate the relationship with environmental factors. |
Pearson correlation analysis for MI and the variations of environmental factors.
4 Discussion
4.1 Growth parameters
Scales have been widely used for age determination and growth estimation (Campana, 2001; Ye et al., 2014; Wang et al., 2015b). According to Luo (2006), scales are more suitable for age determination in C. brachygnathus than other age determination materials, such as vertebra and otolith. In this study, by analyzing thorough analyses of the MIR, we demonstrated that the annuli on the scales were formed once a year. Moreover, the CV of the two scale readings was 2.16%, lower than the threshold CV (5%) recommended by Campana (2001), with an agreement of 92.18% between the double age readings and suggesting high precision for C. brachygnathus age assessment.
The value of b was not significantly different from 3, indicating an isometric growth of this species (Furukawa et al., 2012). The studies of Luo (2006) and Wu et al. (2015), as well as the present study indicated that the asymptotic length (L∞) of C. brachygnathus is no more than 38 cm, and the similar values of growth parameter (Ø) observed in all studies confirmed the accuracy of our growth estimation from scales reading (Tab. 3). The age range of C. brachygnathus was 1–4 years in Swan Oxbow, in the middle reach of Yangtze River, near Shishou City, China (Luo, 2006), which is similar to this study, while Wu et al. (2015) reported that the age of C. brachygnathus was less than 2 years old in Poyang Lake, the largest freshwater lake in the middle Yangtze River, China. As reported previously, C. brachygnathus can reach a length of 200 mm within 6 months lifespan (Tang, 1987; Liu, 2008), indicating that C. brachygnathus has a high growth rate and short life span. According to a previous analysis of the reproduction of C. brachygnathus, it can reach maturity within 5–6 months, and spawning once a year from April to July (Liu, 2008). In addition, the mean absolute fecundity was 14049.00 ± 7745.00 egg, and the absolute fecundity increases with the increase of body length and body weight (Liu, 2008). This very fast generation time is one of the principal parts of a successful life-history trait (Chang and Cao, 1999; Wang et al., 2013), which makes it easy to adapt to the fluctuating environment (Wang et al., 2017).
In the recent decades, due to the overwhelming human activities, such as overfishing and damming, fish resources have been seriously impacted in the Yangtze River floodplain, especially for the river-lake migratory fish (Wang et al., 2005; Zhang and Li, 2007). Our present results showed that the exploitation rate of C. brachygnathus was 0.49, which was smaller than the proposed E ≈ 0.5 (Elliott, 1983), indicating no overfishing on this species in Dongting Lake area. The result was consistent with Wu et al. (2015), who reported that the exploitation rate of C. brachygnathus was 0.37 in Poyang Lake, another river-connected lake in the Yangtze River floodplain. Reproductive strategy may be one of the most important reasons to ensure the present fish species not overfished. More individuals were produced in short time, keeping the population size at a higher level all the time. Feeding may be another reason that favored the population size of the present fish species. As the main food resources of C. brachygnathus, zooplankton, shrimp and small fish were not affected by the severely overfishing, however the number of predators, such as larger fishes and river dolphin, have declined considerably in this area. In addition, the use of fishing nets with small meshes was banned in our study area, which favored the populations of small-body fish species, such as C. brachygnathus and its prey. In conclusion, the very fast generation time and low exploitation rate, which was probably due to the reproduction and feeding, may be some reasons that made C. brachygnathus a dominant species in this area.
Summary of parameters of von Bertalanffy growth equation for Coilia brachygnathus from different studies.
4.2 Movement patterns
Situated in the subtropical monsoon climate zone, the Yangtze floodplain is characterized by flooding during spring-summer (March–September) coincident with maximum rainfall (Chen and Chang, 1995). In an evolutionary time scale, fishes in this region have evolved life history strategies adapting to the flow regime of the Yangtze River. Particularly, many lakes in the Yangtze floodplain, including Dongting Lake, are connected to the main stream forming the river–lake ecosystem (Wang and Dou, 1998; Ru and Liu, 2013). There are two major types of fishes living in these lakes. One type is the non-migratory fishes such as the common carp and crucian carp, which live and reproduce in the lake (Anonymous, 1976). Another type is the river-lake migratory species such as the four major Chinese carps (i.e. Mylopharyngodon piceus, Ctenopharyngodon idella, Hypophthalmichthys molitrix and Hypophthalmichthys nobilis). These fishes migrate between lakes and rivers, which allows them to use the appropriate habitats for development, growth and reproduction (Wang et al., 2005).
Many methods, such as tagging methods and hydro acoustic methods, were used for the study of movement patterns of fishes (Carvalho, 1983; Skov et al., 2008; Jones and Stuart, 2009). However, these methods were not effective in our study due to unfavorable local conditions, such as the low recapture rates related to high-intensity fishing, the noise fishing boat engine in the channel and too much sand from dredging in Dongting Lake. Considering the purposes of this work, we decided to use trinal gillnets to assess the movement patterns of C. brachygnathus. By using this type of gillnet, it was possible to determine the direction that fishes were moving at the time they were captured (Fernandes, 1997). We chose to put the nets close to the west bank (far away from the residential areas) of the channel during night, thus avoiding human activity such as fishing and the noise from boats in the channel during daytime, which may affect the movement of fish. In addition, the MI was used to describe the directional tendency of fish movement, which can minimize the error of the movement direction determining during one study period.
The results of our study indicate that C. brachygnathus is a river–lake migratory species, for there are two main migratory movements of its individuals in the channel connecting Dongting Lake and the Yangtze River. The first one mainly occurs in March–July and is characterized by the movement of it entering Dongting Lake from the Yangtze River; the second one mainly occurs in October–December, with the opposite movement direction. Almost at the same periods, two main peak values of CPUE were observed in one year. This is a pattern that has been observed in other studies of lateral movements of fish (Fernandes, 1997; Hladík and Kubečka, 2003; Ru and Liu, 2013), which can be considered as a sign of migration.
Previous investigations showed that C. brachygnathus spawn mainly in April–July (Liu, 2008), when it was in the period of entering lake. The lake can provide better environment for eggs and larvae development, and more opportunity for survival. This is an advantage for C. brachygnathus compared to the other river–lake migratory fishes such as the four major Chinese carps, which spawn drifting eggs in the mainstream of the Yangtze River in flooding season. Due to the construction of many dams in the upper Yangtze River, the flow regime has altered in mainstream, thus breeding of these drifting-egg-spawning fish is seriously impacted. After October, with the falling of water level, non-migratory fish in lakes such as the common carp are under huge overfishing pressure. However, the C. brachygnathus just leaves the lake and migrate to the mainstream, where it is under less fishing pressure than the non-migratory fishes in the lake. In addition, C. brachygnathus were shown to ingest different food items during the growth stage (Anonymous, 1976; Zhang et al., 2013). Zooplankton, particularly copepods, is a major component of the diet of small anchovy, whereas larger anchovy individuals fed more on shrimp and fish larvae (Zhang et al., 2013). By the way of migration between the Yangtze River and Dongting Lake, it can use the food resource optimally, which improved the development of its population. Therefore, through the river–lake movement, C. brachygnathus could get better habitats for survival, reproduction and feeding, and thus become a dominant species.
4.3 Fish resource conservation
Overfishing and dam construction were considered as two main causes of the decline of freshwater fish populations in river–floodplain ecosystem (Chang and Cao, 1999; Chen et al., 2002; Habit et al., 2007; Tockner et al., 2010; Yoon et al., 2015). Currently, fishing ban (April–June) is being taken as the major measure for fish resource conservation in our study area. However, after this fishing ban period, fishing pressure increased, thus the fish resources cannot recover. Previous studies showed that the abundance of this fish species has declined even not being dominant in some lakes in the Yangtze floodplain (Anonymous, 1976; Hu et al., 2011). Therefore, based on the present study, we suggest that the fishing ban period should be extended to November at least, when most C. brachygnathus individuals have migrated into river.
Hydrological connectivity between rivers and floodplains is essential for the natural functioning of river–floodplain ecosystems (Copp, 1989; Ward and Stanford, 1995; Amoros and Bornette, 2002) and in the life history of many migratory fishes that have evolved to take advantage of seasonal floods and utilize the inundated areas for spawning and feeding (Agostinho et al., 2007). The movements of C. brachygnathus were highly significantly correlated with the variations in water level, discharge and water temperature. Hence, keeping the free connection between Dongting Lake and the Yangtze River is important for the conservation of this species. We suggest opening sluice gates to the disconnected (isolated) lakes for as long as possible during March–November in order to maximize the opportunities for C. brachygnathus migration.
Acknowledgements
This study was funded by the National Natural Science Foundation of China (NSFC 31400359 and 51509239), and the China Three Gorges Corporation (0704097). We thank Mingzheng Li, Fei Liu and other colleagues for their assistance during field work. Thanks are also to Ondřej Slavík and another two anonymous referees for their critical comments on the manuscript. This study was approved by animal ethic committee of Institute of Hydrobiology, Chinese Academy of Sciences, and was conducted according to “Instructive notions with respect to caring for laboratory animals” issued by the Ministry of Science and technology of the People's Republic of China.
References
- Agostinho CS, Pereira CR, Oliveira RJ, Freitas IS, Marques EE. 2007. Movements through a fish ladder: temporal patterns and motivations to move upstream. Neotrop Ichthyol 5: 161–167. [CrossRef] [Google Scholar]
- Amoros C, Bornette G. 2002. Connectivity and biocomplexity in waterbodies of riverine floodplains. Freshw Biol 47: 761–776. [CrossRef] [Google Scholar]
- Anonymous. 1976. Department of Ichthyology, Institute of Hydrobiology, CAS, Fishes of the Yangtze River, Science Press, Beijing (in Chinese). [Google Scholar]
- Bacha M, Amara R. 2009. Spatial, temporal and ontogenetic variation in diet of anchovy (Engraulis encrasicolus) on the Algerian coast (SW Mediterranean). Estuar Coast Shelf Sci 85: 257–264. [CrossRef] [Google Scholar]
- Bagenal T, Tesch F. 1978. Age and growth. In: Bagenal TB, ed. Methods for assessment of fish production in fresh waters. Oxford: Blackwell Scientific Publication, pp. 101–136. [Google Scholar]
- Campana SE. 2001. Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. J Fish Biol 59: 197–242. [CrossRef] [Google Scholar]
- Campana SE, Annand MC, McMillan JI. 1995. Graphical and statistical method of determining the consistency of age determinations. Trans Am Fish Soc 124: 131–138. [CrossRef] [Google Scholar]
- Chang WB. 1982. A statistical method for evaluating the reproducibility of age determinations. Can J Fish Aquat Sci 39: 1208–1210. [CrossRef] [Google Scholar]
- Chang JB, Cao WX. 1999. Fishery significance of the river communicating lakes and strategies for the management of fish resources. Resour Environ Yangtze Basin 8: 153–157 (in Chinese with English abstract). [EDP Sciences] [Google Scholar]
- Chen YY, Chang JB. 1995. Wetlands loss and alteration of the floodplain in the lower and middle reaches of Yangtze River. In: Chen YY, ed. The study of wetland in China. Jilin: Science and Technology Press, pp. 153–160 (in Chinese). [Google Scholar]
- Chen DQ, Duan XB, Liu SP, Shi WG, Wang B. 2002. On the dynamics of fishery resources of the Yangtze River and its management. Acta Hydrobiol Sinol 26: 685–690 (in Chinese with English abstract). [Google Scholar]
- Copp GH. 1989. The habitat diversity and fish reproductive function of floodplain ecosystems. Environ Biol Fishes 26: 1–27. [CrossRef] [Google Scholar]
- Dou HS, Jiang JH. 2000. Dongting Lake. Hefei: Chinese Science and Technology Publishers ( in Chinese). [Google Scholar]
- Fernandes CC. 1997. Lateral migration of fishes in Amazon floodplains. Ecol Freshw Fish 6: 36–44. [CrossRef] [Google Scholar]
- Furukawa S, Ohshimo S, Tomoe S, Shiraishi T, Nakatsuka N, Kawabe R. 2012. Age, growth, and reproductive characteristics of dolphinfish Coryphaena hippurus in the waters off west Kyushu, northern East China Sea. Fish Sci 78: 1153–1162. [CrossRef] [Google Scholar]
- Gayanilo FC, Sparre P, Pauly D. 2005. FAO-ICLARM stock assessment tools II (FiSAT II) user's guide. Rome: FAO Computerized Information Series (Fisheries). [Google Scholar]
- Haas RE, Recksiek CW. 1995. Age verification of winter flounder in Narragansett Bay Trans Am Fish Soc 124: 103–111. [CrossRef] [Google Scholar]
- Habit E, Belk MC, Parra O. 2007. Response of the riverine fish community to the construction and operation of a diversion hydropower plant in central Chile. Aquat Conserv: Mar Freshw Ecosyst 17. 37–49. [CrossRef] [Google Scholar]
- Hladík M, Kubečka J. 2003. Fish migration between a temperate reservoir and its main tributary. Hydrobiology 504: 251–266. [CrossRef] [Google Scholar]
- Hu ML, Wu ZQ, Liu YL. 2011. Fish diversity and community structure in Hukou area of Lake Poyang. J. Lake Sci. 23: 246–250 (in Chinese with English abstract). [CrossRef] [Google Scholar]
- Islam MS, Tanaka M. 2009. Diet and prey selection in larval and juvenile Japanese anchovy Engraulis japonicus in Ariake Bay, Japan. Aqua Ecol 43: 549–558. [CrossRef] [Google Scholar]
- Lagler KF. 1972. Freshwater fishery biology. Dubuque: Brown. [Google Scholar]
- Liu YL. 2008. Species identification and biological characteristics of Coilia brachynathus in the Poyang Lake, Master Dissertation. Nanchang: Nanchang University ( in Chinese with English abstract). [Google Scholar]
- Luo HB. 2006. Age, growth and gonadal development of Coilia brachygnathus in Swan Oxbow of the Yangtze River, Master Dissertation. Chongqing: Southwest University (in Chinese with English abstract). [Google Scholar]
- Megrey BA. 1989. Review and comparison of the age-structured stock assessment models from the theoretical and applied points of view. Am Fish Soc Symp 6: 8–48. [Google Scholar]
- Moreau J, Bambino C, Pauly D. 1986. A comparison of four indices of overall growth performance, based on 100 tilapia population (Fa. Cichlidae). In Maclean JL, Dizon LB, Hosillo LV, eds. The First Asian Fisheries Forum. Manila: Asian Fisheries Society, pp. 201–206. [Google Scholar]
- Pauly D. 1980. On the interrelationships between natural mortality, growth parameters, and mean environmental temperature in 175 fish stocks. ICES J Mar Sci 39: 175–192. [CrossRef] [Google Scholar]
- Pauly D. 1984. Fish population dynamics in tropical waters: a manual for use with programmable calculators. ICLARM Technical Reports, pp. 1–325. [Google Scholar]
- Quinn TJ, Deriso RB. 1984. Quantitative fish dynamics. USA: Oxford University Press. [Google Scholar]
- Reichard M, Jurajda P. 2007. Seasonal dynamics and age structure of drifting cyprinid fishes: an interspecific comparison. Ecol Freshw Fish 16: 482–492. [CrossRef] [Google Scholar]
- Ricker WE. 1975. Computation and interpretation of biological statistics of fish population. Can. J Fish Aquat Sci Bull 191: 382 [Google Scholar]
- Rockstrom J, Steffen W, Noone K, Persson A. 2009. A safe operating space for humanity. Nature 461: 472–475. [CrossRef] [PubMed] [Google Scholar]
- Rolls RJ, Ellison T, Faggotter S, Roberts DT. 2013. Consequences of connectivity alteration on riverine fish assemblages: potential opportunities to overcome constraints in applying conventional monitoring designs. Aquat Conserv: Mar Freshw Ecosyst 23: 624–640. [CrossRef] [Google Scholar]
- Ru HJ, Liu XQ. 2013. River-lake migration of fishes in the Dongting Lake area of the Yangtze floodplain. J Appl Ichthyol 29: 594–601. [CrossRef] [Google Scholar]
- Tang Y. 1987. On the population dynamics of lake anchovy in Taihu Lake and its rational exploitation. J Fish China 11: 61–72 (in Chinese with English abstract). [Google Scholar]
- Tang WQ, Hu XL, Yang JQ. 2007. Species validities of Coilia brachygnathus and C. nasus taihuensis based on sequence variations of complete mtDNA control region. Biodiver Sci 15: 224–231 (in Chinese with English abstract). [CrossRef] [Google Scholar]
- Tockner K, Pusch M, Borchardt D, Lorang MS. 2010. Multiple stressors in coupled river-floodplain ecosystems. Freshw Biol 55: 135–151. [CrossRef] [Google Scholar]
- Wang SM, Dou HS. 1998. Chinese Lakes. Beijing: Science Press (in Chinese). [Google Scholar]
- Wang HZ, Wang HJ. 2009.Ecological effects of river lake disconnection and restoration strategies in the mid-lower Yangtze River. In: Wang ZY, ed. Ecological management on water and sediment in the Yangtze River basin. Beijing: Science Press, pp.379–396 (in Chinese). [Google Scholar]
- Wang LM, Hu HJ, Wang D. 2005. Ecological impacts of disconnection from the Yangtze River on fish resources of Zhangdu Lake. Resour Environ Yangtze Basin 14: 287–292 (in Chinese with English abstract). [Google Scholar]
- Wang T, Wang HS, Sun GW, Huang D, Shen JH. 2012. Length-weight and length-length relationships for some Yangtze River fishes in Tian-e-zhou Oxbow, China. J Appl Ichthyol 28: 660–662. [CrossRef] [Google Scholar]
- Wang T, Huang D, Zhao Y, Wang H, Hu S, Shen J. 2013. Age, growth and mortality of invasive sharpbelly, Hemiculter leucisculus (Basilewski, 1855) in Erhai Lake, China. J Appl Ichthyol 29: 1279–1285. [Google Scholar]
- Wang SH, Wang BH, Hu ML, Wang F, Wu ZQ. 2015a. The complete mitochondrial genome ofCoilia brachygnathus (Clupeiformes: Engraulidae: Coilinae). Mitochondrial DNA 27: 1–2. [Google Scholar]
- Wang T, Gao X, Wang J, Jakovlić I, Dan S.G, Liu HZ. 2015b. Life history traits and implications for conservation of rock carp Procypris rabaudi Tchang, an endemic fish in the upper Yangtze River, China. Fish Sci 81: 515–523. [CrossRef] [Google Scholar]
- Wang T, Gao X, Jakovlić I, Liu HZ. 2017. Life tables and elasticity analyses of Yangtze River fish species with implications for conservation and management. Rev Fish Biol Fish 27: 255–266. [CrossRef] [Google Scholar]
- Ward JV, Stanford JA. 1995. The serial discontinuity concept: Extending the model to floodplain rivers. Regul River 10: 159–168. [CrossRef] [Google Scholar]
- Whitehead PJP, Nelson GJ, Wongratana T. 1988. FAO Species Catalogue. Vol. 7 Clupeoid Fishes of the World (Suborder Clupeoidei). An Annotated and Illustrated Catalogue of the Herrings, Sardines, Pilchards, Sprats, Anchovies and Wolf-herrings. Part 2: Engraulididae. FAO Fisheries Synopsis (7–2). [Google Scholar]
- Wu B, Fang CL, Fu PF, Xiong XY. 2015. Growth Characteristics of Coilia brachygnathus in the Poyang Lake-Yangtze River Waterway. J Hydroeco 36: 51–55 (in Chinese with English abstract). [Google Scholar]
- Xie P. 2003. Three-Gorges Dam: risk to Ancient Fish. Science 302: 1149–1151. [CrossRef] [PubMed] [Google Scholar]
- Xie P, Chen YY. 1996. Enhance researches and conservation of biodiversity in freshwater ecosystem. Bull Chin Acad Sci 4: 276–281 (in Chinese with English abstract). [Google Scholar]
- Xu ZQ, Ge JC, Hung C. 2009. Taxonomy of shortjaw tapertail anchovy Coilia brachygnathus by jaw length and mitochondrial Cytochrome b gene analysis. J Dalian Fish Univ 24: 242–246 (in Chinese with English abstract). [Google Scholar]
- Ye S, Moreau J, Zeng W, Zhang T, Liu J, Li Z. 2014. Growth and mortality of two small fishes, Toxabramis swinhonis Gunther, 1873 and Hyporhamphus intermedius (Cantor, 1842), in a Yangtze shallow lake (China) assessed by length frequency analysis. J Appl Ichthyol 30: 479–484. [CrossRef] [Google Scholar]
- Yi Y, Yang Z, Zhang S. 2010. Ecological influence of dam construction and river-lake connectivity on migration fish habitat in the Yangtze River basin, China. Proc Environ Sci 2: 1942–1954. [CrossRef] [Google Scholar]
- Yoon JD, Kim JH, Jo HB, Yeom MA, Heo WM, Joo GJ, Jang MH. 2015. Seasonal habitat utilization and movement patterns of the threatened Brachymystax lenok tsinlingensis in a Korean river. Environ Biol Fishes 98: 225–236. [CrossRef] [Google Scholar]
- Zhang TL, Li ZJ. 2007. Fish resources and fishery utilization of Lake Poyang. J Lake Sci 19: 434–444 (in Chinese with English abstract). [CrossRef] [Google Scholar]
- Zhang H, Wu GG, Zhang H, Xie P, Xu J, Zhou Q. 2013. Role of body size and temporal hydrology in the dietary shifts of shortjaw tapertail anchovy Coilia brachygnathus (Actinopterygii, Engraulidae) in a large floodplain lake. Hydrobiology 703: 247–256. [CrossRef] [Google Scholar]
- Zitek A, Schmutz S, Unfer G, Ploner A. 2004. Fish drift in a Danube sidearm-system: I. Site-, inter- and intraspecific patterns. J Fish Biol 65: 1319–1338. [CrossRef] [Google Scholar]
Cite this article as: Qin X, Wang T, Lin P, Wang X, Liu H. 2018. Age, growth, mortality and movement patterns of shortjaw tapertail anchovy, Coilia brachygnathus, in the channel connecting Dongting Lake and the Yangtze River in central China. Aquat. Living Resour. 31: 3
All Tables
Standard length and weight distribution of Coilia brachygnathus in different age groups determined by scales reading.
Summary of parameters of von Bertalanffy growth equation for Coilia brachygnathus from different studies.
All Figures
 |
Fig. 1 A map of Dongting Lake and the sampling site in the channel connecting Dongting Lake and the Yangtze River. |
| In the text | |
 |
Fig. 2 A scale of Coilia brachygnathus specimen aged 3 (20.9 cm SL). Black line is the axis for measuring the radius and arrows highlight the annuli. |
| In the text | |
 |
Fig. 3 Standard length frequency distributions of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015. |
| In the text | |
 |
Fig. 4 CPUE (Catch per Unit Effort) of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015. Vertical bars: standard errors. |
| In the text | |
 |
Fig. 5 Mean monthly marginal increment ratio (MIR, Error bars = SD) of Coilia brachygnathus scales. Sample sizes indicated above the data points. |
| In the text | |
 |
Fig. 6 Von Bertalanffy growth curve of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River with observed standard length at age estimated from 563 scales readings. |
| In the text | |
 |
Fig. 7 Movement index (MI) of Coilia brachygnathus in the channel connecting Dongting Lake and the Yangtze River from July 2013 to July 2015, and water level is given as example to illustrate the relationship with environmental factors. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


